Lecture 2 - Organic Chemistry Rev CME 2024
Alkanes and Their Properties
Introduction to Alkanes
Definition of Alkanes
Compounds with only C-C single bonds and C-H bonds (no functional groups).
Can form large or small molecules depending on carbon connections.
General formula: CnH2n+2 (no rings).
Saturated with hydrogen (no additional hydrogen can be added).
Also referred to as aliphatic compounds.
Basic Structures of Alkanes
Examples of Alkanes
Methane (CH4)
Ethane (C2H6)
Propane (C3H8)
Butane (C4H10)
Naming Alkanes
List of Straight-Chain Alkanes
Methane: CH4
Ethane: C2H6
Propane: C3H8
Butane: C4H10
Pentane: C5H12
Hexane: C6H14
Heptane: C7H16
Octane: C8H18
Nonane: C9H20
Decane: C10H22
Properties of Alkanes
Types of Alkanes
Straight-chain (normal) alkanes: C's connected to no more than 2 other C's.
Branched-chain alkanes: C's connected to 3 or 4 other C's.
Types of Carbons in Alkanes
Degree of Substitution
Primary (1°): 1 carbon connected.
Secondary (2°): 2 carbons connected.
Tertiary (3°): 3 carbons connected.
Quaternary (4°): 4 carbons connected.
Types of Hydrogens in Alkanes
Classification of Hydrogens
Primary hydrogens (CH3).
Secondary hydrogens (CH2).
Tertiary hydrogens (CH).
Constitutional Isomers
Definition
Isomers that differ in the arrangement of atoms in chains.
Must have the same molecular formula to be considered isomers.
Isomers
Constitutional Isomers
Isomers that differ in arrangements of chains but the same molecular formula
Compounds other than alkanes can be constitutional isomers of one another
Usually have different physical properties
Types of Isomers
Straight-chain and branched-chain forms (each with 4 carbon atoms) of butane (C4H10).
Physical Properties of Alkanes
Trends in Physical Properties
Boiling and melting points increase with the size of the alkane.
Dispersion forces increase with molecular size, leading to higher melting and boiling points.
Reactions of Alkanes
Combustion
Called paraffins (low affinity compounds) because they do not react as most chemicals.
Alkanes burn in a flame, producing CO2, H2O, and heat.
Non-polar and insoluble in water but soluble in non-polar solvents.
Radical Halogenation
They react with Halogens like Cl2 in the presence of light replaces H’s with Cl’s (not controlled)
Sources of Alkanes
Primary Source
Petroleum, a complex mixture of alkanes and aromatic hydrocarbons.
Distillation of Petroleum
Distillation
First step in refining petroleum.
Separation Process
Components are separated based on volatility.
More than 500 different compounds can be obtained from petroleum distillates.
Boiling Points
<30°C: Natural gas; (C1-C4)
30°C-200°C: Gasoline; (C5-C10)
200°C-250°C: Kerosene, jet fuel; (C12-C18)
250°C-350°C: Diesel fuel, heating oil; (C12<)
350°C-450°C: Lubricating oil; Nonvolatile liquids; (C20<)
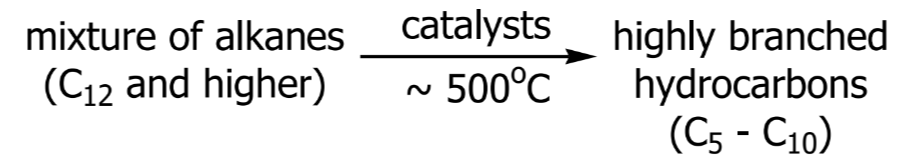
Gasoline and Catalytic Cracking
Gasoline Production
The demand of gasoline is much greater than that supplied by the gasoline fraction of petroleum.
Catalytic Cracking
Converting hydrocarbons from other fractions of petroleum into gasoline.
Naming Alkanes
Nomenclature Rules
Prefix-Locant-Parent-Suffix structure.
Identify the longest carbon chain and number the carbons.
Substituents are identified numbered
Write compound name (single word)
Name a complex substituents as though it was a compound itself.
IUPAC Names for Simple Alkanes
Naming Conventions
All alkane names end with -ane.
Derived from Greek and Latin roots.
one - meth
two - eth
three - prop
four - but
five - pent
Alkyl Groups
Definition
Alkyl group: remove one H from an alkane.
General abbreviation “R”.
Nomenclature of Branched-Chain Alkanes
Naming Process
Use the longest continuous carbon chain as the parent name.
Start numbering from the end nearest the first substituent.
Use the number obtained by Rule 2 to designate the location of the substituent.
For two or more substituents, use the lowest possible individual numbers of the parent chain. The substituents should be listed alphabetically. In deciding alphabetical order, disregard multiplying prefix, such as “di”, “tri” etc.
When two substituents are present on the same carbon, use that number twice.
For identical substituents, use prefixes di-, tri-, tetra-, so on.
When two chains of equal length compete for selection as parent chain, choose the chain with the greater number of substituents.
When branching first occurs at an equal distance from either end of the longest chain, choose the name that gives the lower number at the first point of difference.
Conformations of Ethane
Stereochemistry
Concerned with 3-D aspects of molecules.
Sigma bonds are cylindrically symmetrical
Rotation around C-C bonds is possible in open-chain molecules.
Conformers
Conformation
Different arrangement of atoms resulting from bond rotation.
Sawhorse representation
Newman projection.
Torsional Strain
Stability of Conformers
Staggered conformations are most stable; all 6 C-H bonds are as far away as possible.
Eclipsed conformations are least stable; all 6 C-H bonds are as close as possible.
Conformations of Butane
Types of Conformations
Anti conformation: methyl groups are 180˚ apart.
Gauche conformation: methyl groups are 60˚ apart.
Energy Costs for Interactions in Alkane Conformers
Interaction Energies
Torsional strain and steric strain contribute to energy costs in conformers.

Steric Strain
Steric Strain
Repulsive interaction occurring between atoms forced closer than their atomic radii allow.