Chem Honors First Semester
Polyatomic Ions
Ammonium: NH4+
Acetate: C2H3O2-
Carbonate: CO32-
Cyanide: CN-
Hydroxide: OH-
Nitrate: NO3-
Nitrite: NO2-
Permanganate: MnO4-
Sulfate: SO42-
Sulfite: SO32-
Unit One
Sig Figs
All non-zero numbers are significant: 1, 2, 3
Zeros between two non-zero numbers are significant: 4001
Trailing zeros are only significant if there is a decimal: 100.
Leading zeros are never significant: 0.006
Metric Prefixes + Conversions
King Henry Died By Drinking Chocolate Milk
Scientific Notation: x.y^10(z)
Scientific Method
Observe
Question
Hypothesis
Experiment
Conclusion
Result
Calculation Percent Error
(Correct)-(Output)
——————— = Percent Error
(Correct)
Density: Mass/Volume
Usually g/cm
Classifying Matter
Elements
1 identical atom
Compounds
2 or more elements
Mixtures
2 or more substances
Unit Two
Identify and build atoms
Isotopes
Different mass, same element
Neutrons
Ions
Charge (proton-electron)
Cation/Anion (+/-)
Means gaining electrons to become stable
Neutral
Equal proton and electron
Proton = atomic number
Mass = proton + neutron
Types of materials and characteristics
Metals
Shiny, malleable, ductile, high melting points, high density, good heat conductors
Non-metals
Poor electrical and thermal conductivity, brittle, dull, non-malleable
Metalloids
Typically shiny, brittle at room temperature, semiconductors
Periodic Table
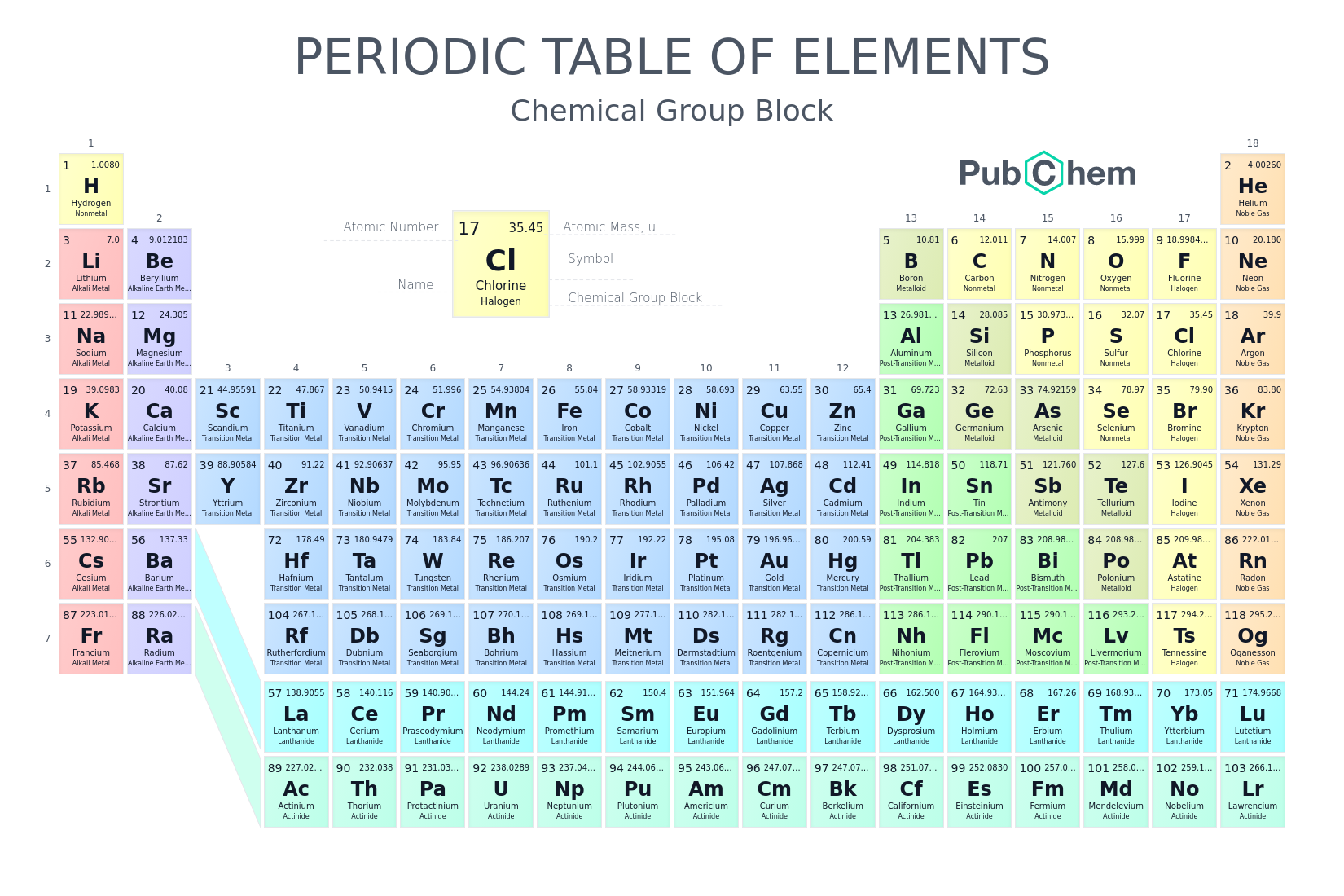
Families
Alkali
Group 1 elements (except hydrogen)
Highly reactive metals
Low ionization energies
Form +1 ions
Soft, silvery metals
React vigorously with water
Alkaline earth metals
Group 2 elements
Reactive metals (less reactive than alkali metals)
Form +2 ions
Higher melting points and densities than alkali metals
Transition
Groups 3-12 elements
Metals with high density and melting points
Often form colored compounds
Can have multiple oxidation states
Halogen
Group 17 elements
