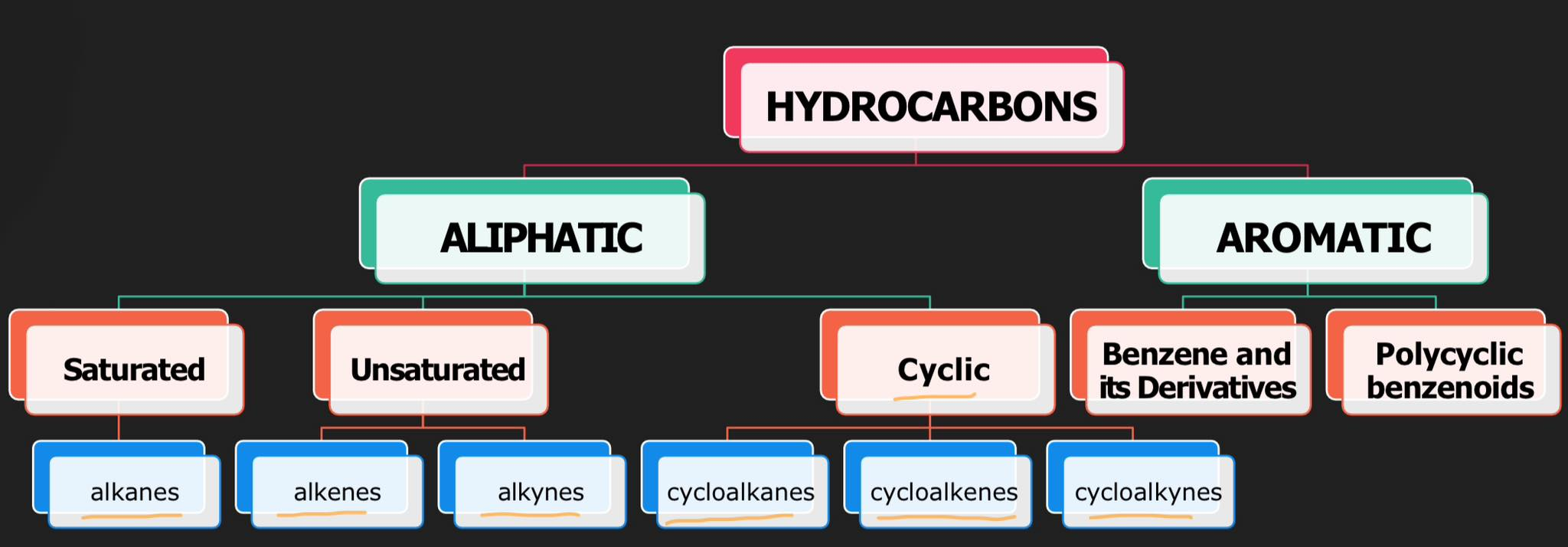
Hydrocarbons
organic compounds composed of carbon and hydrogen atoms only
most hydrocarbons are used as fuel (petroleum products) because of its flammable property
special property to catenate forming seemingly endless chain of carbons (potential combinations estimated up to 106)
non-polar in nature, thereby most of them are insoluble in water
ex. An oil spill in ocean.
Petroleum does not mix with water due to opposite polarities.
