P.7 - Radioactivity
Atoms and Radiation
Some atomic nuclei are unstable. The nucleus gives out radiation as it changes to become more stable. This is a random process called radioactive decay
Activity is the rate at which a source of unstable nuclei decays. Activity is measured in becquerel (Bq)
Radiation can be detected using an instrument called a Geiger Counter
Count-rate is the number of decays recorded each second by a detector
Could be unstable by:
extra neutrons
extra protons
excess energy
Types of Nuclear radiation
Alpha particle (α) - this consist of 2 neutrons and 2 protons, same as helium
Beta particle (β) - a high speed electron ejected from the nucleus as a neutron turns into a proton
Gamma ray (γ) - electromagnetic radiation emitted from the nucleus
Neutron
Atomic and Mass Number
Atomic number — the number of protons inside the nucleus of an atom
Mass number — the number of protons and neutrons in the nucleus of an atom
Changes in the Nucleus
Atoms are very small, having a radius of about 1 × 10-10 meters
Most of the of an atom is concentrated in the nucleus
The radius of a nucleus is less than 1/10,000 of the radius of an atom
All atoms of a particular element have the same number of protons
Atoms have no overall electrical charge
I an atom, the number of electrons is equal to the number of protons in the nucleus
Atoms turn into ions if they lose or gain outer electron/s
Atoms of the same element can have a different number of neutrons, these atoms are called isotopes of that element
In a nuclear equation, an alpha particle may be represented by the symbol: 42He
In a nuclear equation, an beta particle may be represented by the symbol: -10e
Discovery of the Nucleus
Plum Pudding Model
Overall charge is neutral
Number of positive and negative charges are equal
Electrons were in a sea of positive charge
Alpha - scattering experiment