Highly reactive nonmetals
Form -1 ions
Known as "salt-formers”
Noble gases
Group 18 elements
Very unreactive (inert)
Have full valence electron shells (8 valence electrons, except He with 2)
Exist as monatomic gases at room temperature
Periods
Orbital notation
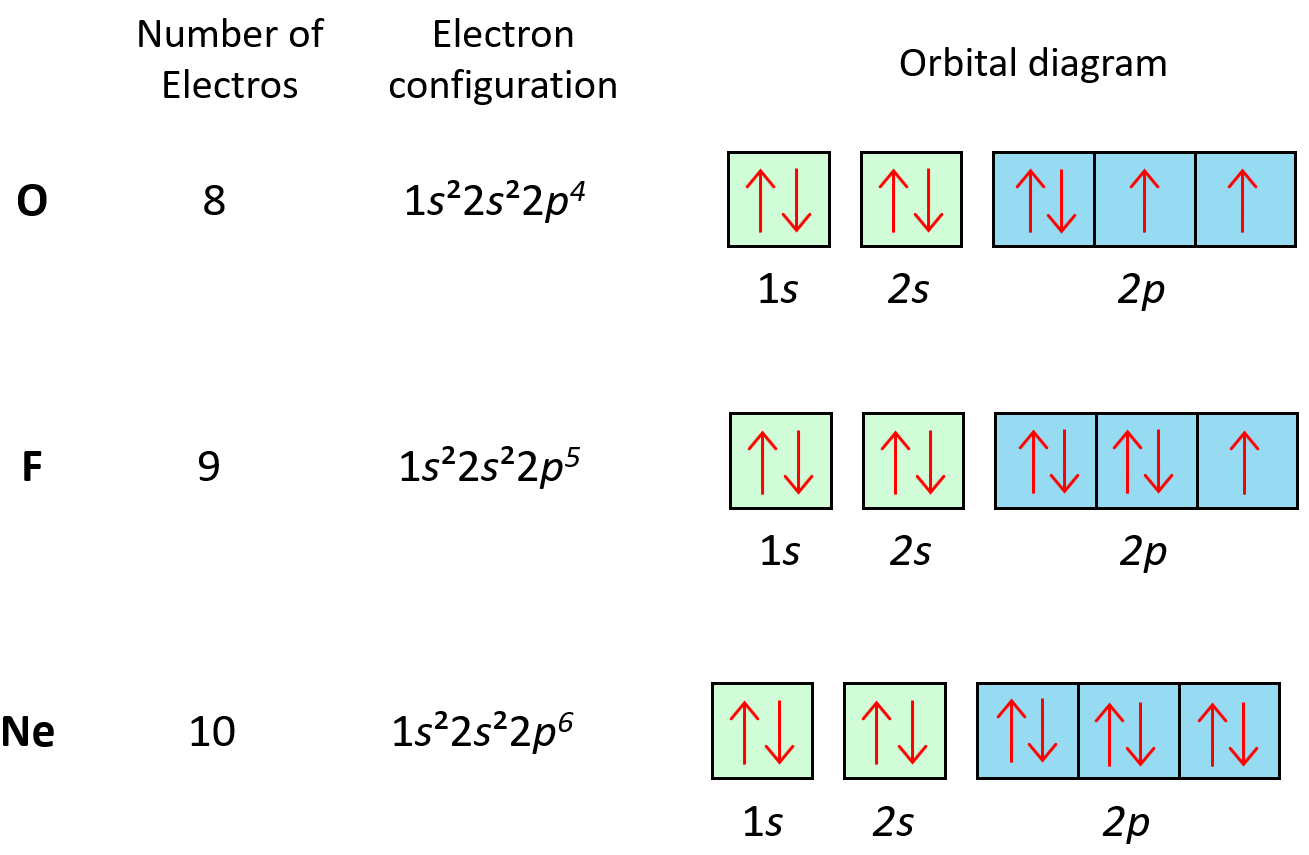
Arrows based on electron configuration
For transition metal ions with a positive charge, take off electrons from the highest energy level
Noble gas configuration
[Noble Gas]…
Electron configuration
#s2#s2#p6#d10
The exponents must add up to the number of electrons in the atom
Average atomic mass
(% x mass) + (% x mass) + (etc)
------------------------------------------
# of masses
Bohr model

Circles
2, 8, 8 - full rings
Electrons
Excited state vs ground state
Ground state: Electrons occupy lowest energy levels possible
Inner shells completed before moving onto next
Excited state: When exposed to energy, an electron may hop to the next energy level
Know how to read nuclear format
AZX
A = Mass
Z = Atomic Number
X = Atomic Symbol
Unit 3
Atomic Structure
Parts of an Atom
Particle
Charge
Location
Proton
+1
Nucleus
Neutron
0
Nucleus
Electron
−1
Electron cloud
Atomic number: number of protons
Mass number: protons + neutrons
Coulombic Attraction
Proton (+) — attracted to — Electron (-) because of charge
The strength of the attraction is affected by…
Distance
As the distance increases, attraction decreases
For example; in a bohr model, the first ring has the strongest coulombic attraction
Does not matter how many electrons, mostly based on distance
Force between electrons and proton on the same ring is all the same
More effect on attraction
Strength of the nuclear charge: Protons
As the nuclear charge increase, attraction also increases
For example: if a nucleus has 3 protons, the electron ring is tighter than a nucleus with 2 protons
PES diagram
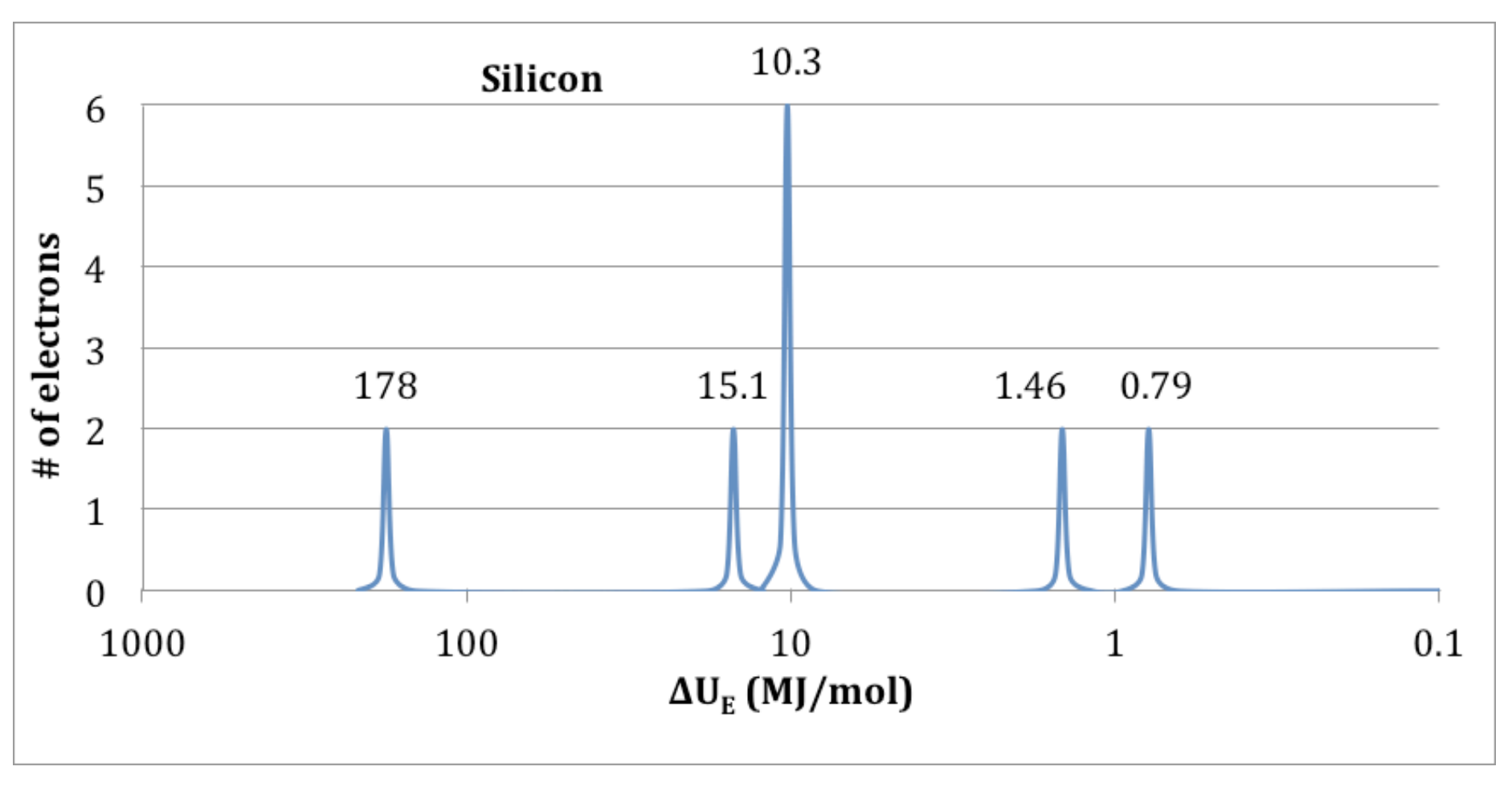
