Oct 24th - Intracellular membrane traffic
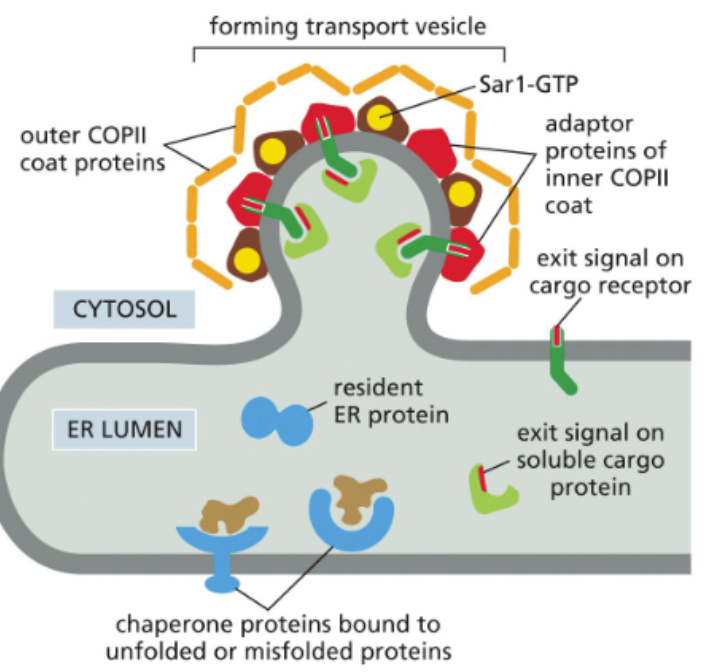
COPII-coated vesicles travel from the ER to the Golgo
soluable cargo proteins have exit signals that are recognized by cargo receptors
vesicles are formed and coated in COPII at ER exit sites
proteins need to be correctly folded and all units assembled in multi-unit proteins (quaternary structure) before departure - prevents misfolded proteins from moving forward
e.g. cystic fibrous
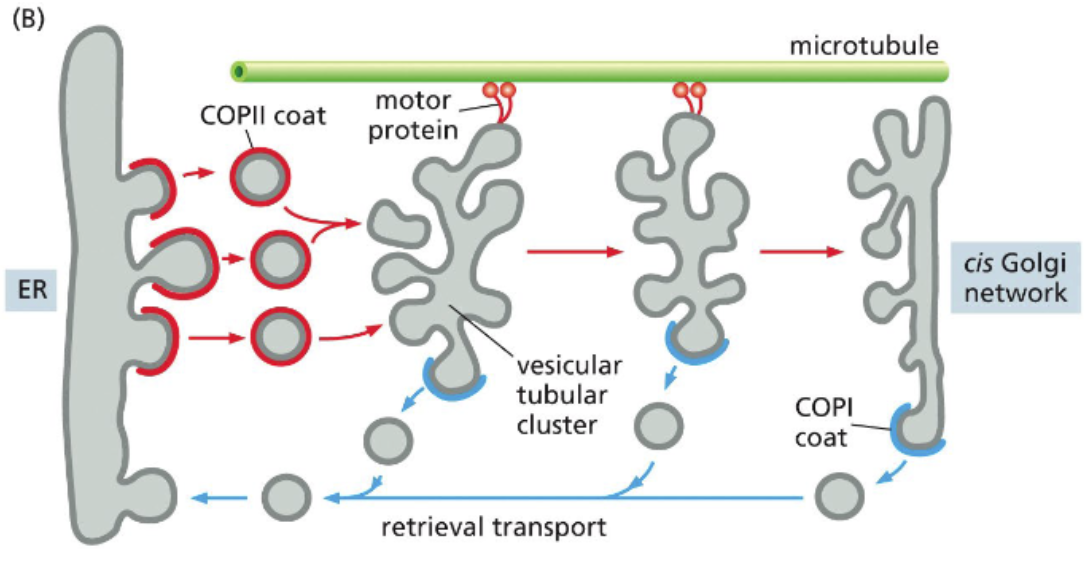
COPII-coated vesicles fuse into the vesicular tubular clusters that are carried to the Golgi
COPII-coated vesicles budding from the ER fuse to form a bigger tubular structure — vesicular tubular structure
