How can we apply what we have learned?
Core and valance electrons
• Opposite charges attract, like charges repel
• Consider a Li atom, electron configuration: 1s22s1
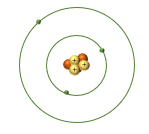
• There are Core electrons inner electrons
• Held tightly to the nucleus due to the electrosatic attraction
• Do not take part in bonding as they are held closely to the elevtron
• Valence electrons
• Electrons in outer shell
• Held less tightly due to shielding from core electrons.Sheilding is when there are core electrons inbetween the nucleus and the valence electrons so the valence electrons experience some repulsion from those elecrtons
• Participate in bonding and responsible for a lot of chemistry
Photoelectric effect
• Discussed in context of wave-particle duality of light in lecture 6
• Energy of the electron emitted depends on energy of electronic shells
• Can distinguish different elements
• Important applications in analytical chemistry and forensics
• Photoelectron spectroscopy (photoemission spectroscopy)
Photoemission spectroscopy
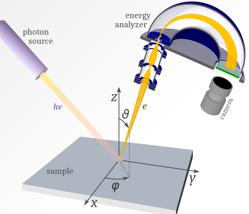
• Lots of different variations:
• Incident (lamda) (X-ray, UV)
• Method of analysis is that a light source which shines light on the sample.The light source has a variable wavelength so the energy of light interacting with the sample can be changed.
• Same principle – measures energy of ejected electrons.The electrons go up into a detector to determine their energy and therefore the sample.
• Light only penetrates into material a few nm so surface analysis technique
• Non-destructive
• Very sensitive so small amounts of elements can be detected so great for trace analysis
• High resolution
XPS vs UPS
X-ray Photoelectron Spectroscopy (XPS)
• Probes core electrons
Ultraviolet Photoelectron Spectroscopy (UPS)
• Probes valence electrons and can look into chemical bonding
• Which is higher energy? (Lecture 6) UV is lower in energy due to its shorter wavelength.X-rays are higher in energy which allows it to probe and remove core electrons.
Basic Principle
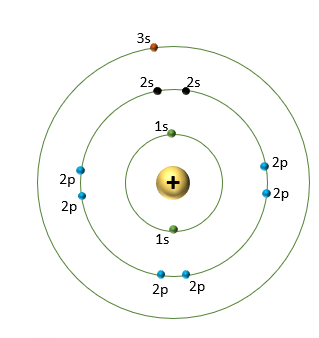 Sodium element number 11 is what we are intrested in detecting. The energy from a photon is used to eject a valence electron. We then end up with a spectra.
Sodium element number 11 is what we are intrested in detecting. The energy from a photon is used to eject a valence electron. We then end up with a spectra.
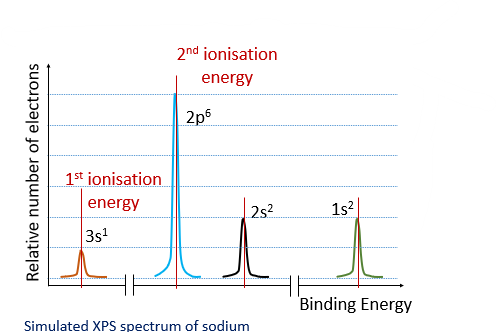 The hight of the peak tells us how many electrons are removed while the position shows how much energy is needed to remove the electrons from around the nucleus.The further along the peak the closer the electron to the nucleus.
The hight of the peak tells us how many electrons are removed while the position shows how much energy is needed to remove the electrons from around the nucleus.The further along the peak the closer the electron to the nucleus.
Real spectra
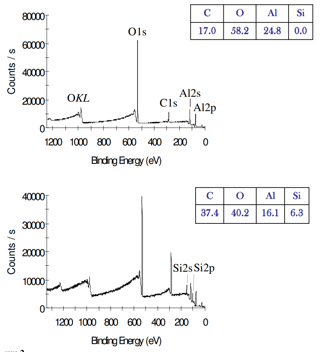
• Specific values of binding energy (where peaks appear) depend on specific atom
• Also used in peak assignment
• Can obtain quantitative information about ratio of different elements
• Real spectra are more complicated but the theory is the same
Chemical analysis of fingerprints
• Can identify a person with fingermarks
• Chemical analysis of fingermarks this is looking at the chemicals deposited in the ridges of the fingerprint.