Peaks
Binding Energy (x)
# of Electrons (y)
1s…2s… on peaks
Trends

Atomic size
The size of an atom, measured from the nucleus to the outermost electrons
Increases down a group because atoms gain additional energy levels, placing outer electrons farther from the nucleus
Decreases left to right across a period because the number of protons increases, pulling electrons closer to the nucleus
Ionization energy
The amount of energy required to remove an electron from an atom
Decreases down a group because outer electrons are farther from the nucleus and less tightly held
Increases left to right across a period because increased nuclear charge strengthens the attraction between the nucleus and electrons
Electronegativity
Atom’s ability to attract electrons in a chemical bond
Decreases down a group because larger atomic size weakens the nucleus’s pull on shared electrons
Increases left to right across a period because more protons increase nuclear attraction
Fluorine has the highest electronegativity
Elements in the bottom-left of the periodic table generally have the lowest electronegativity
Atomic vs Ionic size
Atomic size: size of a neutral atom, measured from the nucleus to the outermost electrons
Ionic size: size of an atom after it has gained or lost electrons to become an ion
Cations (+): form when an atom loses electrons
Smaller than the neutral atom: losing electrons reduces electron-electron repulsion and the same number of protons pull fewer electrons closer
Anions (-): form when an atom gains electrons
Larger than the neutral atom: adding electrons increases electron-electron repulsion, spreading the electrons farther apart
Trend across a period:
Atomic size decreases left to right
Cations are smaller than their neutral atoms; anions are larger than their neutral atoms
Trend down a group:
Both atomic and ionic sizes increase because new electron shells are added
