Chemistry Module 2: Introduction to Quantitative Chemistry
Law of conservation of mass
Mass cannot be created or destroyed, but only change from one form to another.
In a chemical reaction:-
Total mass of reactants = total mass of products.
Number of atoms of each element is conserved.
Types of Chemical Reactions
Synthesis (Combination) Reaction
Two or more reactants combine to form a single, more complex product.
General Form: A + B \rightarrow AB
Example: \text{2Mg(s)} + \text{O}_2\text{(g)} \rightarrow \text{2MgO(s)}
Decomposition Reaction
A single compound breaks down into two or more simpler substances.
General Form: AB \rightarrow A + B
Displacement (Redox) Reaction
An element reacts with a compound, displacing another element from the compound, involving changes in oxidation states.
General Single Displacement Form: A + BC \rightarrow AC + B
Precipitation (Double Displacement) Reaction
Two ionic compounds in solution exchange ions to form a new insoluble compound (precipitate) and another soluble compound.
General Form: AB + CD \rightarrow AD + CB (where one product is a precipitate)
Complete Combustion
A rapid reaction with oxygen, producing carbon dioxide and water vapor (for hydrocarbons) when there's sufficient oxygen.
General Hydrocarbon Form: CxHy + O2 → CO2 + H2O
Produces maximum energy → ALWAYS EXOTHERMIC
Incomplete Combustion
A reaction with oxygen when there's insufficient oxygen, producing carbon monoxide, carbon (soot), and/or carbon dioxide and water.
General Hydrocarbon Form: CxHy + O2 → CO + C + CO2 + H2O
Less efficient, produces less energy, and generates toxic carbon monoxide and soot.
Chemical reactions
Reactions form new substances by breaking and reforming chemical bonds.
New substances have new chemical and/or physical properties.
Atoms are rearranged to form different substances.
General equations (not detailed in transcript).
Stoichiometry
Stoichiometry: study of the quantitative aspects of chemical reactions.
Allows determination of the amount of reactants needed and how much product is made.
Helps to predict yield.
Preparing Solutions (Part 1)
Standard solution preparation steps (example):
Weigh potassium permanganate in a 100 mL beaker.
Dissolve the solid in water.
Transfer the solution to a volumetric flask, ensuring all solid is washed in.
Add water to the flask until just above the neck.
Use a pipette to top up the solution to the mark.
Place the stopper, invert to mix.
Mole concept
Mole is the counting unit for amount of substance.
A mole of substance contains N_A = 6.022 \times 10^{23} particles.
12 g of Carbon-12 contains N_A = 6.022 \times 10^{23} atoms. This one mole.
Mole: Particles
1 mole corresponds to a fixed number of particles, which can be atoms, molecules or ions.
Avogadro’s number
Avogadro’s number = N_A = 6.022 \times 10^{23}.
It is the number of particles in one mole of substance.
Particles may be atoms, molecules or ions.
Purpose: provides the bridge between the macroscopic and microscopic worlds.
Percentage composition
Percentage composition tells you the proportion (by mass) of the different elements in a compound.
Expressed as the percentage of the total mass of the compound.
Compounds are made of atoms in fixed whole-number ratios.
Empirical formula
Empirical formula is the simplest whole-number ratio of elements in a compound.
Molecular formula
Molecular formula is always a whole-number multiple of the empirical formula.
Limiting reagent
When the quantities of reactants are not in the exact stoichiometric ratios, there may not be enough of one reactant for both to fully react.
The limiting reagent is the reactant that is consumed first and limits the amount of product formed.
Part 3: Concentration and Molarity
Different ways to measure concentration:-
Perform a dilution: the reactant completely consumed is the limiting reagent and the one not completely consumed is in excess.
Percent by weight (% w/w):
Describes mass of solute, in grams, per 100 g of solution.
Formula: C = \frac{\text{weight of solute}}{\text{weight of solution}} \times 100\%\; (\% w/w)
Percent by volume (% v/v):
Describes volume of solute, in mL, per 100 mL of solution.
Formula: C = \frac{\text{volume of solute}}{\text{volume of solution}} \times 100\%\; (\% v/v)
Parts per million (ppm):
Used when finding concentration of solute in a solution is very small.
