Module 7 - Ion channels part 2 (P-loop channels, intro to ion selectivity) - 1
P-loop Channels Overview
P-loop channels are integral membrane proteins with significant evolutionary origins in bacteria.
Key Types of P-loop Channels
Nav Channels (Voltage-gated Sodium Channels)
Conduct inward Na+ currents.
Depolarize the membrane potential (Vm) during the rising phase of action potentials.
Kv Channels (Voltage-gated Potassium Channels)
Activated after Nav channels.
Conduct outward K+ currents, repolarizing the membrane potential.
Evolutionary Significance
Both types derive from ancient bacterial channels.
P-loop channels are critical in various physiological processes across organisms.
Emergence of P-loop Channels
P-loop channels originated billions of years ago from an initial prototype: Inward Rectifying Potassium (KIR) Channels.
Structure includes:
2 Transmembrane Helices (TMHs) separated by an extracellular P-loop.
Tetrameric formation (4 subunits required to form a complete channel).
Ion Selectivity
Key amino acids within the P-loops define ion selectivity (specifically for K+ ions).
This forms the ion selectivity filter essential for channel function.
Evolution from KIR Channels
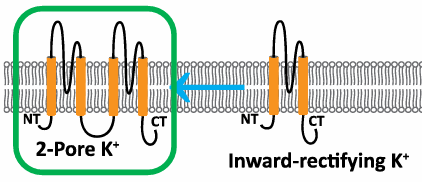
Tandem Duplication of KIR Genes
Gave rise to 2-pore K+ leak channels (K2P channels).
Require only 2 proteins for channel function.
Critical for maintaining resting membrane potential (RMP).
Found exclusively in eukaryotes.
Functional Characteristics
K2P channels, while termed 'leak' channels, exhibit:
Gating and enhanced current flow due to stimuli such as pH, membrane stretch, and temperature.
Ionotropic Glutamate Receptors
Formed from the inversion across the membrane and the addition of a transmembrane helix.
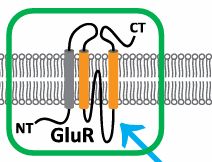
Include AMPA, NMDA, and Kainate receptors, crucial for excitatory synaptic transmission (glutamatergic synapses).
Retain tetrameric structure and are ancient proteins originally from bacterial systems.
Voltage-Gated Channels
Emerged through fusion with a separately evolved bacterial protein (voltage sensor S1-S4). The S1-S4 sensors were fused with the KIR channels
Types include Kv channels, Ca2+-sensitive SK channels, and hyperpolarization-activated channels (HCN).
Some channels lost K+ selectivity, conducting Na+ and/or Ca2+ instead, crucial for sensory biology.
Voltage Sensor Domain (VSD)
Originated as a separate ion channel.
Two human genes (TPTE and TPTE2) conduct voltage-sensitive currents regulated by pH.
Mutations in VSD can create leak cation currents harmful to cells (example: Arginine→Histidine mutation in Nav1.4).
BK Channels
Evolved with an addition of a transmembrane helix at the N-terminus of Kv/SK/TRP channels.
SK channels refer to small conductance calcium-activated potassium channels, which are ion channels that open in response to increases in intracellular calcium levels.
TRP channels stand for transient receptor potential channels, a group of ion channels that are involved in various sensory modalities such as temperature and pain sensation.
Their gating is influenced by rises in cytoplasmic Ca2+ levels.
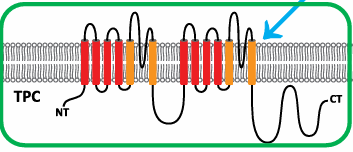
Two Pore Channels (TPC)
Resulted from a tandem duplication of Kv-like channels.
Dimeric structure; cationic (Ca2+, Na+).
Involved in calcium release from endo-lysosomes into the cytoplasm.
4-Domain Channels
Formed through tandem duplication of TPC-like channels:
Include Voltage-gated Na+ channels (Nav, cell depolarization during action potential) and Ca2+ channels (Cav, synaptic transmission, muscle contraction, gene expression).
NALCN is a Na+ leak channel that helps maintain RMP in neurons, crucial for neuronal function.
Yeast Ca2+ channel 1 is found in yeast and homologous with NALCN
Clinical Implications of Mutations
Mutations in the pore region are linked to congenital contractures, hypotonia, and developmental delays (CLIFAHDD).
Voltage sensor mutations result in infantile hypotonia and associated neurological effects (IHPRF1).