Classified based on the presence of multiple bonds:
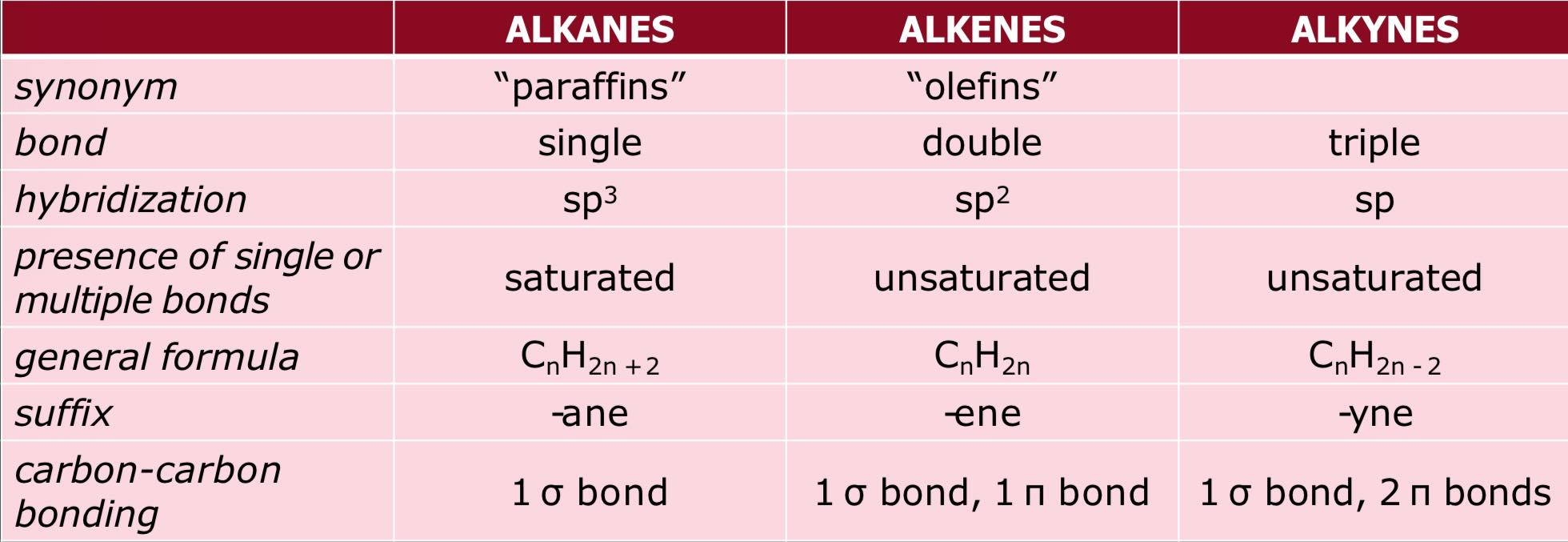
Saturated - carbon atoms are bonded with SINGLE bond (1σ)
possesses one sigma bond which is relatively strong type of bond
Unsaturated - carbon atoms are bonded with DOUBLE (1σ and 1π) or TRIPLE (1σ and 2π) bonds
possesses a sigma and pi bonds; pi bonds are relatively weak type of bond
this makes unsaturated hydrocarbons more reactive than saturated ones
Gasoline is composed of mixtures of paraffins, olefins, naphthenes and benzene.
Classification based on their structures:
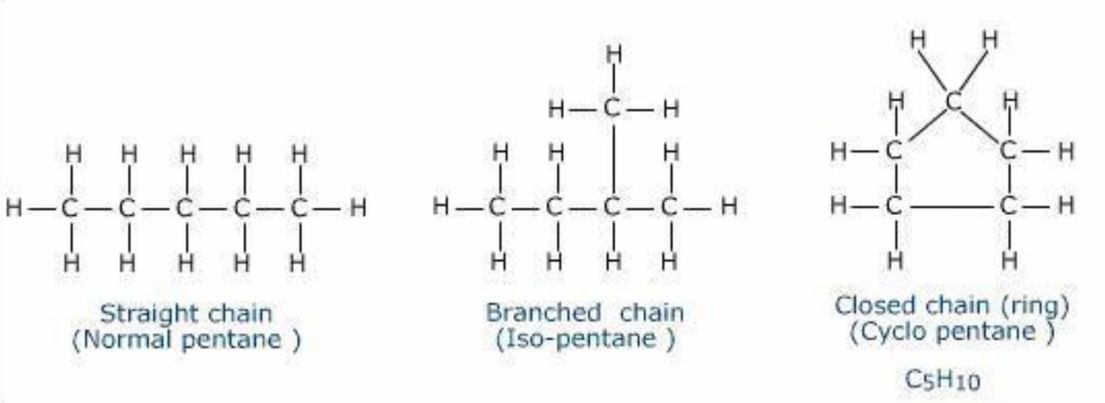
Open-chain
linear structure
could be straight or branched chains
Closed-chain
ring structure
could be aliphatic or aromatic
ALIPHATIC HCs (open-chain)
AROMATIC HCs
are cyclic hydrocarbons with delocalized pi electrons between carbon atoms of ring
