Chemistry of Baking
Elements Compounds and Mixtures
Elements and Compounds
2Mg2 + O2 —> 2MgO(s) + CO2 (g)
(element) (molecule) (compound ionic) (compound molecule)
Diatomic:
A lot of non-metals are diatomic
H2, N2, F2, Cl2, Br2, I2
Chemical Element: A neutral substance that cannot be broken down into simpler substances using chemical bonds
Compound: is a pure substance formed when two or more chemical elements combine together. A compound has different properties from its component of elements.
Molecule: They consist only of atoms that contain the same number of protons although the atoms can contain a different number of neutrons. There are 188 known chemical elements.
Mixtures
Mixture: is a physical combination of pure substance which can be separated into its pure components by physical means. Contain more than one element and/or compound that are not chemically bonded so keep their individual properties.
Homogenous mixture: all the components are in the same phase (air, coffee). They have uniform properties
Heterogenous mixture: The components are in different phases (oil and water, sand and salt). Non uniform properties.
Emulsion: Two immiscible mixtures that are turned into a mixture, where one mixture are tiny balls of liquid dispersed in another (milk which is fat and water )
Immiscible: Two mixtures that cannot make a homogenous mixture when mixed together.
Saturation: Maximum solute that a solvent can hold
Separating Mixtures
Fractional distillation: Allows to separate liquids of close temperature. Where the one with higher boiling point will vaporize but then condense back down as it has to travel more compared to normal distillation

Dissolving and evaporate: One use case is to separate salt from sand. Sand is not soluble by water so by dissolving the salt in water and using funnel paper to separate the sand from the water. Then evaporate water and be left with salt
Dissolve and Filter: Separating sand from salt. Mix the salt with water and then filter the solution through filter paper and funnel finally pour mixture into the filter paper rinse with water to dissolve extra salt. Left with only sand in funnel
Distillation: Heating of a liquid and then let it condense it back. Lower boiling point solution rises first and condenses while high boiling points stays in round bottom flask. Example Separating Alcohol from water. Only useful if boiling points are quite far apart
Fractional Distillation: With closer boiling points or more than two solutions. When ethanol boils at 78 water vapor would also go with it contaminating. Meaning that a fractionating column is needed to condense the water vapor and return to round bottom flask
Magnet: For example iron from sulphur use strong magnet to attract the iron from sulphur.
Separation funnel: (immiscible liquids). For example oil and water, oil is at the top water at the bottom, water goes down while oil stays
Chromatography: Separating components of different mixtures. This is because they move differently when in a solution. Such as different dyes, put dyes on chromatography paper, allow solvent to rise and the dyes separate.
Gel electrophoresis: Applying electric fields to separate based on charge and size as they move faster or slower depending on size and charge. Some things that can be separated amino acids move differently than protein so then separated
KMT and States
States
Melting: Solid transforming to liquid (Endothermic)
Freezing: Liquid transforming to solid decrease in potential energy (exothermic)
Evaporation: Phase change from liquid to gas at lower than boiling point, Increase in potential energy. Occurs at the surface molecules to gas. (Endothermic)
Vaporization: Phase change from liquid to gas, increase in potential energy (endothermic)
Crystallization: Liquid to Solid. Saturated solution allow to cool and as it cools and the solute is no longer dissolved. Can be better than evaporation to remove solvent. (exothermic)
Boiling: Gas to liquid. When vapor pressure=external pressure. produces gas bubbles that rises to the top of the liquid. (Endothermic)
Condensing: Gas to liquid (Exothermic)
Sublimation: Solid to gas directly without being in liquid. Example is dry ice, from solid dry ice to CO2 gas directly (Endothermic)
Deposition: Gas to solid without being in liquid. Example is snowflakes, from water vapour to solid snowflakes (Exothermic)
Boiling point of water:
As water heats up some molecules have enough energy to escape the intermolecular forces and turn into a vapor (gas). The gas molecules cause a pressure called the vapor pressure. As the water heats up more and more particles escape the surface of the liquid to become a vapor.
Boiling: Water vapor pressure= (100kPa)=atmospheric pressure =(100kPa)
The lower the pressure, the lower the boiling point for water and vice versa. Example if pressure is 120kPa then boiling pure water boils at 120C.
Kelvin: Increase at the same rate of celcius but starts at absolute zero which is -273C. To convert add or subtract 273
Energy:
When a substance in undergoing a phase change the temperature remains constant which show that there is no change in Ek. The energy changed is Ep due to the change in the inter molecular forces.

Kinetic Molecular Theory (KMT)
Postulates of the Kinetic Molecular Theory:
Gases have a very small volume (if compressed volume is negligible)
Gases are constantly moving at high speeds in random, but in straight line paths (Brownian motion)
Gases experience no electrostatic attractions between molecules.
The speed of a gas molecules is directly proportional to its mass and Temperature
Ek=1/2mv2
Perfectly elastic collisions
These assumptions are good for ideal gases and are used quite often with little error. However, when one needs to be exact you must account for the fact that REAL gases have mass and experience electrostatic attraction.