Units can be expressed in several ways:-
\text{ppm} = \frac{\text{mass of solute (mg)}}{\text{mass of solution (kg)}}
\text{ppm} = \frac{\text{volume of solute (mL)}}{\text{volume of solution (kL)}}
\text{ppm} = \frac{\text{mass of solute (mg)}}{\text{volume of solution (L)}}
Dilution: adding water to a solution.
Solution of known concentration can be made by diluting a more concentrated solution.
Dilution example (Steps for generating dilutions):
Transfer 25.00\text{ mL} of Solution 1 into a 100.00\text{ mL} volumetric flask using a digital pipette.
Fill the volumetric flask to the calibration mark following method Part A.
Complete the table for Solution 2.
Repeat steps 1–3 to create 2 successive dilutions, resulting in 4 volumetric flasks of dilutions.
Practical note: For dilutions, the key relationship is C1V1 = C2V2
Part 4: Gas laws
Gases
Properties of gases:-
Low density: gas molecules dispersed in a given volume; less dense than solids or liquids.
Indefinite shape or volume: gases have no definite shape or volume; they expand to fill the container.
Compressibility and expandability: respond to changes in temperature and pressure.
Diffusivity: gases mix quickly to form homogeneous mixtures (diffusion).
Pressure: gas molecules in constant motion exert pressure on container walls; depends on amount of gas, volume, and temperature.
Kinetic theory of Gases
Explains the effects of temperature and pressure on matter.
Assumes all particles are in constant motion.
Four assumptions for ideal gases:
All gases are composed of small particles and the volume of gas particles is negligible (most of the volume is empty space).
The particles move in continual, random, rapid motion with no attraction or repulsion between particles.
All collisions are perfectly elastic (energy is conserved in collisions).
The average kinetic energy of particles increases as temperature increases.
Ideal gas vs real gases
Real gases have small attractive and repulsive forces between particles; ideal gases do not.
Real gas particles have volume; ideal gas particles have negligible volume.
Real gas collisions may be inelastic (energy can be lost); ideal gas collisions are elastic.
The term "Real gas" refers to actual gaseous substances; "Ideal gas" is a hypothetical model.
Summary of gas behavior
Real gas: experiences intermolecular attractions; has finite particle volume; collision energies can change.
Ideal gas: no intermolecular attractions; point particles; perfectly elastic collisions; constant kinetic energy per temperature.
Gas quantities and models
Avogadro’s law / Gas concept: equal volumes of gases contain the same number of molecules at the same temperature and pressure (translated through discussion of Avogadro’s number and molar volume).
Avogadro’s law (and related notes)
Vi/ni = Vf/nf
equal volumes of all gases, at the same temperature and pressure, have the same number of molecules
at STP (0°C, 100kPa) → molar volume of all gases is 22.71L
at RTP (25°C, 100kPa) → molar volume of all gases is 24.79L
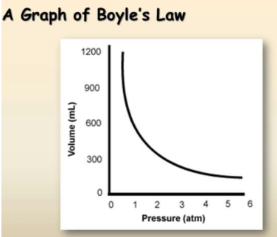
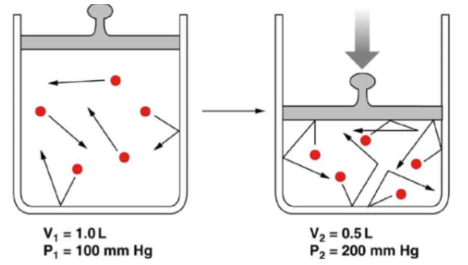
Boyle’s law
Relationship between pressure and volume at constant temperature and constant amount of gas:-
Real gas: may approximate the law under certain conditions.
Ideal gas: PV is constant at constant temperature and moles, thus P1V1 = P2V2 when T and n are constant.
Key point: As volume decreases, pressure tends to increase (for a fixed amount of gas).
Ideal gas law (general)
The ideal gas law relates pressure, volume, amount of gas, and temperature:-
PV = nRT
Where:-
P = pressure,
V = volume,
n = number of moles,
R = ideal gas constant,
T = absolute temperature (Kelvin).