Most particles passed straight through
Some particles passed through at different angles
Some particles deflected back
Electrons can move up an orbit by absorbing electromagnetic radiation
Electrons move down an orbit by emitting electromagnetic radiation
The electromagnetic radiation is a photon
Alpha, Beta, Gamma
Types of Radiation | Notation | What is it? | Ionising power | Penetrating power | Stopped by? | range in air |
Alpha | 24He | 2 neutron and protons, same as helium | High | Low | Paper | 5 cm |
Beta | -10e | high speed electron ejected from the nucleus as a neutron turns into a proton | medium | medium | aluminium sheets around 5mm | 1 metre |
Gamma rays | 00γ | electromagnetic radiation emitted by the nucleus | low | High | lead, several cm thick | infinite |
Radioactive contamination is the unwanted presence of material containing radioactive atoms on other materials
Irradiation is the process of exposing an object to nuclear radiation. The irradiation object does not become radioactive
It is important for the findings of studies into the effect of radiation on humans to be published and shared with other scientists so that the findings can be checked by peer review
Activity and Half-Life
Activity- number of nuclei that decay per second — activity = number of nuclei decayed / time
Count rate - number of counts per second from a Geiger Counter — count rate = number of counts from Geiger Counter / time
Half life (1) - time taken for the count rate to reduce to half of it’s initial value
Half life (2) - time taken for the number of un-stable radioactive isotope nuclei to half
Half life (3) - time taken for mass of unstable radioactive isotope nuclei to half
Nuclear radiation in medicine
Nuclear radiations are used in medicine for the exploration of internal organs and control or destruction of unwanted tissue
