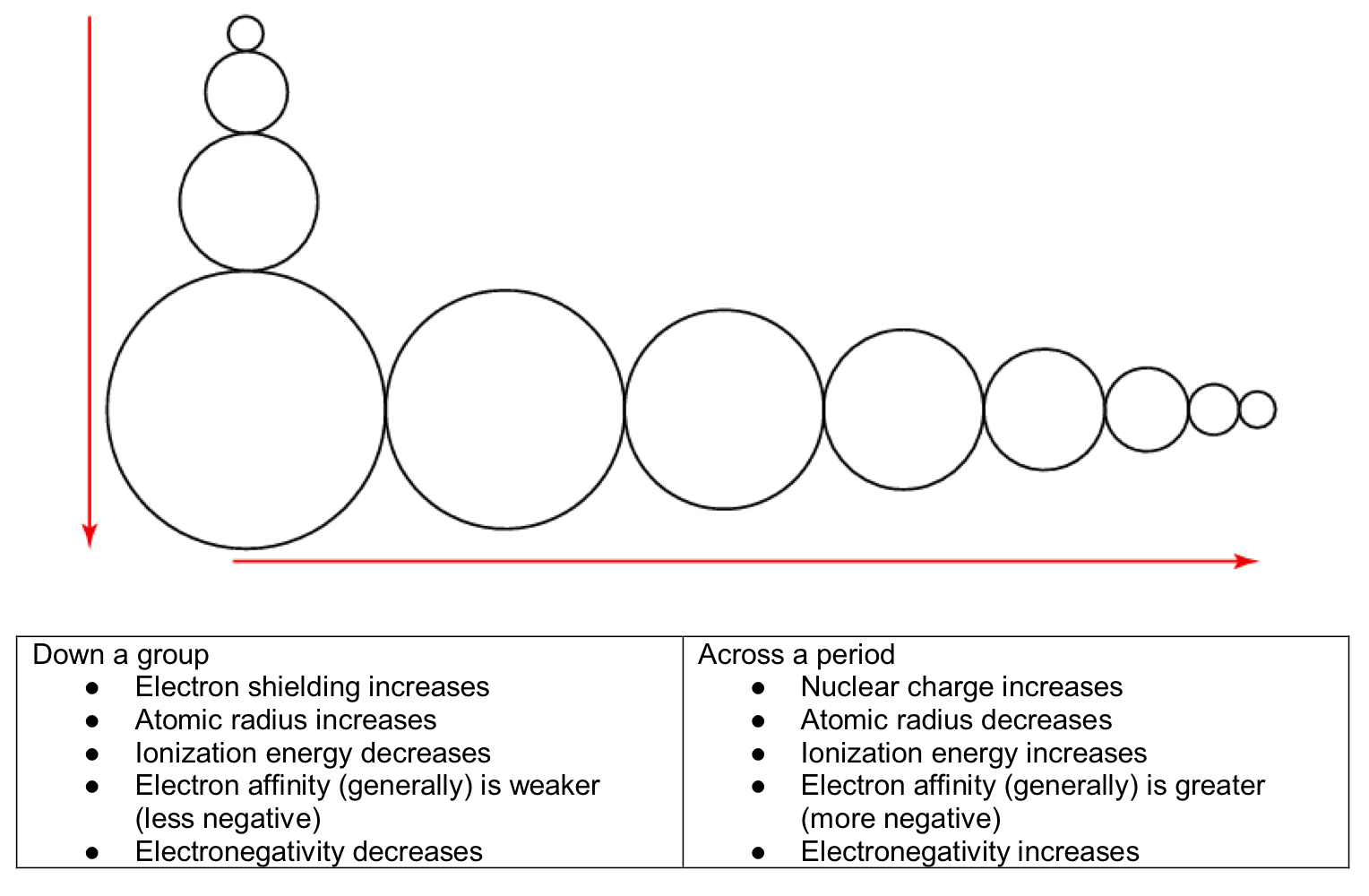
Periodicity
Periodicity - the trends in properties of elements across a period and down a group.
Atmoic radius
Ionic radius
Ionization energy
Electronegativity
Electron afinitniy
Effective Nuclear Charge - the attraction of the positively charged nucleus acting on the valence electrons, taking into account electron shielding from core electrons
Atomic Radius
Used to describe the size of an atom.
