P5: nuclear physics
The nucleus
Composed of: protons, neutrons -> overall positive charge
protons = electrons (atomic number, Z)
Nucleon number, A (mass number) = protons + neutrons
Nuclide notation: a group of atoms containing the same number of protons and neutrons
Isotopes:
some are radioactive
occur naturally
Protons, neutrons and electrons
relative charge: the ratio of the charge of a particle compared to the fundamental charge (no units)
proton: +1
neutron: 0
electron: -1
Fission and fusion
Nuclear fission: splitting of a large, unstable nucleus into 2 smaller nuclei
Nuclear fusion: 2 light nuclei join to form heavier nucleus
requires extremely high temperature and pressure
Ionising nuclear & background radiation
ionisation: an atom becomes negatively/positively charged by gaining/losing electrons
background radiation: radiation that exists around us all the time
natural sources: radioactive elements that have always existed
man-made: from human activity
sources: radon gas, rocks and buildings, food and drink, cosmic rays, medical sources, nuclear waste, nuclear accidents
Detecting radiation: using a counter
detecter uses count rate measured in counts/s or counts/min (number of decays per second)
decreases the further away the detector is from the source (radiation becomes more spread)
Geiger-muller tube detects count rate
absorbs radiation -> transmits an electrical pulse to a counting machine
higher count rate = more radiation is being absorbed
Types of radioactive decay
Radioactive decay: change in unstable nucleus that can result in the emission of:
alpha (a) particles
beta (B-) particles
gamma (Y) radiation
spontaneous and random
Alpha:
same as a helium nucleus
charge of +2
low penetrating (stopped by paper)
highly ionising
Beta particles:
fast moving electrons
-1 charge
moderate (stopped by aluminium)
moderately ionising
Gamma:
electromagnetic
have highest energy
no charge
high penetrating (stops at lead)
low ionising
Ionising effect of radiation
depends on: kinetic energy and electric charge
Nuclear radiation can ionise the atoms it hits by removing an electron so the atom loses a negative charge (positive charge overall)
Effects of kinetic energy and charge on ionising power:
greater the charge → more ionising it is
higher the kinetic energy → the more ionising it is
Deflection in electric fields:
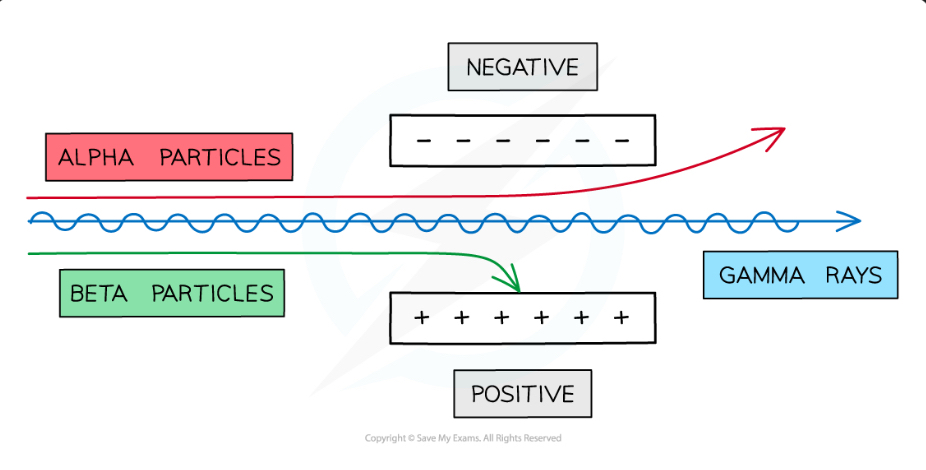
Deflection in magnetic fields:
alpha and beta particles are deflected in opposite directions due to opposite charges
Radioactive decay
isotopes of an element may be radioactive due to an excess of neutrons → nucleus is too heavy
too many protons creates a repulsive force → neutrons repel each other
decays into several smaller nuclei due to imbalance of protons/neutrons until stable nuclei
nucleus too heavy → forces keeping protons and neutrons together will be weaker
mass number of the element reduces during nuclear decay
During alpha and beta decay, nucleus changes to new element
alpha decay: alpha particle is emitted
beta decay: neutron → proton + electron
Decay equations:
Alpha:
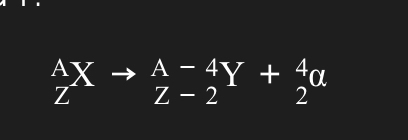

Half-life: time taken for half the nuclei of that isotope in any sample to decay
Uses of radiation: the type of raditation emitted and the half-life of an isotope determine which isotope is used
alpha radiation: fire alarms
alpha radiation ionises the air in the detector → creates a current
alpha emitter is blocked when smoke enters the detector
alarm is triggered when sensor no longer detects alpha particles
gamma rays: sterilization of equipment and irradiating food to kill bacteria
most penetrating → can penetrate enough to irradiate all sides
can sterilize without removing packaging
kills microorganisms → food lasts longer and reduces food-borne infections
beta radiation: measuring thickness of materials
can be partially absorbed by most materials
material moves across a radiation source: if material gets thicker → more particles are absorbed → less gets through to be detected
machine makes adjustments to keep thickness of the material constant
gamma rays: diagnosis and treatment of cancer
radiotherapy: kills living cells
beams of gamma rays are directed at the cancerous tumor → can penetrate the body and can be moved around to minimise harm to healthy tissue
tracer: radioactive isotope that can be used to track the movement of substances around the body
Dangers of radiation: can damage living things such as human cells and tissues at high doses
cell death
mutations
cancer
Safe storage
lead-lined boxes and keep them at a distance
minimize the amount of time you handle sources
keep far away from the sources
use a pair of tongs at arms length
transported in special containers like casks
radioactive waste is buried underground