Carboxylic Acids
Objectives
Determine the physical and chemical properties of carboxylic acids, their derivatives, amines, and amides.
Learn the proper naming conventions for carboxylic acids and esters.
Familiarize with the structures of carboxylic acids and esters.
Identify key chemical reactions involving carboxylic acids and esters.
Carboxylic Acids
A. Structure and Derivatives
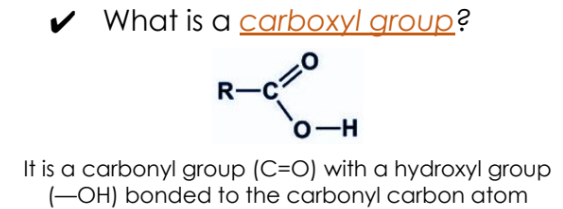
Definition: An organic compound with a carboxyl functional group (-COOH).
Carboxyl Group Structure: A carbonyl group (C=O) with a hydroxyl group (—OH) attached to the carbonyl carbon.
Simplest carboxylic acids contain a hydrogen atom attached to the carbon.
Cyclic carboxylic acids aren’t real
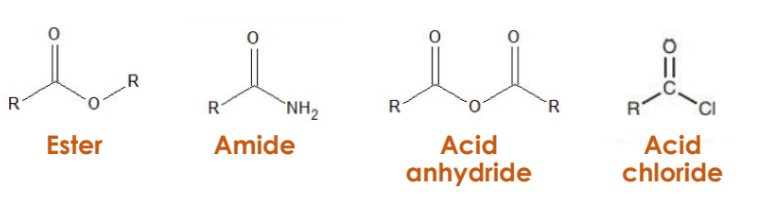
Organic compound can be synthesized or converted in a Carboxylic Acid
B. IUPAC Nomenclature and Common Names
Monocarboxylic Acids
Have one carboxyl group.
Naming Steps:
Locate the carbon of the carboxyl as the parent chain.
Select the longest chain including the carboxyl carbon.
Change the “-e” to “-oic” acid for the parent chain name.
Assign #1 to the carboxyl carbon for numbering.
Note other substituents and number their location according to standard naming rules.
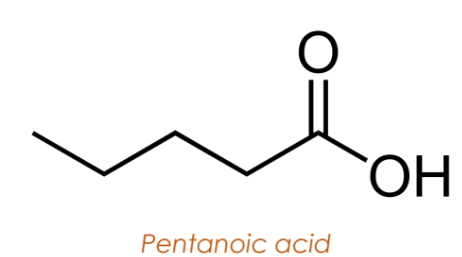
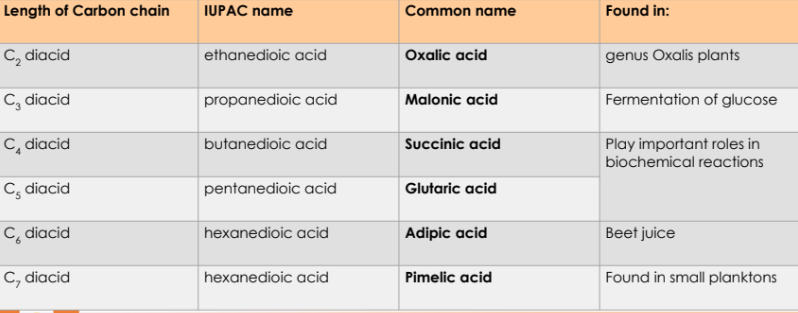
Dicarboxylic Acids
Have two carboxyl groups located at each end of the carbon chain.
Named by appending “-dioic acid” to the alkane name.
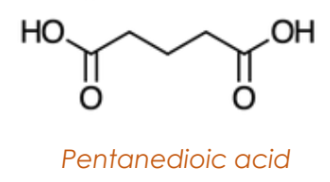
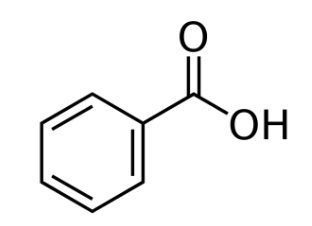
Aromatic Carboxylic Acids
Simplest example: Benzoic Acid.
