Enthalpies of Formation
The enthalpy of formation of an element in it’s standard state is equal to zero.
The enthalpy change when one mole of a substance forms from its elements in their standard states.
Examples: For example, when you combine two hydrogen gas molecules (H2) and one oxygen gas molecule (O2) to create one water molecule (H2O) at standard conditions (like room temperature and pressure), it releases around 285.8 kilojoules of heat energy. This heat energy release is a characteristic standard enthalpy change for this chemical reaction.
*STANDARD STATES OF ELEMENTS AND DIATOMIC MOLECULES (AT 25 degrees and 100 kPa)
mercury and bromine are liquids
noble gases are diatomic (HOFBrINCl) are gases
everything else is a solid
Formula: 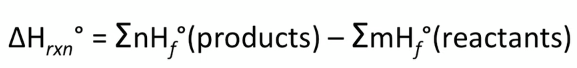 Practice:
Practice:
Ex. 1: Calculate the enthalpy change for the combustion of one mol of glucose (C6H12O6) to carbon dioxide and water.
Step 1: Write the balanced chemical equation
C6H12O6 + O2 = CO2 + H2O
Step 2: Look up the standard enthalpies values for the products and reactants on table and place in equation.