their natural characteristics are described as aromaticity
contain conjugated pi bonds, that is alternating double bonds
this special arrangement of pi bonds exhibits resonance (same structure kahit bali-baliktarin), which causes the conjugative stability of the aromatic compounds
According to Hückel's Molecular Orbital Theory, a compound is particularly stable if all of its bonding molecular orbitals are filled with paired electrons
the number of pi electrons = 4n + 2= n, where n = 0 or any positive whole integer
final answer should be whole number
example:
4n + 2 = 6
4n = 6 - 2
4n = 4
/ 4 = / 4
n = 1
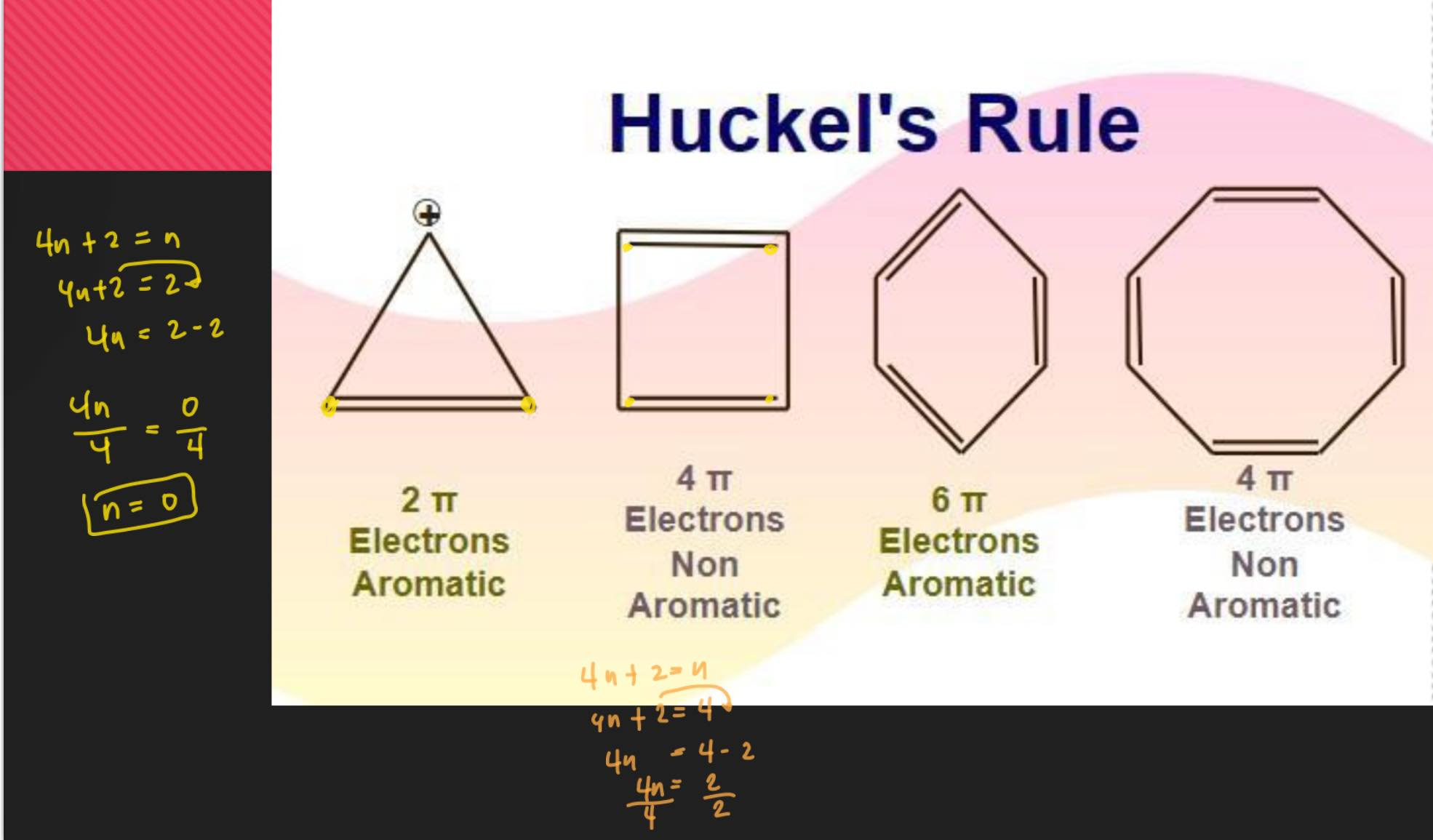
criteria for aromaticity:
must be cyclic (clove) in structure
must be flat or planar in configuration
must have conjugated double bonds
must follow Hückel’s rule of aromaticity
*tumatalon-talon yung electrons kaya may space in bet.