Medical use | How does it work? | Type of radiation | Why is that type of radiation used? |
exploration of internal organs (medical tracer) | radioisotopes is injected into the patient and their progress around the body is followed using a detector | Gamma | Highly penetrative - pass out of body. Weakly ionising - minimalizes damage to body tissue. Short half-life sample - not in body for long |
control, or destruction of unwanted tissue | High dose of radiation are directed at cancerous cells to kill them | Gamma - radiotherapy Beta - implant method | Highly penetrating - pass out of body. High does - compensate for weakly ionising. Long half-life - won’t decay too quickly |
Background radiation is around all the time
Radiation does is measured is sieverts (Sv) 1000mSv = 1Sv
Level of background radiation does may be affected by occupation and/or location
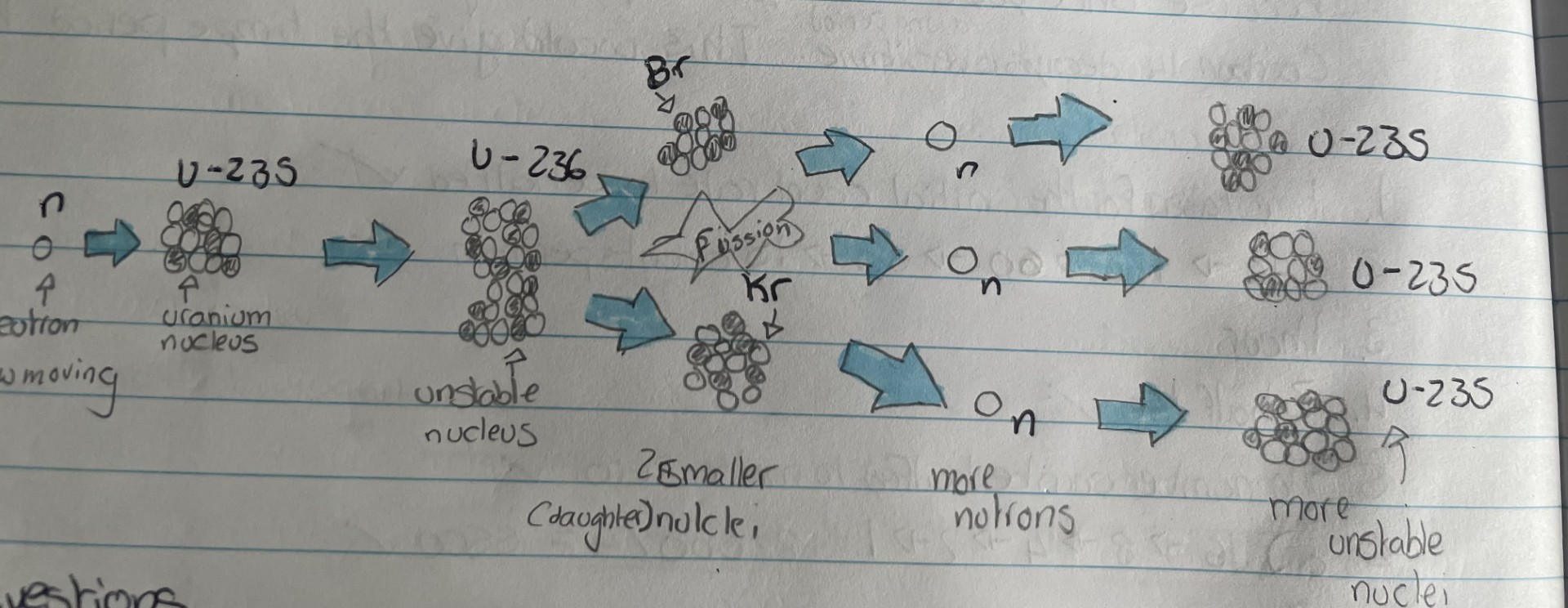
Nuclear Fission
Nuclear fission is the splitting of a large, unstable atomic nucleus
Uranium-235 and Plutonium-239 are fissionable materials commonly used in fission reactors
The nucleus must absorb a neutron for fission to occur
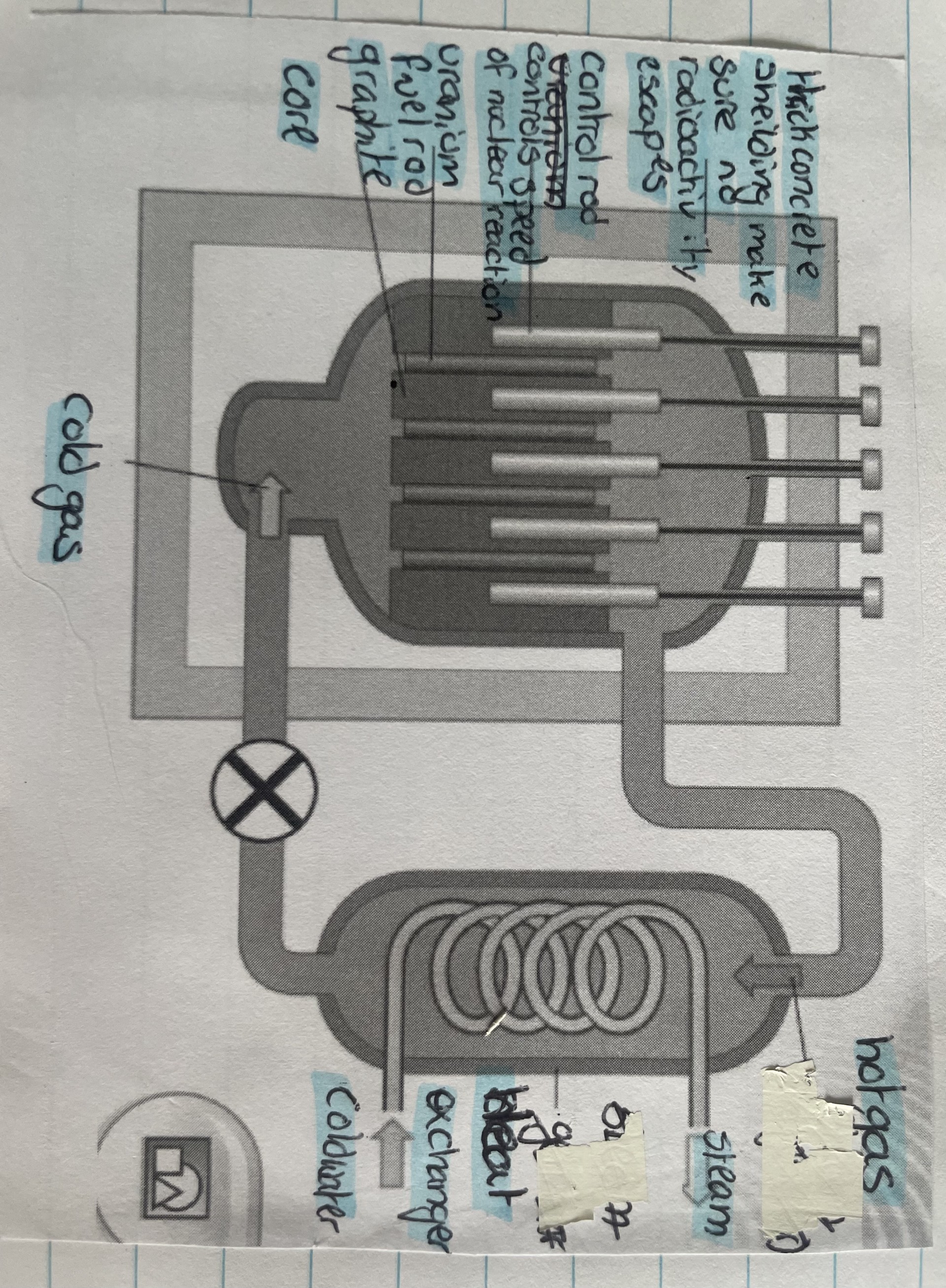
Cor at the centre contains rods of enriched Uranium fuel surrounded by a moderator
Moderator - slows down fast neutrons so the fission chain reaction doesn’t break down
Control rods control the speed of the chain reactions
As control rods are pushed into the core, they slow down the fission reaction, as they are puller out, the chain reaction increases
Control rods absorb neutrons
Pros | Cons |
doesn’t produce CO2 lasts many years more energy produced per 1kg of fuel | expensive to build radioactive waste to be stored non - renewable slow start up time dangerous |
Nuclear Fusion
Nuclear fusion is the joining of 2 light nuclei to form a heavier nucleus
In this process, some of the mass may be converted into the energy of radiation
