Unit 08: Bond Types
Valence Electrons Reminders
The e- is the outermost energy level of an atom.
The e- involved in bonding.
The number of valence electrons- determines how many bonds the atom will form.
They determine what type and charge ion is formed by an element.
Octet Rule Reminders
In forming compounds, atoms tend to achieve the e- configuration of a Nobel gas.
An octet is a set of 8.
Electron configurations will end with
They will lose, gain, or share e- in order to obtain this state.
01: Covalent Bonds
Formed between nonmetal atoms
Make up molecules
Share electrons to form bonds
Both atoms will have a full valence shell
All covalent bonds are weaker than ionic bonds
Compounds with these bonds can be liquids, solids, or gases at room temperature
Most have low melting points
Most have relatively low boiling points
Do not conduct electricity
The diatomic elements (BrINClHOF) Form covalent bonds
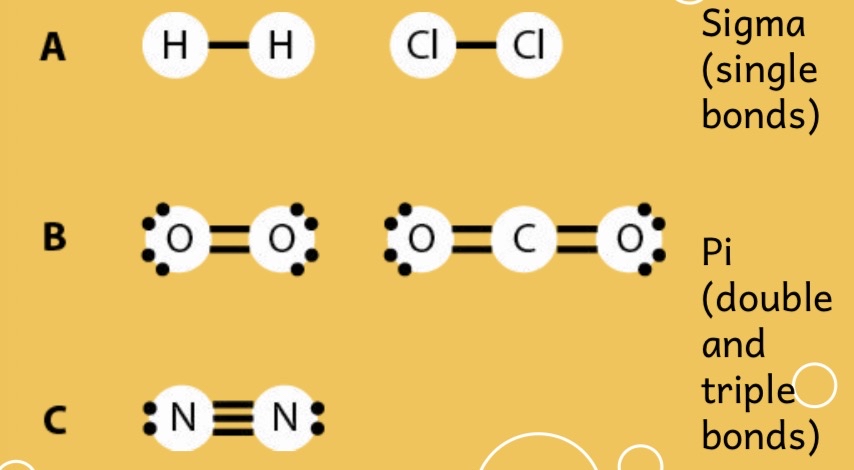
There are two types of covalent bonds:
Sigma bonds - these are single bonds between elements
Pi bonds - these are multiple bonds between elements
Double bonds =
Triple bonds (three lines)
Remember each dash represents two e-
Types of Covalent Bonds!:
02: Metallic Bonds
Metal bonds are arranged in very compact and orderly patterns.
The e- are not shared, gained, or lost to form these bonds.
Outer energy levels in the metal atoms will overlap.
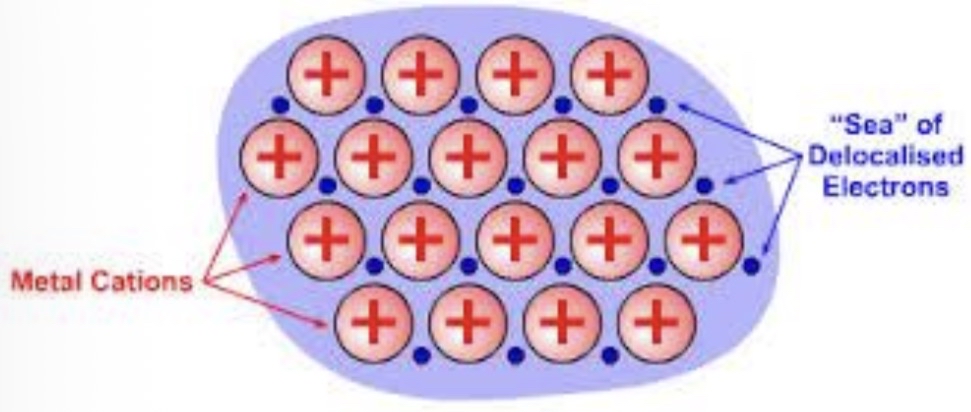
The metal atoms in a metallic solid contribute their valence e- to form a “sea of e-“
The e- are not held to any certain atom, they move freely from one atom to the next.
These e- are known as delocalized e-.
They do not have a specific location in the bond.
Think of a metallic bond as a peanut butter sandwich.
The two pieces of bread represent the atoms.
The peanut butter represents the “sea of e-“
The peanut butter hold the sandwich together and it is not specific to either piece of bread
The e- hold the bond between the atoms together and they are not specific to either atom
When the valence e- leaves to join the sea of e-, it leaves behind a metallic cation (the individual atoms have a positive charge)
The bond is the attraction of a metallic cation for delocalized e-
Compounds containing metallic bonds have the following properties:
Malleable
Ductile
Durable
Fairly high boiling points
Melting point vary, but are generally moderate
Alloys are a mixture of elements with metallic properties
The properties of the alloy are often superior to the properties of their individual parts
Alloys are important because they increase the usage of metals
Some examples of alloys are steel, pewter, and brass
There are two types of alloys:
Interstitial
Small holes in the metal lattice are filled with smaller atoms
Ex: carbon steel - Iron crystals are filled with carbon atoms producing a stronger, harder, and more ductile substance
Substitutional
Atoms of the original metal are replaced by atoms of similar size of another metal
Ex: sterling silver, brass, and pewter
Alloys

03: Ionic Bonds
Formed between oppositely charged ions
Metals form cations
Nonmetals form anions
E- are transferred from one atom to another
The formation of ionic compounds are always exothermic
Ionic bond form crystal lattice structures
Ionic bonds are different from metallic bonds, but share some of the same properties
They are relatively strong bonds and require large amounts of energy to break apart
Compounds will have high melting and boiling points
Compounds are hard, brittle solids at room temperature
In their solid state, they do not conduct an electric current
When they are dissolved in water, the ions dissociate to form ions
These are known as electrolytes
Electrolytes do conduct electricity very well
04: Bond Types Based on EN Difference
Differences in electronegativity (EN) determine whether a bond will be classified as nonpolar covalent, polar covalent, or ionic
EN is how strongly atoms attract bonding e- to themselves
They higher the EN, the stronger the “pull” that atom has for e-
You can look at it like a tug of war for the e-
A nonpolar covalent compound is one in which the e- are shared equally or fairly equal in the bond
The tug of war is essentially equal
The difference in the atoms’ electronegativities will be no greater than 0.4
An example of a nonpolar covalent compound would be any of the diatomic molecules since the EN difference is 0
Another example would be SeS. Se has an EN of 2.4 and S has an EN of 2.5, giving a difference in EN of 0.1
In a polar covalent compound, there will be one atom that has a stronger attraction for the e-
Because of this difference in attraction, there will be a slightly positive end and a slightly negative end to the compound
In our tug tug of war, one element is pulling more than the other
The difference in EN will be 0.4 to 1.5 in a polar covalent compound
In a ionic compound, there will be one atom that has a much larger attraction for the e-
The tug of war will be extremely one-sided. It would be like the Atlanta Falcons defensive line versus a group of kindergarten students
The difference in EN will be 1.6 or greater