Derivatives of benzoic acid are named based on substitutions on the aromatic ring.
The carboxyl group is always 1
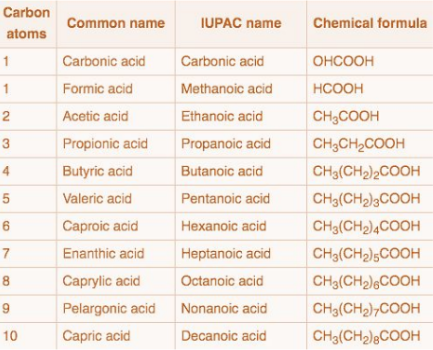
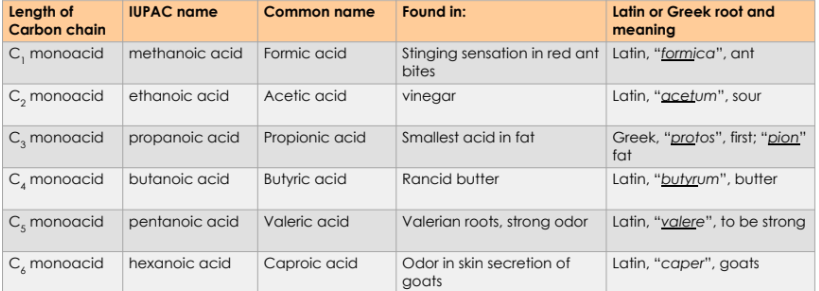
Common names of Carboxylic Acids
Common names is more prevalent for carboxylic acids
Earliest classes of organic compounds studied are abundant
Derived from Latin or Greek words
C. Polyfunctional Carboxylic Acids
Definition: Contain additional functional groups alongside one or more carboxyl groups.
Types:
Unsaturated Acids: Mono: Acrylic acid (Propenoic acid). Di: Maleic acid, and Fumaric acid (Butenedioic acid)
Hydroxy Acids: -OH functional group: Glycolic acid (2-Hydroxyethanoic acid) and Lactic acid (2-Hydroxypropanoic acid)
Keto Acids: e.g., Pyruvic acid (2-oxopropanoic acid).
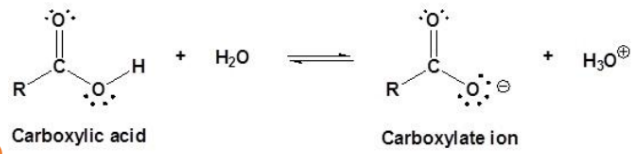
D. Acidity of Carboxylic Acids
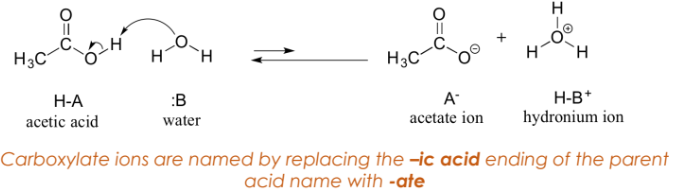
Carboxylic acids are classified as weak acids.
Reaction in water produces hydrogen ions, resulting in hydronium and carboxylate ions.
Carboxylate Ion
Negative ion produced when a carboxylic acid loses one or more acidic hydrogen atoms.
Monocarboxylic acids always carry -1 charge
Dicarboxylic acids possess -2 charge
Physical Properties and Reactions
A. Carboxylic Acids
Physical Properties:
Straight-chain carboxylic acids with 1-9 carbon atoms are liquids with strong odors.
Longer chains (>10 C atoms) are waxy solids without strong odors.
Strong polar groups lead to high melting and boiling points due to H-bonding.
