C3: Stochiometry
Element symbols
E.g: H is hydrogen
Na is sodium
K is potassium
Molecular formula of a compound
the number and type of atoms in one molecule
e.g: H2, H2O, NaCl,..
Chemical formulae and equations
Structural formula (simplified): CH3CH2CH2CH3
molecular formula: C4H10
Formulae of compound ions:
Made up of atoms covalently bonded together
Have a negative charge (gained electrons to become stable
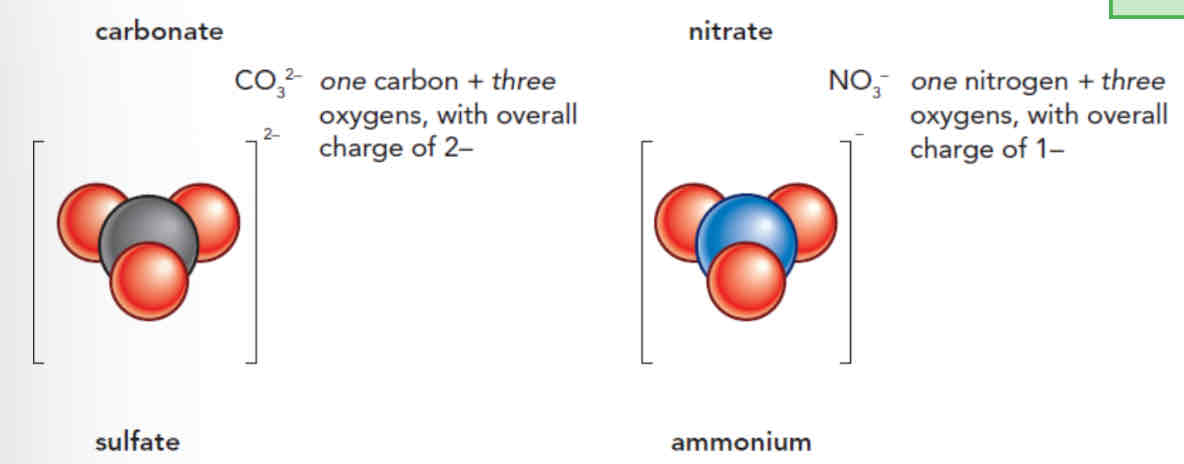
Formulae of compounds:
Group I-IV= group number
Group V-VII = 8 - group number
Group VIII = 0
The cross-over method:

Molecular formula: number of each type of atom present
Formulae of ionic compounds: charges must balance each other
Rules:
Metals and hydrogen = positively charged
Non-metals = negatively charged
Transition elements: charge given in brackets

Relative masses of atoms and molecules
Relative atomic mass of elements: the average mass of the isotopes of an element compared to 1/12th of the mass of an atom of 12C
Molecular mass of compounds
Relative molecular mass (Mr): total masses of atoms/ions in the formula
The mole and the Avogadro constant
Concentration can be measured in g/dm³
The Mole (mol):
unit of amount of a substance
one mole contains 6.02 × 10²3 particles (atoms, ions, molecules) → avogardo constant
Molar mass = mass of one mole of any substance will be equal to the Mr
Equation:

Molar gas volume: 24 dm³ at room temp
mol = V dm³/24 dm³/mol