States of matter
DEFINITION:
In physics, the state of a matter is one of the distinct forms in which matter can exist. The physical state of a substance depends upon its physical properties.
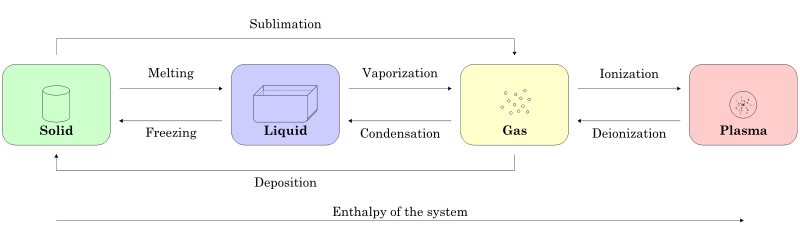
There are four states of matter that are observable in everyday life: Solids, liquids, gases and plasma. Many intermediate states of matter like liquid crystals also exist. The first state of matter is solid and according to the big bang theory the first state of matter is plasma.
PLASMA:
The fourth state of matter is referred to as plasma. In this state, equal amounts of positive and negatively charged particles are present in the substance that are produced when the gas becomes ionized. it does not have a definite shape or volume like gases. The presence of negative particles in the form of free electrons makes plasma electrically conducting medium. gases are converted to plasma in two ways:-
- by applying high voltage between two points
- by exposing gases to extremely high temperatures
this heating causes the electrons to leave the atoms creating positive hole on the atoms.
plasma occurs in mostly in stars like the sun and in interstellar space. aurora, lightning, some types of flames, neon and fluorescent bulbs ,etc.
LIQUID CRYSTAL:
DEFINITION AND USES:
The state of matter which is an intermediate between solid crystals and liquids is called liquid crystal. this matter can flow but exhibits crystal symmetries. the liquid crystal state exists between two temperatures i.e. melting temperature and clear temperature. they have properties of liquid like surface tension and viscosity within a certain temperature range.
- they are used as temperature sensors. this is because they change their color with change in their temperature
- they are used to measure temperature changes.
- used to locate veins and tumuors
- they are use to find the point of potential failure in micro circuits.
- used to display letters and numbers in electrical devices like digital watches and calculators
Liquid Crystal
- Liquid crystal is a state of matter that has properties of both liquids and solids.
- It is a type of material that has a molecular structure that is somewhere between that of a liquid and a solid.
- Liquid crystals are used in a variety of applications, including displays for electronic devices such as televisions, computer monitors, and mobile phones.
- They are also used in thermometers, mood rings, and other types of sensors.
- Liquid crystals are made up of long, thin molecules that are arranged in a regular pattern.
- When a voltage is applied to the liquid crystal, the molecules change their orientation, which changes the way light passes through the material.
- This property is used in liquid crystal displays (LCDs) to create images on a screen.
- LCDs are made up of two layers of glass with a layer of liquid crystal material in between.
- When a voltage is applied to the liquid crystal, it changes the orientation of the molecules, which allows light to pass through or blocks it, creating the image on the screen.
- Liquid crystals have a number of advantages over other types of displays, including low power consumption, high contrast, and a wide viewing angle.
- They are also lightweight and thin, making them ideal for use in portable devices such as laptops and smartphones.
GASES:
Gas is the state of matter that has neither a fixed volume nor a fixed shape at a particular temperature. The varying shape and volume adapts to fit its container .A liquid may be converted to a gas by heating at constant pressure to the boiling point, or else by reducing the pressure at constant temperature.
properties of gases:
indefinite volume and shape. they diffuse to fill the volume of the container available to them.
the gas molecules have enough kinetic energy that the effect of intermolecular forces is zero. there are no attractive or repulsive forces between the molecules.
the molecules are far away from each other. the distance between the molecules is much greater than the size od the molecule.