vesicular tubular structure travels along microtubules to fuse with the Golgi
as it moves, COPI-coated vesicles bud off, carrying ER cargo
uses retrieval pathway to transport proteins like BiP and SNAREs back to the ER
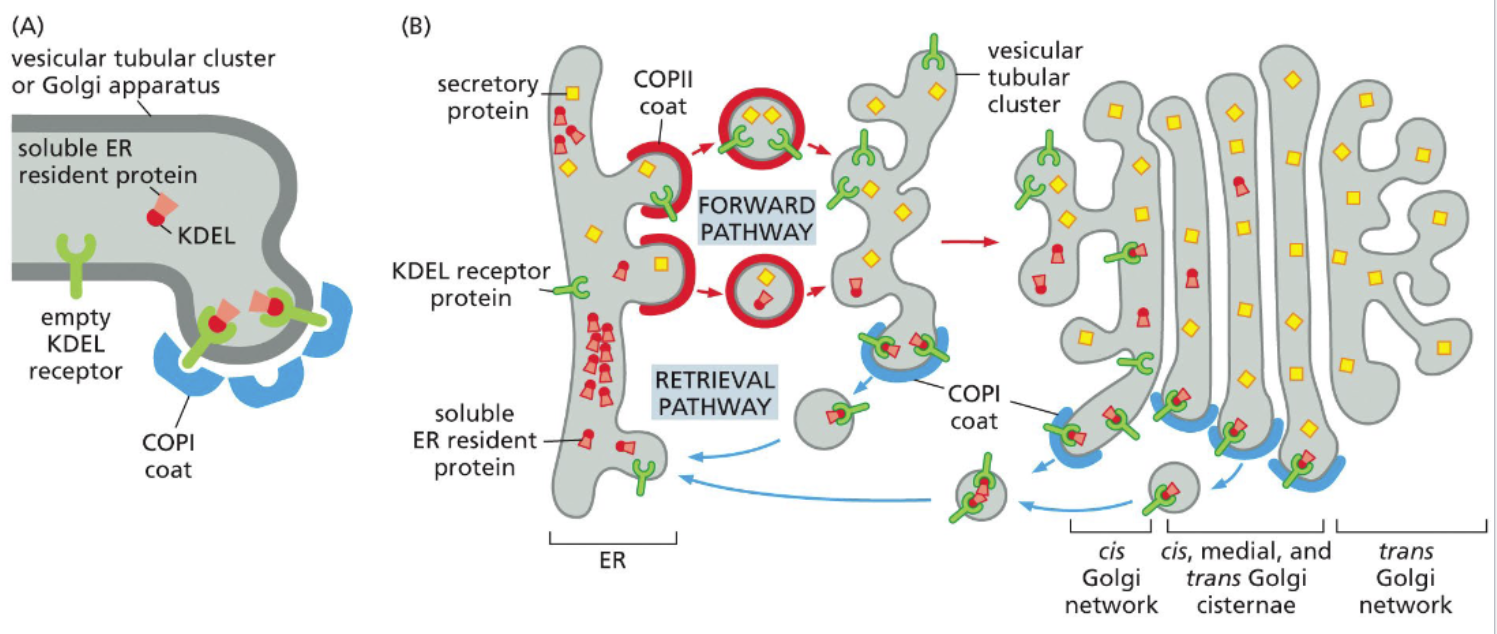
KDEL is a return signal for resident ER proteins
cargo that are retrieved have retrieval signals
e.g. KDEL in BiP is recognized by KDEL receptors — bind escaped BiP
this is followed by COPI-coated vesicle formation
the Golgi apparatus is a protein modifying and sorting system
it is a collection of flattened membranous compartments called cisternae
protein modification happens progressively as they move up from cis to trans cisternae