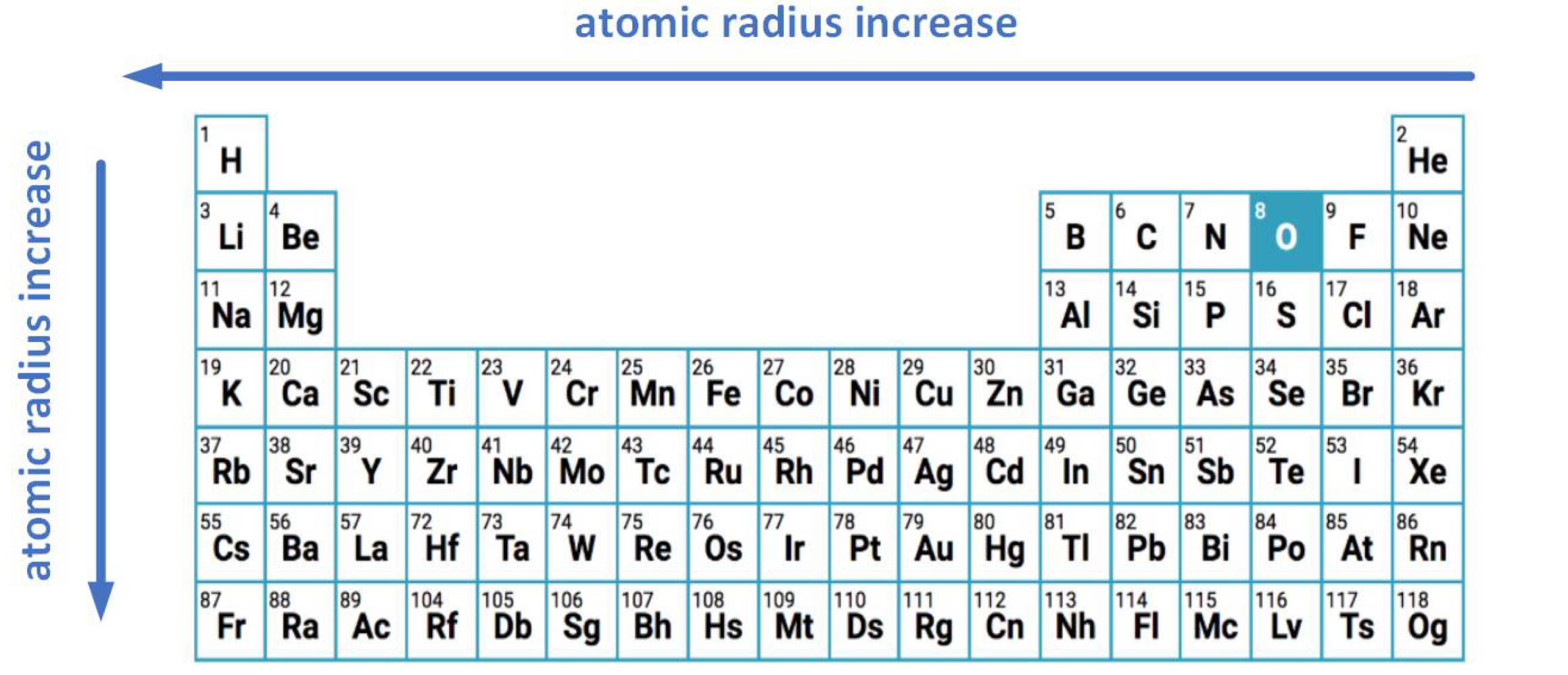
Across the period: decreases
increase of # of protons and # of electrons (in the same valence shell)
nuclear charge increases
no change in electron shielding
Down the group: increases
increasing number of main electron shells where valence electrons are added to higher energy levels (further from nucleus)
increasing number of inner shells
increases electron shielding effect (reduces attraction to valence electron from nucleus
Ionic Radius

Radius of cation < Radius of parent atom
same # of protons, but less electrons —> smaller radius
less electron-electron repulsion
Radius of anion > Radius of parent atom
greater electron-electron repulsion
Isoelectronic ions: Ion with more protons will have a smaller radius
