Ch6c: Gases
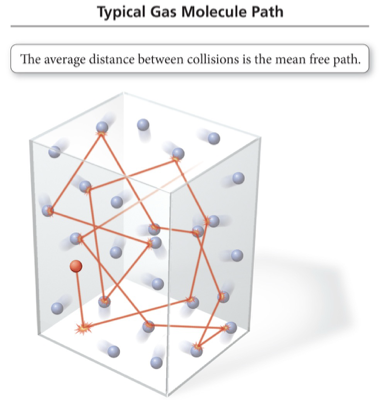
Mean Free Path
Molecules in a gas travel in straight lines until they collide with another molecule or the container.
The average distance a molecule travels between collisions is called the mean free path.
Mean free path decreases as the pressure increases.
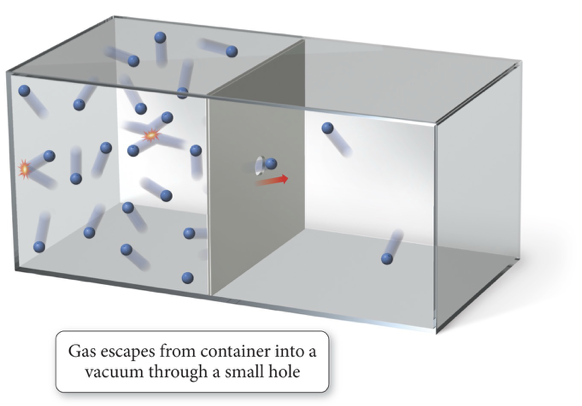
Concepts of Effusion and Diffusion
The process of a collection of molecules spreading out from high concentration to low concentration is called diffusion.
The process by which a collection of molecules escapes through a small hole into a vacuum (low pressure) is called effusion.
The rates of diffusion and effusion of a gas are both related to its rms average velocity.
For gases at the same temperature, this means that the rate of gas movement is inversely proportional to the square root of its molar mass.
Graham’s Law of Effusion
Relation to Root Mean Square Average Velocity:
For gases at the same temperature, the rate of gas movement correlates inversely to the square root of its molar mass.
Equation Involving Diffusion and Effusion:
For two different gases (A and B) at the same temperature, the ratio of their rates of diffusion or effusion is expressed as follows:
RateA/RateB= sqrt MB/MAWhere:
MA = Molar mass of gas A
MB = Molar mass of gas B
Thus, the rate of gas A is inversely proportional to its molar mass (in denominator).
Interpretation:
If the ratio (RateA / RateB) >1, gas A effuses faster than gas B
If the ratio (RateA/RateB) <1, gas B effused faster than gas A.
Practical Example: Uranium Purification
Context:
The example discussed concerns the purification of uranium, crucial for sustaining a chain reaction in nuclear fission to produce energy.
Isotopes of Uranium:
Natural uranium consists primarily of three isotopes:
Uranium-238: 99.2739%
Uranium-235: 0.711%
Uranium-234: 0.005%
Purification Goal:
The objective is to isolate uranium-235 from the mix of isotopes.
Chemical Reaction Leading to Purification:
Uranium is reacted with fluorine to produce uranium hexafluoride (UF6) gas, which occurs at 56 degrees Celsius and 1 atmosphere pressure.
Method for Purification:
The separation of uranium isotopes can be approached using the diffusion method, where UF6 gas is produced. Uranium-235 is slightly lighter than uranium-238, which influences the effusion rates.
Calculating Molar Mass:
Molar mass of uranium-238 UF6: (238+6(19)): 352.04
Molar mass of uranium-235 UF6: (235+6(19)): 349.03
Calculation leads to a ratio calculated as , which estimates to 1.004298
Since this value is greater than one, uranium-235 effuses faster.
Centrifugation (Enrichment of Uranium)
Centrifuge Operation:
Greater centrifugal force enables separation of isotopes based on weight differences, the lighter uranium-235 effusing faster than uranium-238.
Process Description:
When introduced to a gas diffusion setup with low pressure and holes, uranium-235 is enriched while uranium-238 is removed.
Understanding Effusion under Vacuum Conditions
Bottom Line on Effusion:
Effusion necessitates the presence of a vacuum (low pressure) to draw gas through small openings.
Vacuum Conditions Clarification:
Only the space further out is maintained as a vacuum, while the initial gas space remains at high pressure.
Real Gases vs. Ideal Gases
Understanding Real Gas Behavior:
Real gases deviate from ideal behavior predominantly at high pressures and low volumes due to intermolecular attractions and significant molecular volumes.
Ideal Gas Law Limitations:
Ideal gas law assumes:
no attractions between molecules
that gases do not occupy any volume
At low temperatures and high pressures these assumptions are not valid.