Size depends on nuclear charge, number of electrons, and electron-electron repulsion
Identifying elements based on their ionization energy
More energy = closer to noble gas/inner rings
Outer rings are valence electrons
Isoelectronic
Same number of electrons
Families and valence electrons
Unit 4
Naming
General
Determine whether the compound is ionic or covalent first
Ionic = metal + nonmetal or contains polyatomic ions
Covalent = nonmetal + nonmetal
The type determines the naming rules used
Covalent naming
Uses prefixes to show number of atoms
Prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-
First element keeps its full name (no “mono-” if only one)
Second element ends in -ide
Example: CO₂ → carbon dioxide
Ionic naming
Name the cation (positive ion) first, then the anion (negative ion)
Do not use prefixes
Monatomic anions end in -ide
Example: NaCl → sodium chloride
Ionic compounds with transition metals
Transition metals can have multiple possible charges
The charge must be specified using Roman numerals
Example: FeCl₃ → iron(III) chloride
Roman numerals
Show the charge of the metal cation
Roman numeral equals the positive charge
Determined by balancing total charges in the compound
Charge
Total charge of an ionic compound must equal zero
Charges of ions determine subscripts in formulas
Oxidation states help identify metal charge
Polyatomic ions
Groups of covalently bonded atoms with an overall charge
Act as a single unit in reactions and naming
Common examples: NO₃⁻, SO₄²⁻, NH₄⁺
Names stay the same in compounds
Resonance
Occurs when multiple valid Lewis structures exist
Actual structure is an average of resonance forms
Only electrons move; atom positions stay the same
Often seen in molecules with delocalized electrons
Lewis structures
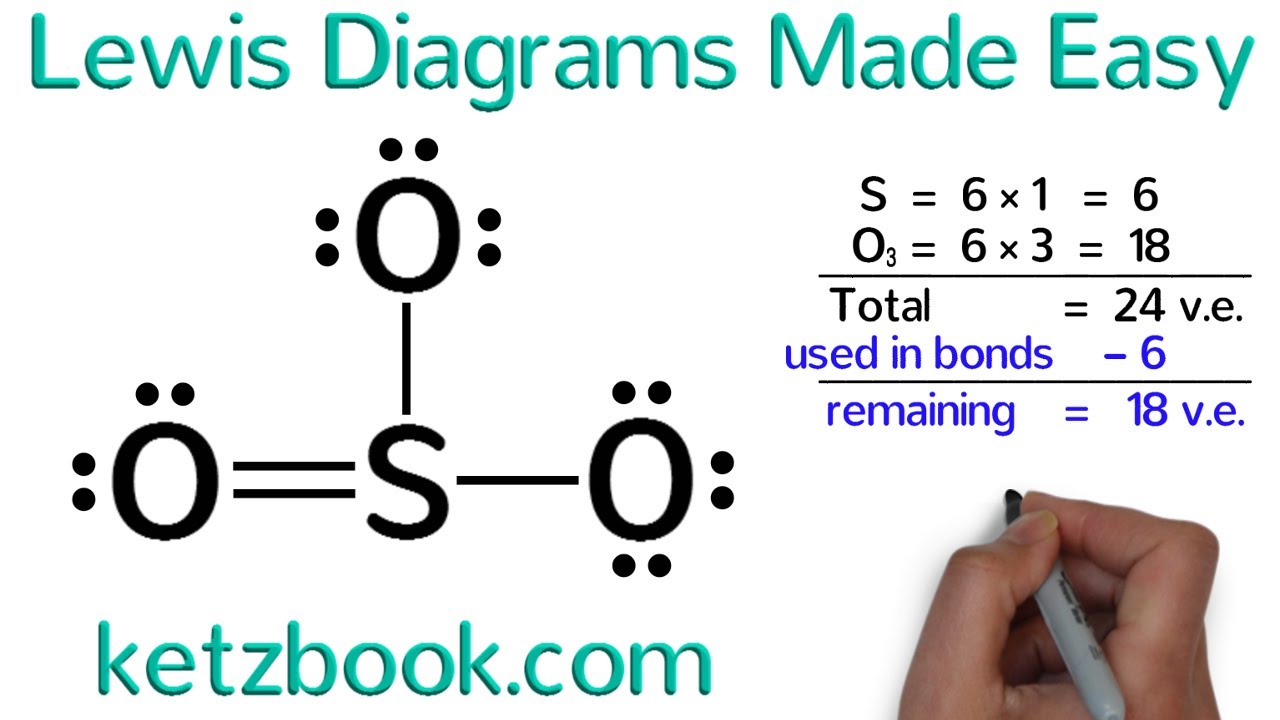
Different forms of Lewis structures
Expanded octet (elements in period 3 and below)