• Link person and activity
• E.g. gun shot residue
Atomic and emmission spectra from lecture 6
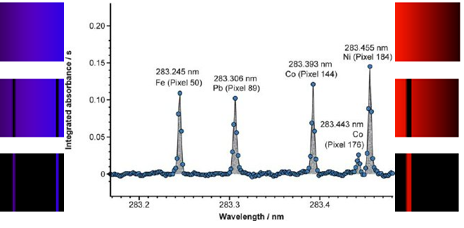
The sample is mixed with acetylene and oxygen so it can catch light and is then burnt in a flame a light is passsed through the flame and the light will encounter atoms from the sample and excite electrons, and certain regions of the light spectrum will be absorbed.Then we can observe the particular wavelengths and energies.The sample gets destroyed in the process.
• AAS applications
• Quality control, determination of origin – analytical chemistry
• Counterfeiting – forensic science
Why does understanding bonding and reactivity matter?
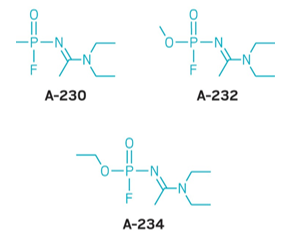
• Novichok nerve agents
• Developed in secret in former Soviet Union
• Classified in West
• Salisbury poisonings 2019, a former KGB agent was poisened by one of these agents
• How to determine what the poison was?
• Native form,how it came in the bottle
• Binding to other chemicals
• Decomposition products,how does it react withe the air ect.
• Requires a fundamental understanding of the bonding and reactivity
Covalent boding
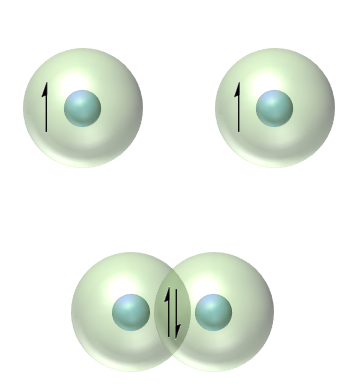
• More on this later in the course
• Covalent bonds form when electrons are shared between atoms (particle model of electron)
• Actually a result of interference of waves (wave model of electron)
• If there are valence electrons that are tightly held close to the nucleus covalent bonding ocours
• Close overlap required as the electrons are relativley close to the nucleus
• Short range, directional
Hydrogen
• Waves of atomic orbitals can interact in-phase or out-of-phase see lecture 6
• Create molecular orbitals out of atomic orbitals
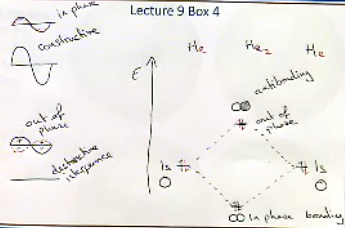 In lecture 6 we saw that waves can be added together constructivly which occurs when waves are in phase.Destructive interferance occours when the waves are out of phase and the two waves cancel out.
In lecture 6 we saw that waves can be added together constructivly which occurs when waves are in phase.Destructive interferance occours when the waves are out of phase and the two waves cancel out.
If we put two hydrogen atoms on an energy scale we can see how they come together to make a H2 molecule.The two atoms can come together in phase and have constructive interferance to create a bigger wave.We can see this molecular orbital at the bottom of the diagram.The atoms can also come together out of phase,the two diffrent colours shoe the diffrent phases.
The out of phase H2 is higher in energy on the diagram.Where there is an in phase combination we call it a bonding orbital.Where there is an out of phase combination or where there is no electron denisty between the atoms we call it an antibonding orbital.In a bonding orbital the energy of both the electrons is decreased o this ia a more favourable outcome in nature.
• Valence electrons a long way from the nucleus, not held very tightly
• Multiple atoms can overlap without nuclei repelling
• Does not require close overlap
• Electrons can delocalise across the whole system
• Conduct electricity
• Bond energy = 4 – 120 kJmol-1
• Oxygen is electronegative so O-H bonds are polarised
• Not formal +ve and –ve charges
• Separation of charge to form a dipole
• Can interact with other dipoles to form H-bonds
• Very important in biological chemistry
Bond energy < 5 kJmol-1
Polarisation of an electron cloud by adjacent nucleus
Weak electrostatic interaction
Ion-induced dipole