Temperature and Ek
Temperature (T) is a measure of average kinetic energy (Ek)
Temperature is not Thermal Energy. Thermal
Kelvin+273=Celsius
Celsius-273=Kelvin
Goes up the same rate
Heat goes in and out cold does not go in and out.
Enthalpy change (∆h):
The heat usually transferred either into or out of a system
Is equal to the change of energy of chemical bonds
Is only applied to reactions that occur at standard pressure (100kPa)
Introduction to the mole
Chemical formulae: Letters are symbols for elements
Ionic compounds: For ionic compound put the cation (positive) and then the anion (negative)
Covalent compound: Exact number of each element present in molecule order doesn’t matter
Hydrated compounds: Ionic compounds with water attached (electrostatic attraction) to form a larger compound)
6.02×1023 is Avogadro’s number with he number called a mole. A mole of something equals 6.02×1023 quantity of that thing.
Atomic Mass
Atomic mass
Ratio of the atomic mass per isotopes of an element compared to one twelfth of the mass of the C-12 isotope.
Relative Atomic Mass:
Ratio of the atomic mass per atom of an element compared to one twelfth of the mass of the C-12 isotope.. Dependent on the naturalabundance of different isotopes of an atom.
Empirical and Molecular Formulas
Percentage composition:
It is the percentage by mass of the species in a chemical formula
Empirical formula
is the smallest whole number ratio of atom of each element in a compound
is applicable to both ionic and covalent compounds
Method
% to mass, mass to moles, divided by the smallest ratio, x to make it whole.
Molecular formula:
Is the actual of atoms of each element in the compound
is applicable to only ionic compounds
can sometimes be the same as the emperical formula
is always a whole number multiple of the empirical formula
Therefore the emperical formula is CH2. The molecular formulae are CH2,C2H4
Chemical Equations
Reactant: a substance that changes in a chemical reaction; a substance that is present starting a chemical reaction
Product: a substance that is created by a chemical reaction; a substance present after the reaction
Net ionic equations show only the ions that are involved in the reaction, does not include the spectator ions.
Word equations are equations with chemical symbols and numbers replacing the names of the reactants and products
Neutralization reactions:
An acid and base produce a salt that contains the anion of the acid and the action of the base. monoprotic acids give one hydrogen ion. Diprotic acids like H2SO4 give two hydrogen ions (require two OH-)
Acid- Carbonate Reactions
Acids react with carbonates and hydrogen carbonates in a type of neutralization
Solubility Rules:
Equipment
Number in Image to know : 1,2,3,4,5,6,13,14,16,18,24,26,27,28,29,30
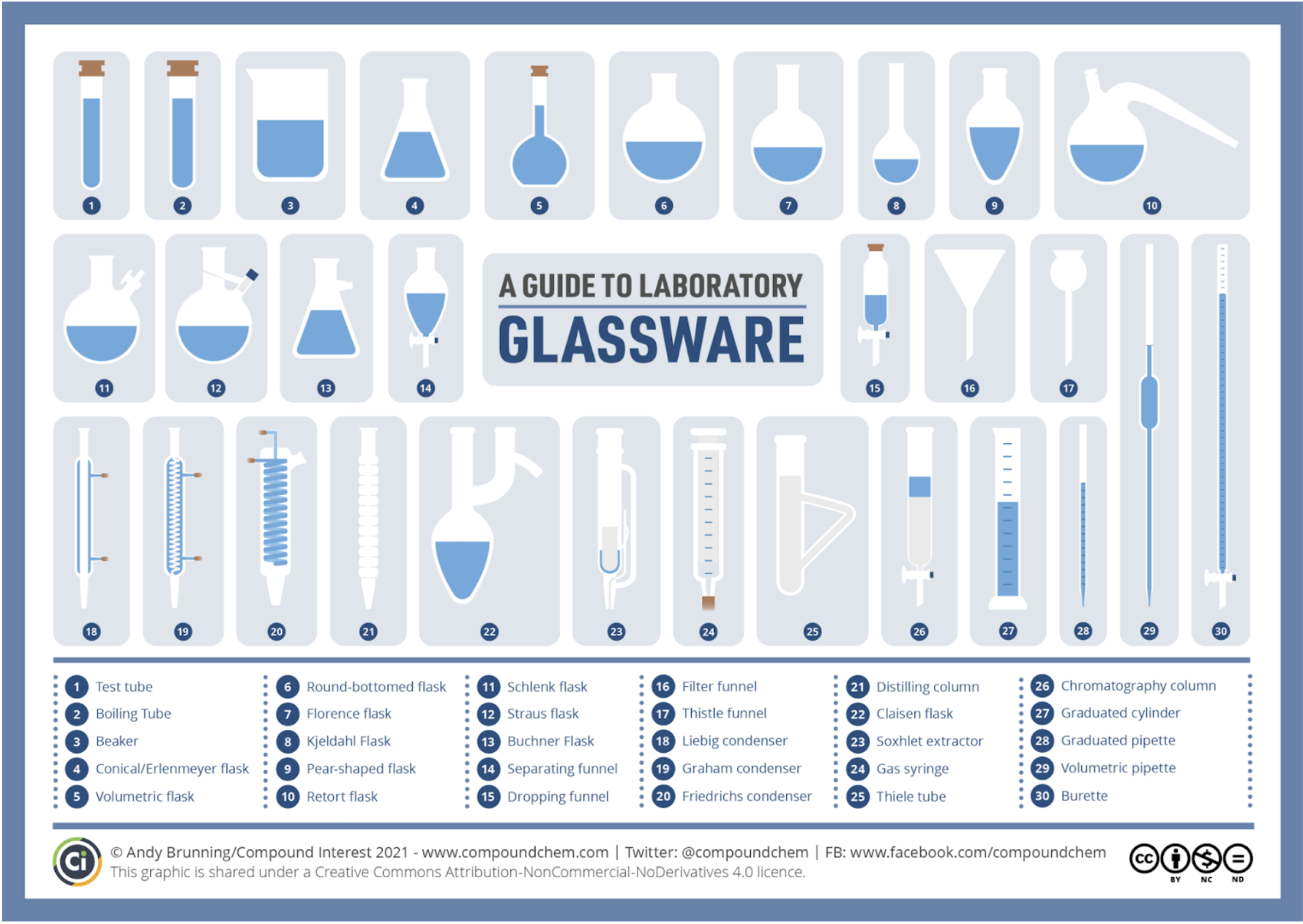
Equipment uncertainty is (smallest measurement)/2 for analog
Digital uncertainty is equal to smallest reading
Sometimes need to use manufactures uncertainty in digital devices
How a standard aqueous solution can be prepared:
Determine the number of moles of solute needed to make desired concentration.
Calculate and then using that measure the amount of mass needed