Incomplete octet (boron compounds)
Resonance structures
Structures with formal charges
Lewis structures – purpose
Show valence electrons
Show bonding and lone pairs
Help predict molecular geometry and polarity
Drawing Lewis structures
Count total valence electrons
Identify central atom (usually least electronegative)
Connect atoms with single bonds
Distribute remaining electrons to satisfy octets
Add multiple bonds if needed
Check formal charges
Molecular geometry
Determined by number of electron groups around central atom
Lone pairs and bonds both affect shape
Geometry minimizes electron repulsion
Identify valence electrons
Based on group number for main-group elements
Used to determine bonding capacity
Critical for Lewis structures
Lone pairs
Nonbonding electron pairs on an atom
Increase electron repulsion
Reduce bond angles compared to ideal geometry
Bonding angles
Depend on number of bonds and lone pairs
Lone pairs cause greater repulsion than bonding pairs
More lone pairs → smaller bond angles
Multiple bonds
General
Form when single bonds do not satisfy octet
Share more than one pair of electrons
Increase bond strength and decrease bond length
Single bonds
One shared pair of electrons
Longest and weakest type of bond
Double bonds
Two shared pairs of electrons
Shorter and stronger than single bonds
Triple bonds
Three shared pairs of electrons
Shortest and strongest type of bond
Bond length vs bond strength
Shorter bonds are stronger
Longer bonds are weaker
As bond order increases, length decreases and strength increases
HONC rule
Typical bonding patterns:
H → 1 bond
O → 2 bonds
N → 3 bonds
C → 4 bonds
Helps check if Lewis structure is reasonable
Formal charges
Used to evaluate the best Lewis structure
Formula:
Formal charge = valence electrons − (nonbonding electrons + ½ bonding electrons)
Helps determine electron distribution accuracy
Determine the best structure