Ionization Energy
The energy required to remove one electron from each atom in one mole of gaseous atom
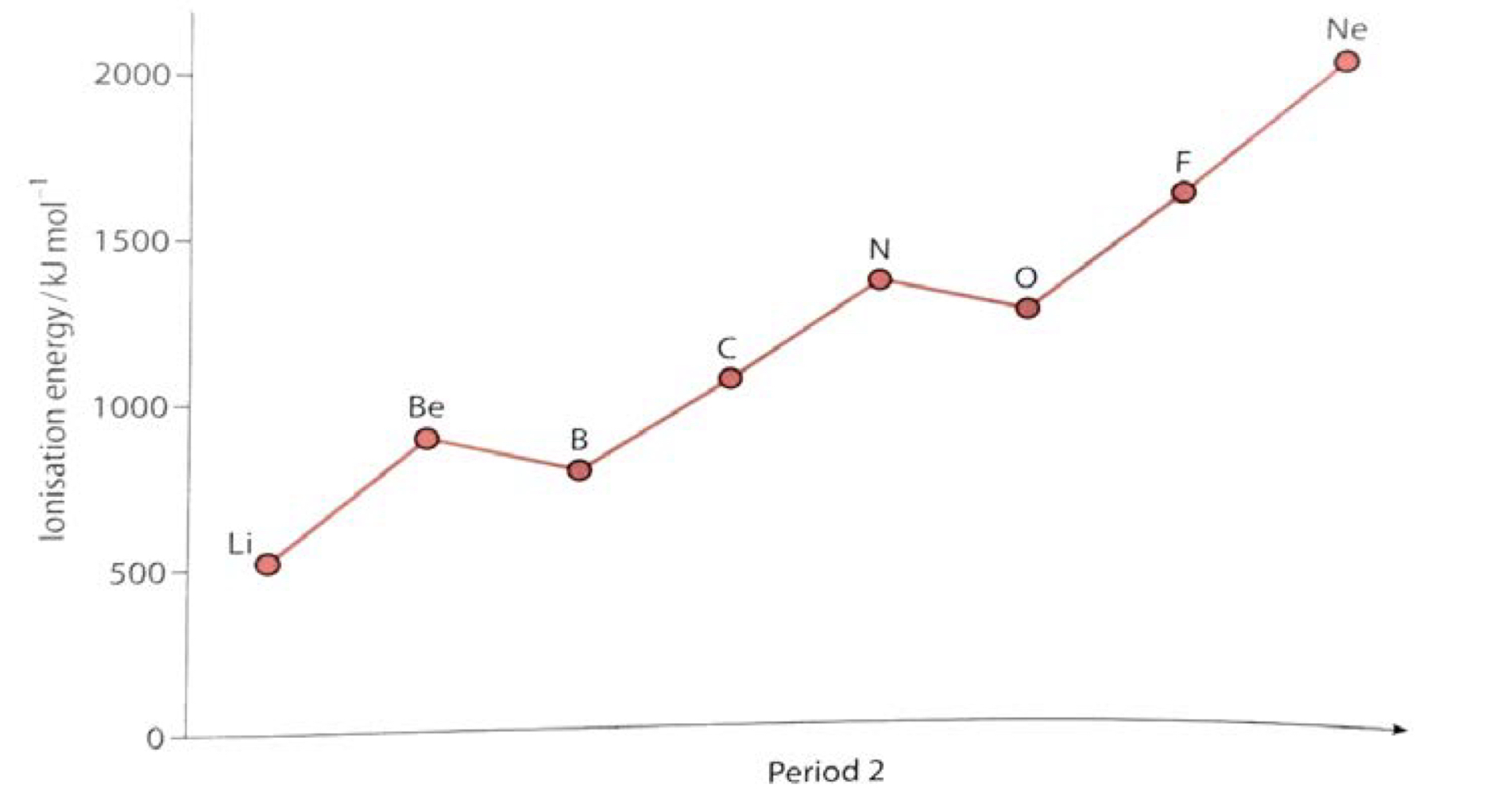
Across the period: increases
atomic radius decreases, nuclear charge increases
valence electrons are closer to the nucleus
Down the group: generally decreases
atomic radius increases down a group, so the valence electron is further away (more shielding)
Electronegativity
The relative attraction that an atom has for the shared electron in a covalent bond
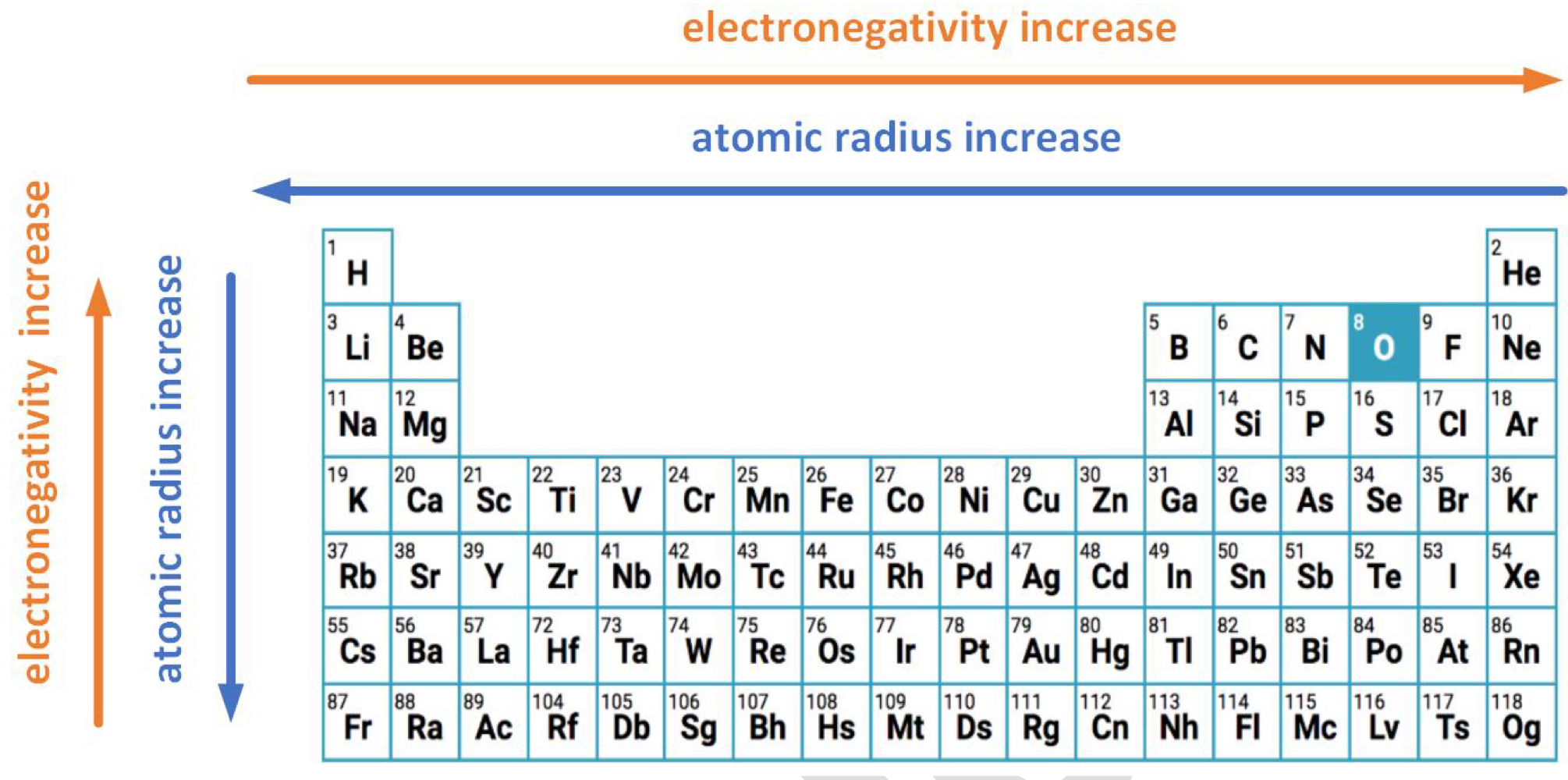
Across the period: increases
# of protons increase → nuclear charge increases
atomic radius decreases, so nuclear attraction to electrons in the bond becomes stronger
Down the group: decreases