Add solute to small beaker and add sufficient distilled water to fully dissolve the solute (has to be less than required volume)
Pour the solute and solvent into a volumetric flask
Fill up the volumetric flask up to the line using distilled water (use the beaker to fill
Invert shake volumetric flaks
Molar concentration
Solute: The substance that is dissolved
Solvent: the substance that is in liquid form the substance the solute is dissolved in
Solubility:the amount of a solute that will dissolve in a given volume of the solvent
Saturated a solution in which no more solute will be dissolved at that temperature
Precipitate When a solid is produced in a chemical reaction in a solution it is called y
Concentration (C) is the amount in moles contained with a given volume (V) of a solution
Molarity (molar Concentration) the molarity of a solution is the concentration in moldm-3
Avogrado’s law: At constant temperature and pressure, a volume of gas always has the same number of particles
1 mole =22.7dm3 at STP (0 and 100kPa)
Limiting Reagents
Identifying limiting reagent:
Take the number in the problem and divide by the coefficeint from the balanced reaction
The smallest ratio gives the limiting reagent
Percentage yield
Theoretical Yield: The amount of the product calculated using stoichiometry
Actual Yield the amount of product actually made when the reaction is done in a laboratory
Percent Yield The amount of product actually produced divided by the amount of product that could be produced theoretically (as a percentage)
Atom Economy
Percentage Yield of the mass of product that is desired over total mass of reactants
Exothermic and Endothermic Reactions
Exothermic reaction
∆h is negative
Chemical potential→ heat energy
Energy has been given to the surroundings
Endothermic Reaction
∆h is positive
heat energy → chemical potential
Energy has been absorbed from the surroundings
Activation energy: minimum energy required for a collision reaction to be successful

Enthalpy change: Difference between chemical Ep of Reaction and Products
Calculation of Enthalpy Changes
Enthalpy changes (ΔH) are related to the number of moles in the reaction. If all the coefficients are doubled, then the value of ΔH will be doubled.
When a reaction is carried out in aqueous solution, the object will gain or lose heat from (or to) the reactants.
Therefore, the change in energy (ΔH) can be calculated from Q = m x c x ΔT.
The ΔH value can then be calculated back to find the molar enthalpy change for the reaction.
Calorimeters are used to measure the heat released (Q system) by a chemical reaction.
A device called a calorimeter is used to measure the heat released in a combustion reaction.
Heat absorbed = (mc ΔT) liquid + (mc ΔT) calorimeter
Density of solution=density of water @ 25C
Specific heat capacity of solution is the same as H2O (4.18Kjkg-1K-1)
Hess’s Law
Hess’s law: it doesn’t matter how many seps takes for a reaction to occur, or what those steps are, the enthalpy will be the same.
Usually before reactions are added together, some of them must be reversed and/or multiplied by a factor n in order that they sum to the desired reaction. in this process the rules are:
Whenever you multiply a reaction by n, ∆H for the reaction is also multiplied by n.
If you reverse a reaction, ∆H changes sign.