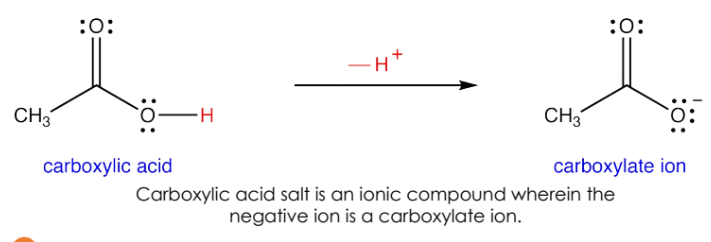
B. Carboxylic Acid Salts
Formed by reaction of carboxylic acids with strong bases.
Naming:
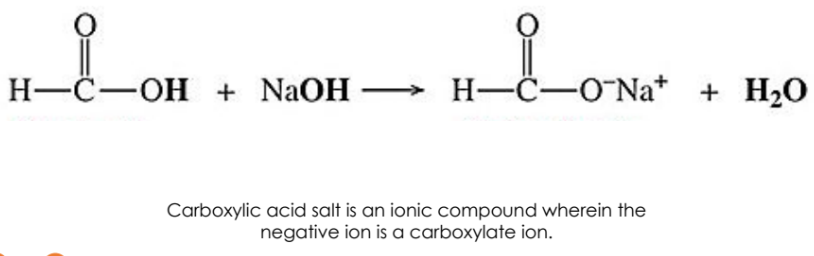
Name the positive ion first.
Use a separate word for the carboxylate ion resultant from the negative carboxylic portion.
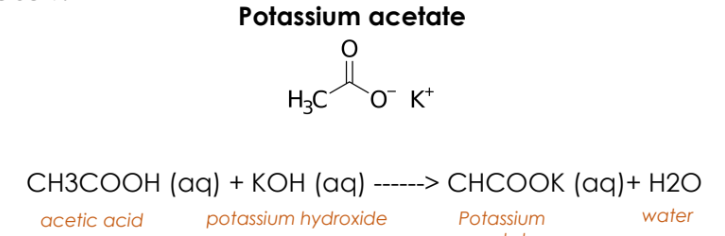
Example: Formic Acid + Sodium Hydroxide ➔ Sodium Formate + Water
C. Naming –COOH salts
Write the chemical equation for the given carboxylic acid salt
Carboxylic acid salts are ionic compounds with the carboxylate ion as the negative part.
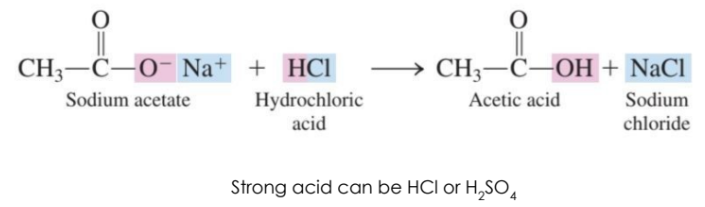
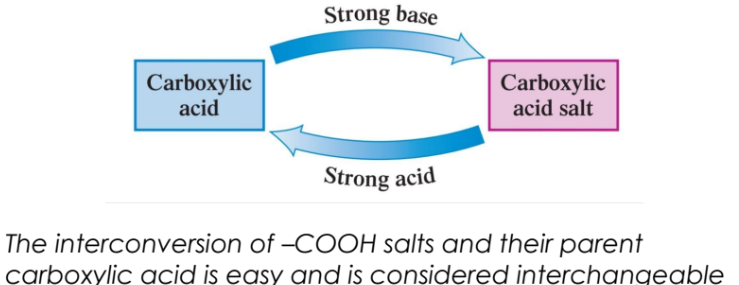
Salts can be converted back to carboxylic acids using a strong acid.
React the salt with a strong acid to convert a –COOH salt back to a carboxylic acid
Uses of Carboxylic Acid Salts
Enhance medication solubility.
Benzoate salts effective against yeast and mold in foods.
Sorbates inhibit microbial growth in perishable products.
Propionates used to preserve baked goods.