atomic radius increases, so valence electrons are in a higher energy level
nuclear attraction to electrons in the bond becomes weaker
Relationship between electronegativity difference and bond character
Higher difference in electronegativity = higher ionic character
Lower difference in electronegativity = higher covalent character
Electron Affinity
The enthalpy change when an electron is added to an isolated atom in a gaseous state
The energy released when one electron is added to each atom in one mole of gaseous atoms
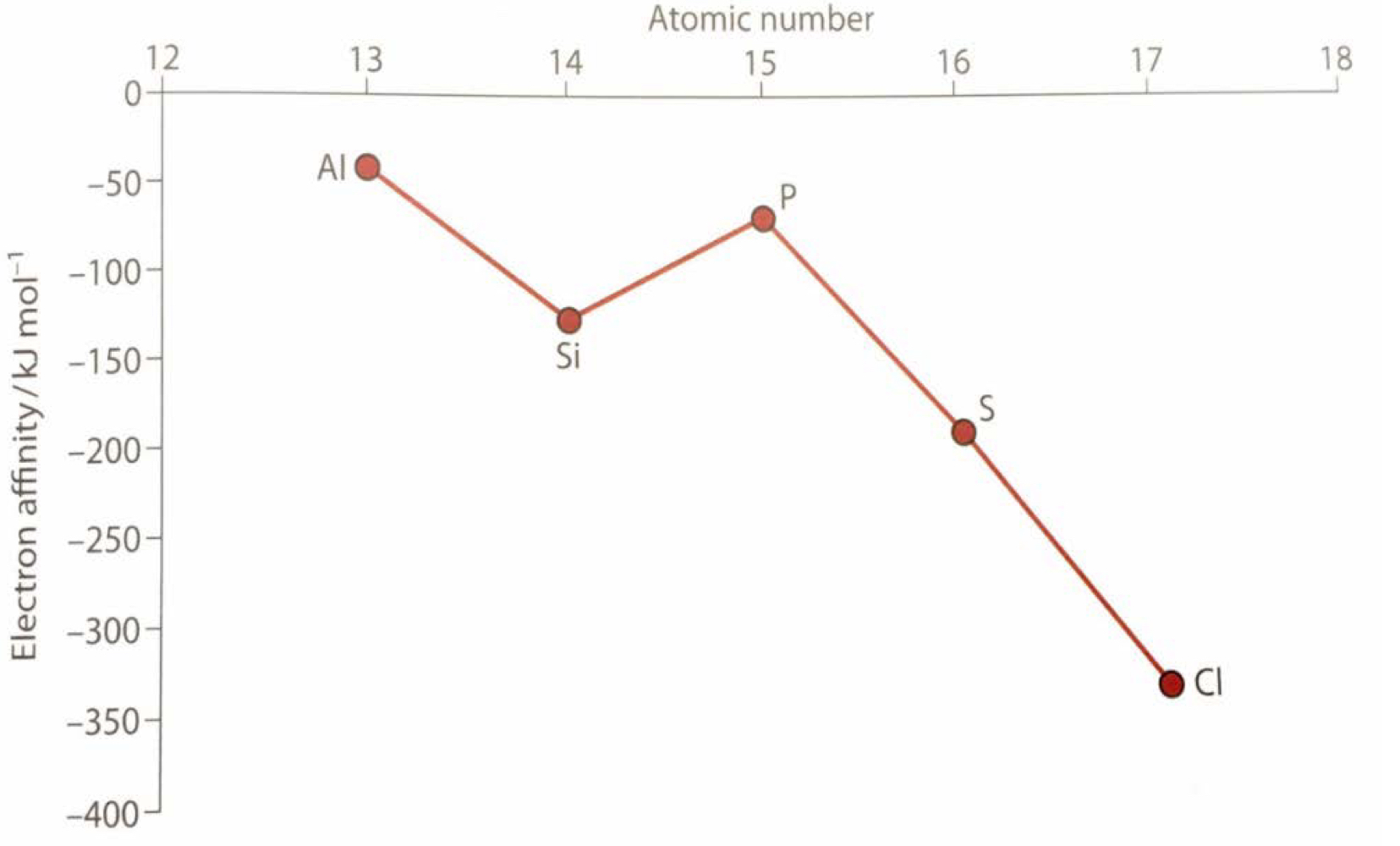
Stronger affinity for a free electron → more energy will be released
Always a negative value
energy is released (exothermic reaction)
the more negative, the more exothermic
Across the period: stronger (more energy)
# of protons increases
stronger attraction between valence electrons and the nucleus
Halogens (Group 17) are the strongest (most negative)
Gaining one more electron → noble gas (stable)
Exception: when an electron is added to half-filled orbitals, it will be less exothermic, since the orbital already contains an electron
Down the group: weaker
number of energy level increases → atomic radius increases
weaker bond between added electrons and the nucleus
Exception: in very small atoms, the electron affinity may be less exothermic, because of the electron-electron repulsions
F has less electron affinity than Cl, since F is so small that when an electron is added, there are electron-electron repulsions
Summary of Trends