energetics
when a reaction is exothermic, it gives out energy and so gets HOTTER
when a reaction is endothermic, it takes in energy and so gets COLDER
polystyrene cups are used in displacement reactions because they act as an insulator and reduce the amount of heat lost to the surroundings
(general) equations…
amount of energy produced per gram = amount of energy produced / mass of (ethanol) burnt
Q = mc*∆T*
∆H = -(E / 1000) / moles (of the limiting reactant)
concentration = moles/volume (in dm^3)
moles = mass / Mr
reactions
displacement reactions are exothermic
combustion reactions are exothermic
bonds breaking is exothermic
bonds forming is endothermic
DISPLACEMENT REACTION

COMBUSTION REACTION

CALCULATIONS
Calculating heat energy change is done by
Q = mc∆T
“M” is the mass of the LIQUID
“C” is the specific heat capacity and is always 4.2 unless you are told to use 4.18
“∆T” is the change in temperature, whether the reaction was exothermic or not it will always be a positive number
Then, to calculate the enthalpy change…
∆H = -(E / 1000) / moles (of the limiting reactant)
Amount of energy transferred (in kJ)/ number of moles (mol)
The moles used must be the limiting amount, and if the reaction was EXOTHERMIC ∆H will be NEGATIVE and if the reaction was ENDOTHERMIC ∆H will be POSITIVE
The units are kJ / mol
ENERGY LEVEL DIAGRAMS
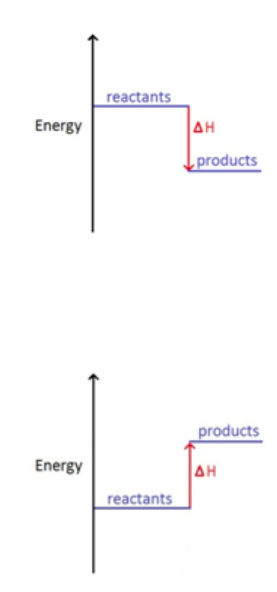
In these diagrams , when a reaction is exothermic the enthalpy change is negative so the products line is below the reactants as energy has been given out
When a reaction is endothermic the enthalpy change is positive and so the products line is above the reactants as energy has been taken in
BONDS !
Breaking bonds needs energy and so is an endothermic reaction, making bonds is exothermic
To find the overall enthalpy change you need to compare the amount of energy taken to make and break the bonds
The overall enthalpy change is the energy needed to break the bonds - the energy released from making new bonds