Introduction to Organic Chemistry Part 2
Carboxylic Acid
Weak acid
Responsible for the tart flavour of sour-tasting foods
Ethanoic/acetic acid: the main ingredient found in vinegar
Botanic acid: partially responsible for the locker/ unwashed sock smell
Boiling Point
They have a higher boiling point than alcohol alcohols because they can form hydrogen-bonded dimers. This occurs when two carboxylic acid molecules interact through hydrogen bonding between the hydroxyl group of one molecule and the carbonyl group of another. These dimers are more stabilized than individual molecules, resulting in stronger intermolecular forces, and consequently, having a higher boiling point than alcohols, which cannot f0orm such stable timers
Solubility
The solubility of carboxylic acid in water depends on the size of the molecule. Smaller molecules, five carbon atoms or fewer, are soluble in water.
Chemical Properties: Acidity
Differences in acid strengths carboxylic acids, phenols and alcohols
Two of the factors which influence the ionization of an acid are:
the strength of the bond being broken.
the stability of the ion being formed
For example in the carboxylic acid, ethanoic acid. The molecule is able to lose the H+ ion to form an ethanoate ion, the oxygen that was bonded to the hydrogen now becomes negatively charge. The negative charge is delocalized across the two oxygen atoms. The more you can spread the charge around, the more stable an ion becomes.
Carboxylic Acid is more acidic than phenols
When the hydrogen-oxygen bond in phenol breaks, you get a phenoxide ion. In the phenoxide ion, the single oxygen atom is still the most electronegative atom, and the delocalized system will be heavily distorted towards it. Delocalization makes the phenoxide ion more stable than it would otherwise be, and phenol is acidic to an extent. However, the delocalization did not share the charge around very effectively. As a result the negative charge is around the oxygen to which hydrogen ions will be attracted and so the phenol will readily reform. Phenol is therefore only very weakly acidic.
Phenols are more acidic than ethanol
Variation in Strengths between Carboxylic Acids
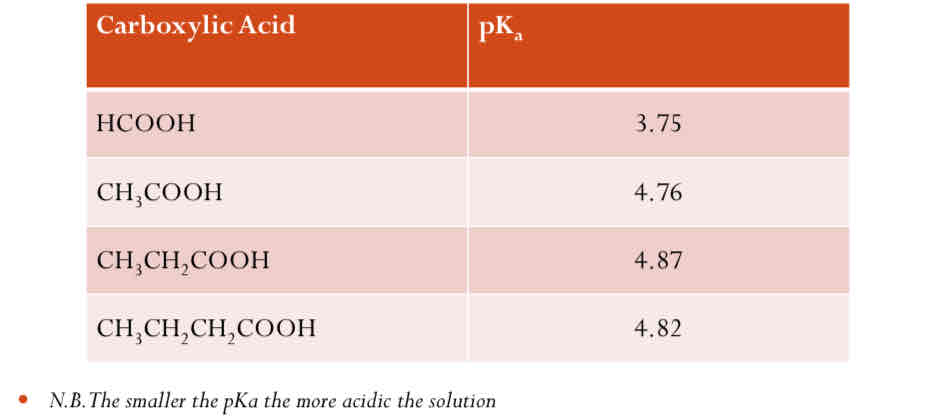
Alkyl groups are electron-donating groups. This means they push electron density toward the carboxyl group (-COOH), making the electron density on the oxygen atoms of the carboxylate anion (-COO⁻) slightly higher.
The increased electron density on the oxygen atom results in a reduced ability to stabilize the negative charge on the conjugate base (the carboxylate anion). This destabilization makes the conjugate base less stable and makes it more likely to recombine with a proton (H⁺), which in turn makes the acid weaker.
The bigger the alkyl group, the weaker the acid.
Comparing the effect of alkyl groups on the acidity of Carboxylic Acid
Strengthening the Acidity of Carboxylic Acids
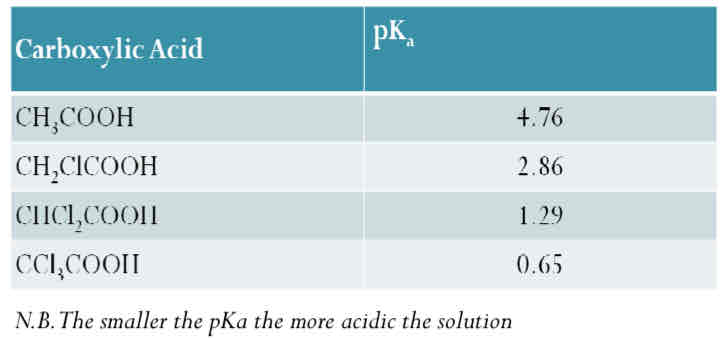
The acidity can be strengthened by pulling charge away from the -COO-end This can be done by attaching an electronegative atom (halogen) to the chain.