Gas has low density
diffusion is the movement of molecules from a region of higher concentration to lower concentration. diffusion occurs in gases because they move freely.
the escaping of a gas molecule through a small hole. effusion occurs in gases because there is no intermolecular forces binding the molecules to each other and they can thus escape freely from the hole one at a time.
compression: because there are large spaces between the atoms applying pressure brings the atoms colser together and thus gases can be compressed. volume of gas decreases.
expansion: The thermal expansion of gases is a change in their volume in response to a change in temperature. Heating a gas increases the kinetic energy of the particles, causing the gas to expand. The thermal expansion of gases is larger than that for solids and liquids
there is no effect of gravity on a single gas molecule
gases kinetic energy depends on temperature. increasing temperature.
The Ideal Gas Law is a fundamental equation in thermodynamics that describes the behavior of an ideal gas. It states that the pressure, volume, and temperature of an ideal gas are related by the equation PV = nRT, where P is the pressure, V is the volume, n is the number of moles of gas, R is the gas constant, and T is the temperature in Kelvin.
Ideal Gas Law
The ideal gas law is a fundamental equation in thermodynamics that describes the behavior of gases under various conditions. It is expressed as:
PV = nRT
Where:
- P is the pressure of the gas
- V is the volume of the gas
- n is the number of moles of gas
- R is the gas constant
- T is the temperature of the gas
Gas Constant
The gas constant, R, is a constant that relates the properties of a gas to its temperature, pressure, and volume. Its value depends on the units used for pressure, volume, and temperature. The most commonly used value of R is 8.314 J/(mol·K).
Units
The units used for the variables in the ideal gas law depend on the system of units used. In SI units, pressure is measured in pascals (Pa), volume in cubic meters (m³), temperature in kelvins (K), and the number of moles in moles (mol).
Limitations
The ideal gas law is only applicable to ideal gases, which do not exist in reality. Real gases deviate from ideal behavior at high pressures and low temperatures. To account for these deviations, various modifications to the ideal gas law have been proposed, such as the van der Waals equation.
The ideal gas law is a fundamental equation in thermodynamics that describes the behavior of gases under various conditions. It is a combination of Boyle's law, Charles's law and Avogadro's law. Boyle's law states that the pressure of a gas is inversely proportional to its volume at a constant temperature, while Charles's law states that the volume of a gas is directly proportional to its temperature at a constant pressure. Avogadro's law states that the volume of a gas is directly proportional to the number of moles of gas at a constant temperature and pressure.
The ideal gas law equation, PV = nRT, relates the pressure, volume, temperature, and number of moles of an ideal gas. The pressure of the gas is directly proportional to the number of particles in the gas and their average kinetic energy. The volume of the gas is the space that the gas particles occupy. The temperature of the gas is a measure of the average kinetic energy of the gas particles. The number of moles of gas is a measure of the amount of gas present.
The gas constant, R, is a constant that relates the properties of a gas to its temperature, pressure, and volume. Its value depends on the units used for pressure, volume, and temperature. The most commonly used value of R is 8.314 J/(mol·K). The units used for the variables in the ideal gas law depend on the system of units used. In SI units, pressure is measured in pascals (Pa), volume in cubic meters (m³), temperature in kelvins (K), and the number of moles in moles (mol).
The ideal gas law assumes that the gas is composed of a large number of small particles that are in constant random motion. It also assumes that the particles are not subject to any intermolecular forces, except for perfectly elastic collisions with each other and with the walls of the container. These assumptions are valid for most gases at low pressures and high temperatures.
The ideal gas law is used in many areas of science and engineering, including chemistry, physics, and engineering. In chemistry, the ideal gas law is used to calculate the behavior of gases in chemical reactions. In physics, the ideal gas law is used to study the behavior of gases in different conditions. In engineering, the ideal gas law is used to design and optimize gas-based systems, such as engines and turbines.
However, the ideal gas law is only applicable to ideal gases,