Benzene (most common aromatic compound)
generally undergoes electrophilic aromatic substitution reactions
*hindi nasisira yung structure
it undergoes substitution reaction wherein the ring system is not destroyed and therefore resist addition of substituent groups within the pi bonds
this explains the reason why aromatic compounds resembles reactions of saturated rather than unsaturated hydrocarbons
Experiment results
Ignition Test

combustion reaction
hydrocarbon reacts with oxygen to produce carbon dioxide, water, and heat

incomplete combustion reaction:
hydrocarbons react with insufficient amount of oxygen, instead of producing CO2, produces CO, H2O, and C as products
When a fuel (e.g. gasoline, kerosene) burns in plenty of air, it receives enough oxygen for complete combustion, thereby producing non-luminous flame
When a fuel burns in a limited space (e.g. car engine), there is no enough oxygen to completely oxidize the fuel, thereby producing black smoke/soot
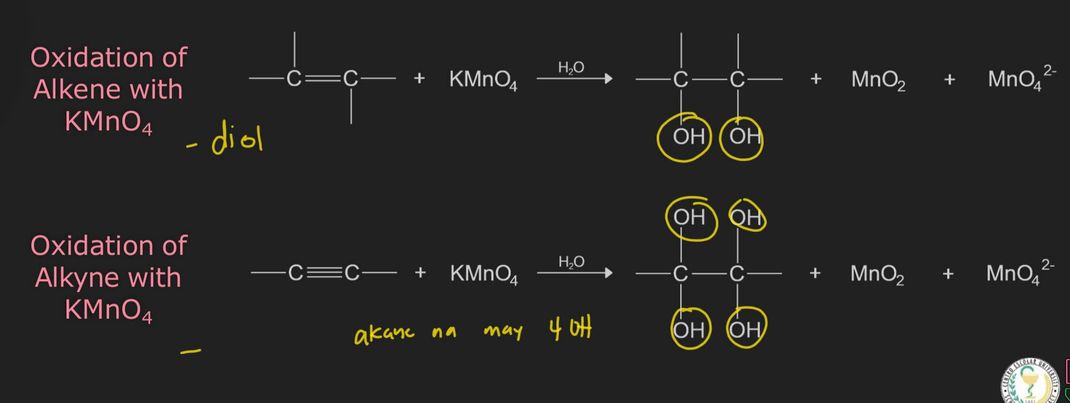
Baeyer’s Test for Unsaturation

named after Adolf von Baeyer
used as a qualitative test for unsaturation
Baeyer's reagent is an alkaline solution of potassium permanganate (KMnO4) (strong oxidizer)
reaction with double or triple bonds (-C=C- or -C≡C-) in an organic material causes the color to fade from purple to brown precipitate
oxidation reaction
an alkene/alkyne is oxidized by KMnO4 producing products of a diol (for alkene), an alkane with four hydroxyl groups (for alkyne), a manganese dioxide (MnO2) and permanganate ion (MnO4-2)

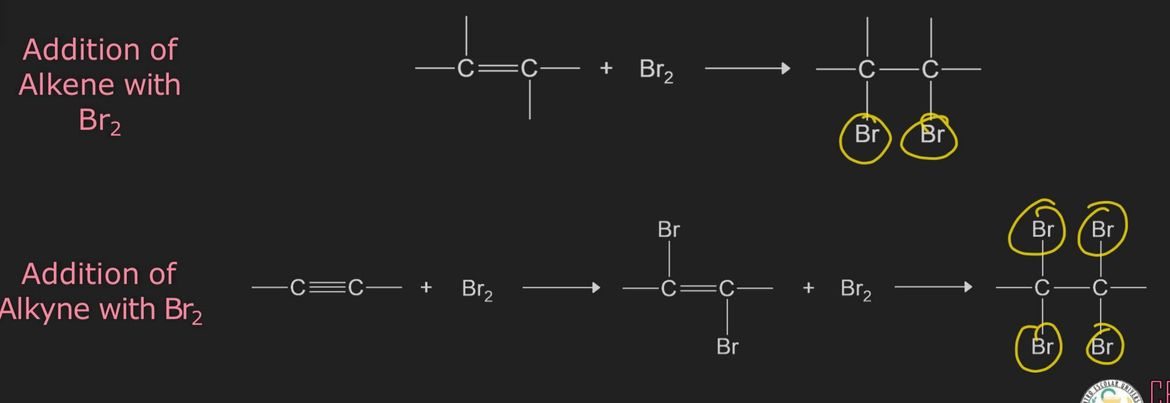
Bromine Test for Unsaturation
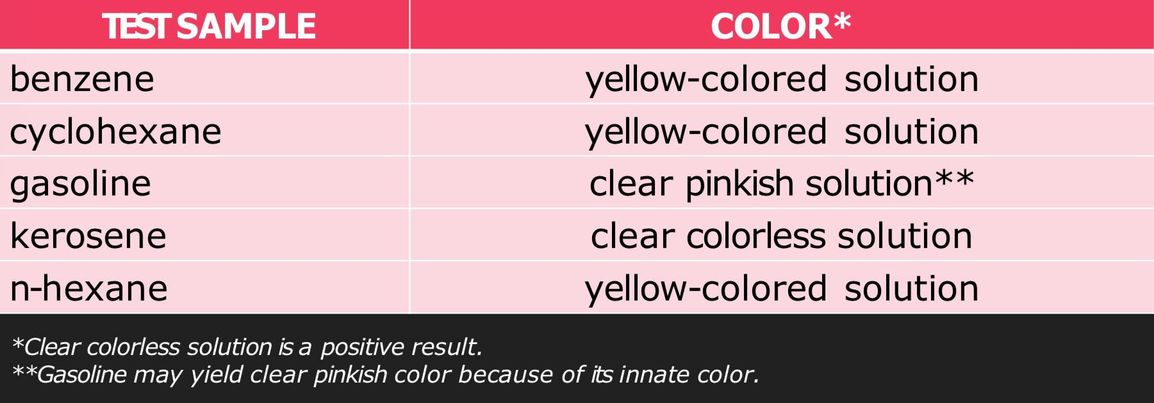
used as a qualitative test for unsaturation
bromine is dissolved either in dichloromethane (CH2Cl2), chloroform (CHCl3), or carbon tetrachloride (CCl4) and the alkene/alkyne sample is added to it