The effect of an electronegative atom on the acidity of the carboxylic acids
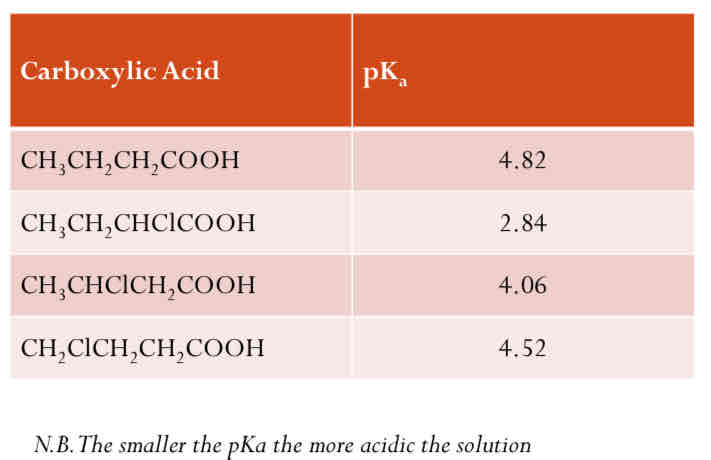
Position of the Electronegative Atom
The further the chlorine atom is the lesser the effect on the –COO-
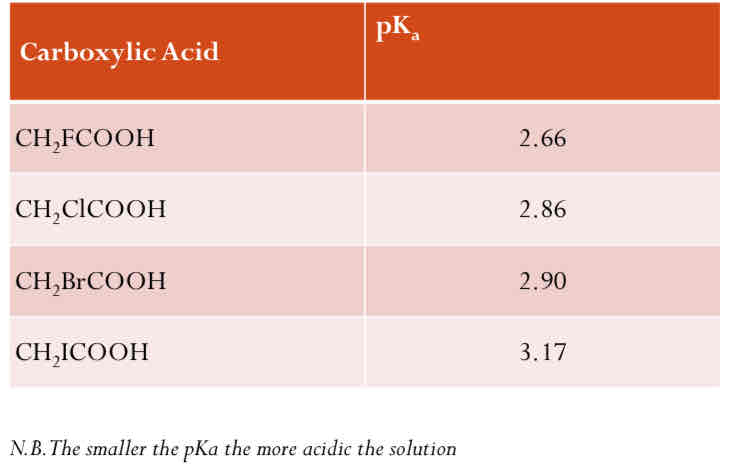
Other Halogens
Attaching different halogens also makes a difference Fluorine is the most electronegative therefore it is most successful at pulling charge away from the -COO-end and so strengthening the acid.
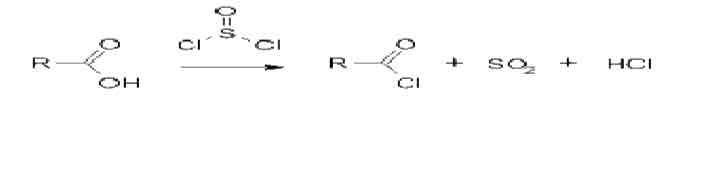
Reaction with thionyl chloride: formation of acid chloride
The thionyl chloride reacts with carboxylic acids to produce an acyl chloride Sulphur dioxide and hydrogen chloride gases are given off as byproducts. When a carboxylic acid reacts with thionyl chloride, it undergoes a substitution reaction where the hydroxyl group (-OH) of the carboxylic acid is replaced by a chlorine atom (Cl), forming an acyl chloride. The reaction also produces two gases as byproducts: sulfur dioxide (SO₂) and hydrogen chloride (HCl).
Mechanism:
Activation: Thionyl chloride reacts with the carbonyl group (C=O) of the carboxylic acid, and the sulfur atom bonds to the oxygen of the hydroxyl group (-OH), activating it for nucleophilic attack.
Formation of Acyl Chloride: The -OH group is eliminated as water (H₂O) and replaced by a chlorine atom, forming the acyl chloride (R-COCl).
Byproducts: The elimination of the hydroxyl group as water leads to the formation of SO₂ and HCl as byproducts.
For instance, if acetic acid (CH₃COOH) reacts with thionyl chloride, the reaction would be:
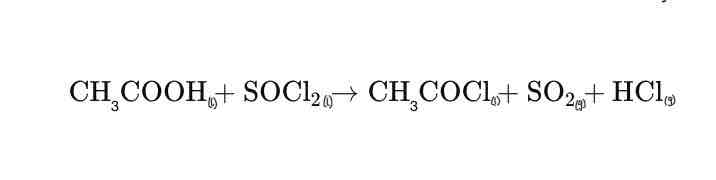
 A
A