Structure with the lowest total formal charge is preferred
Structures with most atoms having a formal charge of 0 are best
If charges exist, negative charge should be on the more electronegative atom
Polarity
Describes how unevenly electrons are shared in a bond or molecule
Results from differences in electronegativity between atoms
Covalent bonds
Electrons are shared
Nonpolar covalent bond
Electrons are shared equally
Electronegativity difference is very small or zero
Example: H–H, O=O
Polar covalent bond
Electrons are shared unequally
One atom pulls electrons closer
Creates partial charges: δ⁺ and δ⁻
Example: O–H, N–H
Ionic bond
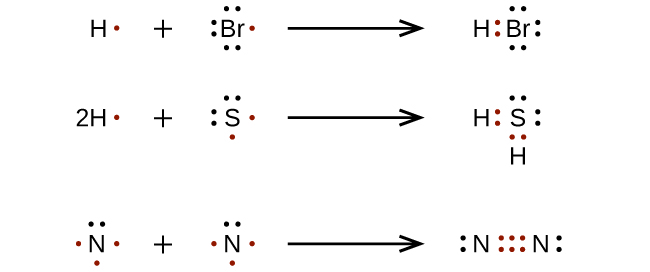
Very large electronegativity difference
Electron transfer occurs
Full positive and negative charges form
Bond polarity
Determined by electronegativity difference between bonded atoms
Larger difference → more polar bond
Molecular polarity
Depends on both bond polarity and molecular geometry
A molecule can have polar bonds but still be nonpolar overall
Symmetry
Symmetrical molecules cancel out dipoles
Result in a nonpolar molecule
Example: CO₂, CH₄
Asymmetry
Asymmetrical shape causes dipoles to add
Results in a polar molecule
Example: H₂O, NH₃
Dipole moment
Direction of electron pull in a bond
Points toward the more electronegative atom
Effect of lone pairs
Lone pairs increase asymmetry
Often make molecules polar
Example: H₂O is bent due to lone pairs
Steps to determine polarity
Determine if bonds are polar
Identify molecular geometry
Check if dipoles cancel or reinforce
Decide if molecule is polar or nonpolar
Solubility rule
“Like dissolves like”
Polar substances dissolve in polar solvents
Nonpolar substances dissolve in nonpolar solvents
Common highly electronegative atoms
F, O, N, Cl
Intermolecular forces
Intermolecular forces are attractions between molecules, not within them