reaction with double or triple bonds (-C=C- or -C≡C-) in an organic material causes the color to fade from brown to clear colorless solution
addition reaction
an alkene/alkyne reacts with Br2 producing products of a dibromo for alkene and ultimately a tetrabromo for alkyne
the weak pi bond in alkenes and alkynes are susceptible to addition reactions
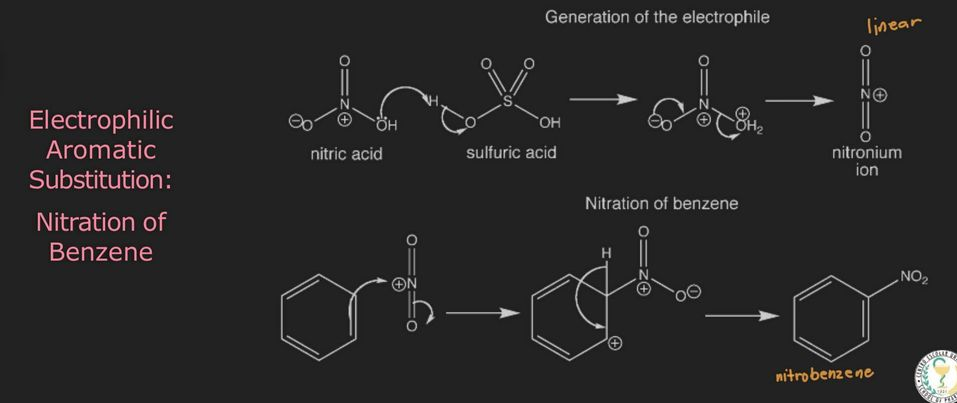
Test for Aromaticity (nitration)

benzene is nitrated using a nitrating acid (HNO3 + H2SO4) to produce nitrobenzene
this nitration test predicts the presence of aromatic ring through the formation of yellow colored layer in solution after warming the test sample
only aromatic compounds will proceed through
electrophilic aromatic substitution
electrophilic aromatic substitution reaction:
nitration of benzene firstly involves the formation of a very powerful electrophile, the nitronium ion; which is linear
this occurs following the interaction of sulfuric and nitric acid
sulfuric acid is stronger and it protonates (transfer proton) the nitric acid on the OH group so that a molecule of water can leave
benzene attacks the positively charged nitrogen atom of the electrophile,
where one of the N=O bonds is broken at the same time
this is followed by rapid loss of a proton to regenerate the aromaticity → nitro benzene
SUMMARY:
Test Sample | Ignition Test flame/presence of soot | Baeyer’s Test | Bromine Test | Bromine Test |
|---|---|---|---|---|
benzene |
| purple soln. | yellow soln. | pale yellow layer in soln. |
cyclohexane |
| purple soln. | yellow soln. | clear colorless soln. |
gasoline |
| dark-brown ppt | clear pinkish soln. | deep yellow layer in soln. |
kerosene |
| reddish-brown ppt | clear colorless soln. | yellow layer |
n-hexane |
| purple soln. | yellow soln. | clear colorless soln. |