vesicles carrying the modified proteins bud off from trans cisternae
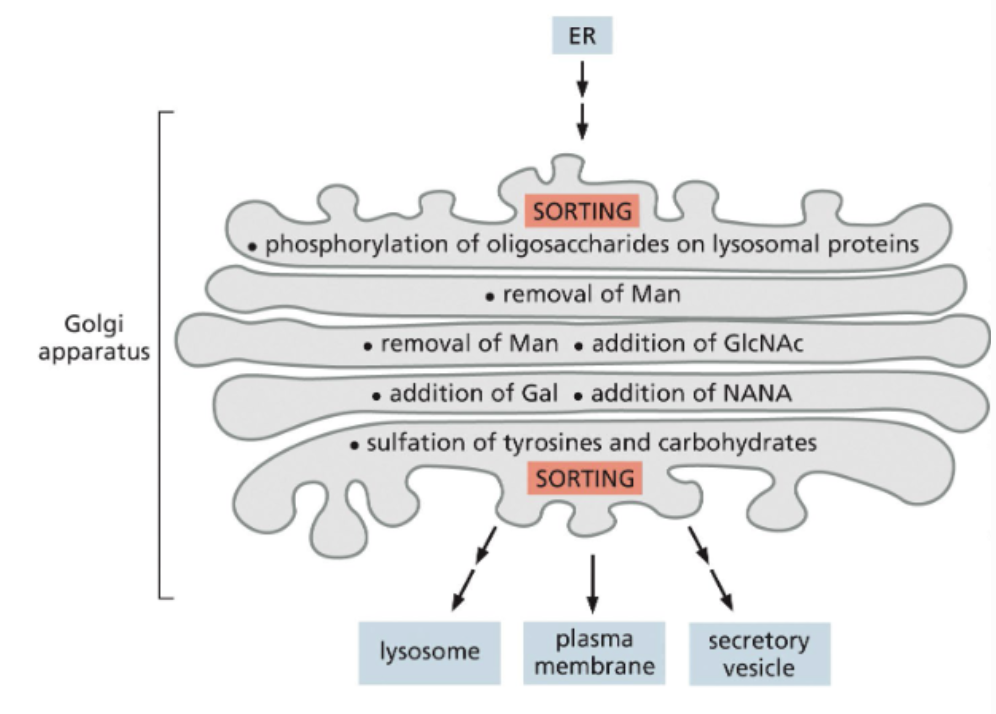
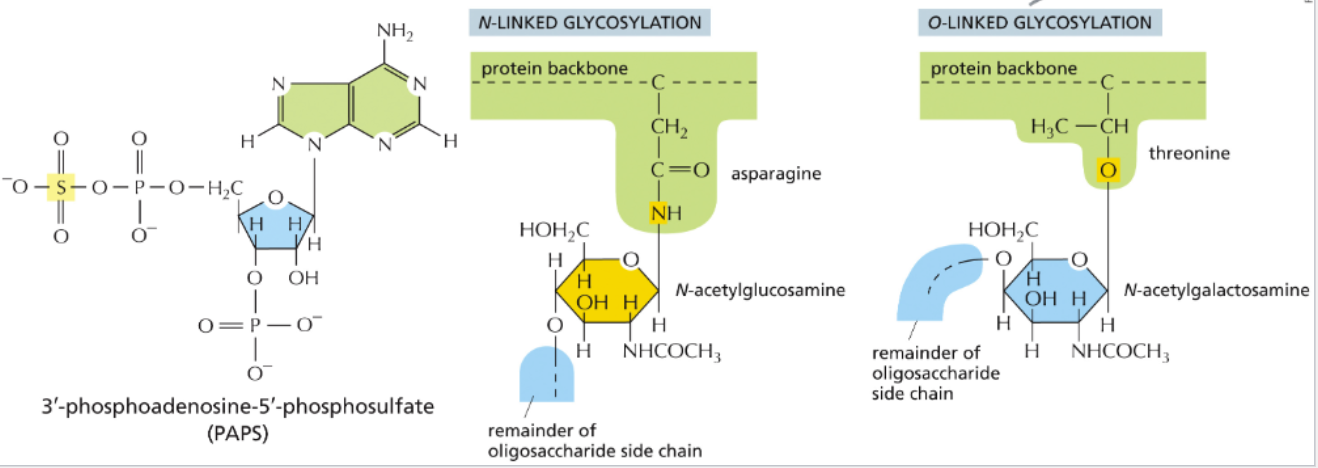
N-linked oligosaccharides started in the ER are modified in the Golgi
glycosylation contributes to the multiple functions
protection of proteins from proteases
facilitating folding by shielding hydrophobic regions, preventing aggregation and recruiting chaperones that recognize a “glycol-code” of sugar patterns
fine-tuning protein-protein interactions
the glycocalyx might have evolved as an animal version of a cell wall
a group of glycoproteins called proteoglycans contains sulfated sugars that are modified in the Golgi