They are weaker than covalent or ionic bonds but affect boiling point, melting point, and solubility
Dipole–dipole forces

Occur between polar molecules
The positive end (δ⁺) of one molecule attracts the negative end (δ⁻) of another
Stronger than London dispersion forces
Example: HCl molecules attracting each other
Hydrogen bonding
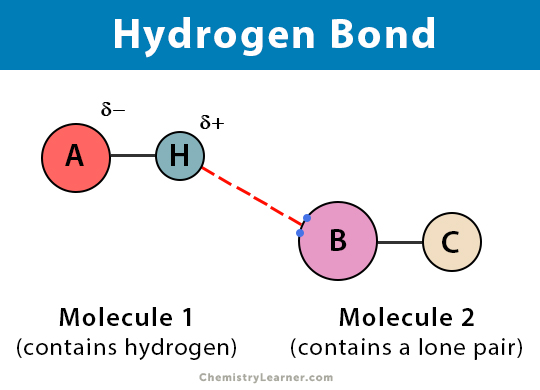
A strong type of dipole–dipole force
Occurs when hydrogen is bonded to N, O, or F
Hydrogen is strongly attracted to N, O, or F on nearby molecules
Explains high boiling point of water
London dispersion forces
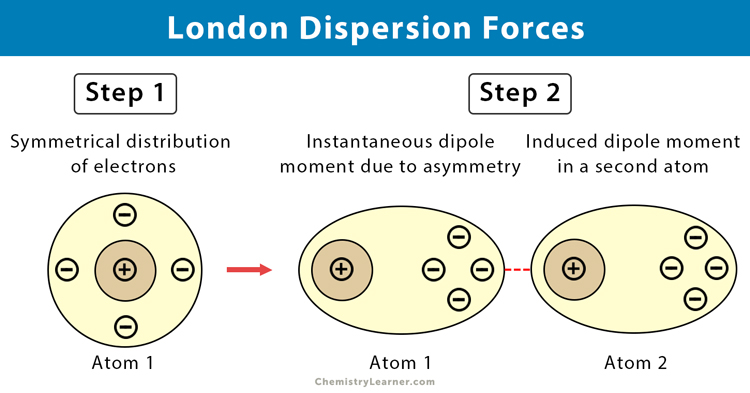
Occur in all molecules
Caused by temporary, uneven electron distribution
Only force present in nonpolar molecules
Weakest intermolecular force
Strength increases with larger molecules and more electrons
Strength order (weak → strong)
London dispersion
Dipole–dipole
Hydrogen bonding
Unit 5
Moles to grams
Used when given moles and asked for mass
Multiply by molar mass (g/mol)
Formula:
grams = moles × molar mass
Molar mass is found by adding atomic masses from the periodic table
Example idea: more moles → more grams
Grams to moles
Used when given mass and asked for amount
Divide by molar mass
Formula:
moles = grams ÷ molar mass
Always use correct units (grams and g/mol)
Balancing chemical equations
Based on the law of conservation of mass
Number of each type of atom must be equal on both sides
Only change coefficients, never subscripts
Start by balancing elements that appear once on each side
Leave hydrogen and oxygen for last
Check your work by recounting atoms
Coefficients
Numbers placed in front of formulas
Multiply the entire compound
Example: 2H₂O = 4 H atoms and 2 O atoms
Common balancing tips
Balance metals first, nonmetals next
Treat polyatomic ions as a single unit if unchanged
Reduce coefficients to the smallest whole numbers
Double-check all atoms at the end