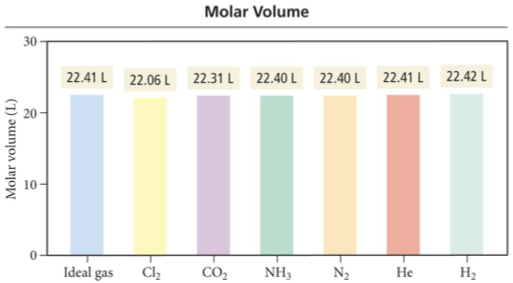
Real Gas Behavior
At low pressures, the molar volume of argon is nearly identical to that of an ideal gas.
But as the pressure increases, the molar volume of argon becomes greater than that of an ideal gas.
At the higher pressures, hence lower volume, the argon atoms themselves occupy a more significant portion of the gas volume, making the actual volume greater than that predicted by the ideal gas law.
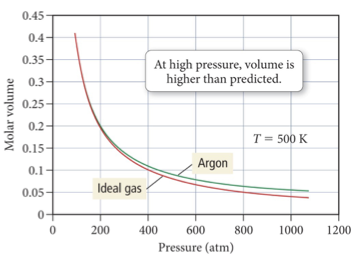
Because real molecules take up space, the molar volume of a real gas is larger than predicted by the ideal gas law at high pressures.
Van der Waals
In 1873, Johannes van der Waals (1837–1923) modified the ideal gas equation to fit the behavior of real gases at high pressure.
The molecular volume makes the real volume larger than the ideal gas law would predict.
Van der Waals modified the ideal gas equation to account for the molecular volume.
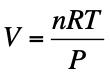

b is called a van der Waals constant and is different for every gas because their molecules are different sizes.
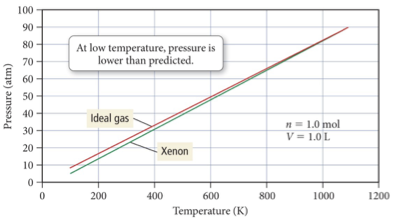
Van der Waals: Low Temperature
At high temperature, the pressure of the gases is nearly identical to that of an ideal gas.
But at lower temperatures, the pressure of gases is less than that of an ideal gas.
At the lower temperatures, the gas atoms spend more time interacting with each other and less time colliding with the walls
The actual pressure less than that predicted by the ideal gas law.
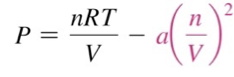
a is another van der Waals constant and is different for every gas because their molecules have different strengths of attraction.
Van der Waals Complete Equation
Combining the equations to account for molecular volume and intermolecular attractions we get the van der Waals equation.
Used for ideal to real

You are taking the measured, real-world gas and applying corrections so that it behaves as if it were ideal — so you can still use the ideal gas law nRT on the right side.
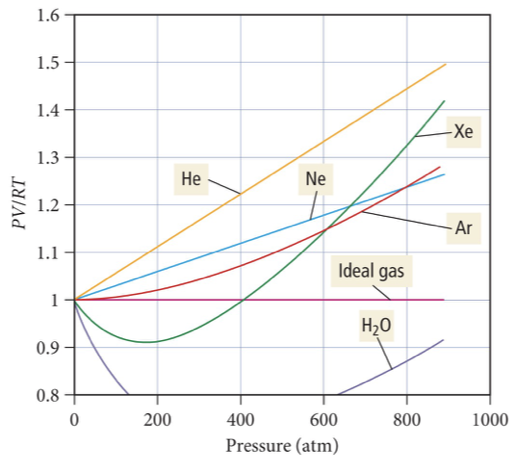
Real Gas Graphs
For 1 mole of gas, the ideal gas law says:
PV=RT
So:
PV/RT = 1
If a gas were perfectly ideal, this value would always equal 1, no matter the pressure or temperature.
So on a graph of PV/RT vs. P, an ideal gas would be a flat line at 1.
🧩 Now for real gases
Real gases deviate because:
They attract each other (intermolecular forces).
They take up space (finite molecular volume).
Both of these make the gas behave non-ideally, but in opposite directions.
🧲 At low pressures — PV/RT < 1
When pressure is low:
The gas molecules are far apart.
The attractive forces between them pull them slightly closer together.
That means they hit the container walls less often → lower pressure than expected for an ideal gas.
Because P is smaller, the ratio PV/RT becomes less than 1.
👉 Main factor: Intermolecular attractions.
🧱 At high pressures — PV/RT > 1
When pressure is high:
The molecules are packed close together.
Their finite size (molecular volume) starts to matter — they can’t compress as much.
This causes the measured volume to be higher than predicted by the ideal gas law.
So PV/RT becomes greater than 1.
👉 Main factor: Molecular volume (repulsions due to size).