Carboxylic acid
how to test for carboxylic acid = add a carbonate (effervescence = positive test)
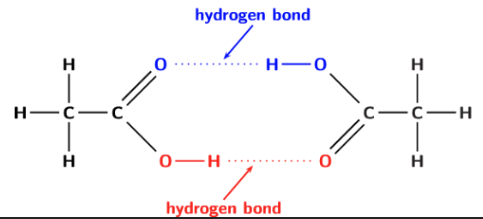
the first 4 carboxylic acids in the homologous series are very soluble in water
why are carboxylic acids soluble?
C=O bonds are polar and O-H bonds are polar —> this allows them to form hydrogen bonds w water
as the chain length increases the non-polar chain forms a larger proportion and has a greater effect in the overall polarity of the module
as chain length increases solubility decreases (more London forces surrounding carbon making it harder for non-polar bonds to interact)
carboxylic acids only partially dissociate in water making them weak acid
Carboxylic acid reactions
carboyilic acids undergo the following reactions
redox reactions with metals
2CH3CH2COOH (aq) + Mg (s) 🡪 (CH3CH2COO-)2Mg2+ (aq) + H2 (g)
the following neutralisation reactions:
reaction with metal oxide
2CH3COOH (aq) + CaO(s) 🡪 (CH3COO-)2Ca2+ (aq) + H2O (l)
reaction with alkalis
CH3COOH (aq) + NaOH(aq) 🡪 CH3COO-Na+ (aq) + H2O (l)
reaction with carbonates
2CH3COOH (aq) + Na2CO3(aq) 🡪 2CH3COO-Na+ (aq) + H2O (l) + CO2