The transcript notes mention a form: \frac{Vi}{ni} = \frac{Vf}{nf}, which reflects Avogadro’s principle (equal volumes contain equal numbers of molecules) rather than the full ideal gas law. This transcript line is kept for reference alongside the standard ideal gas law.
Molar volume and STP
At STP (as defined in transcript: approximately 100 kPa and 0 °C, i.e., 273.15 K), the molar volume is approximately:-
V_m = 22.71\ \text{L mol}^{-1}
Molar volume (Vm) is the volume occupied by one mole of any gas at a given temperature and pressure.
The amount of gas can be expressed as:-
n = \frac{V}{V_m}
At STP, Vm is the same for all gases (ideal condition).
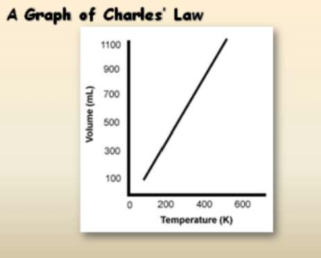
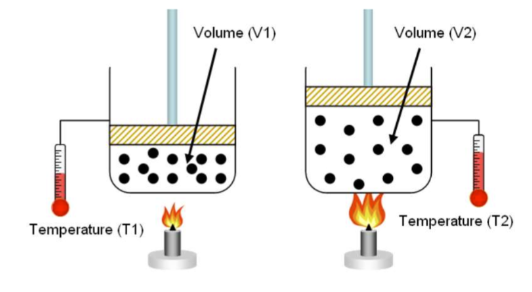
Charles’ law
Also known as a gas law describing how gases tend to expand when heated:-
When pressure is held constant, the kelvin temperature and volume are directly related.
V1/T1 = V2/T2
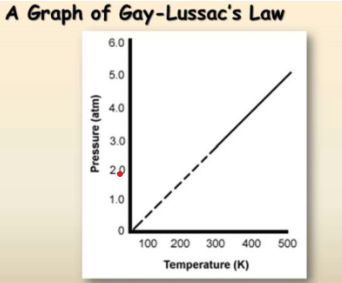
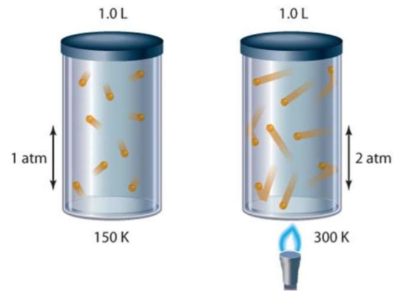
Gay Lussac’s law
Describes how gases expand with temperature at constant volume (in the transcript):-
P1/T1 = P2/T2
Combined gas law
The transcript notes it is also known as the law of volumes and describes how gases tend to expand when heated.
Given relations in transcript:-
Temperature-related form: \frac{V}{T} = \text{constant} (for a process at constant n and P or under a specified condition in the context).
Pressure-related form: \frac{P}{T} = \text{constant} (for another context described).
Important general form (not explicitly stated in transcript but standard):-
\frac{P V}{T} = \text{constant} for a fixed amount of gas.
In full form (combined): \frac{P V}{n R T} = 1 \Rightarrow PV = nRT.
Connections and big-picture notes
The law of conservation of mass underpins all chemical equations: matter is neither created nor destroyed in a closed system, guiding stoichiometric calculations.
Stoichiometry connects macroscopic measurements (grams, liters) to the microscopic world of atoms and molecules via molar amounts.
Avogadro’s number provides the bridge between moles and actual particles, enabling conversion between microscopic and macroscopic scales.
Concentration concepts (% w/w, % v/v, ppm) support quantitative solution chemistry, including dilutions and preparation of standard solutions.
Gas laws unify the behavior of gases under varying P, V, T, and n, enabling quantitative predictions of gas behavior in real-world contexts such as chemical reactions in gases, gas mixtures, and industrial processes.
The STP molar volume concept (Vm) offers a convenient, approximate way to relate gas volumes to the amount of gas when conditions align with STP, highlighting the idea that many gases behave similarly under these standardized conditions.
Practical and ethical/philosophical implications
Quantitative chemistry underpins safe, efficient chemical manufacturing, environmental monitoring, and pharmaceutical development; precise measurement and calculation are essential to avoid hazardous conditions and ensure product quality.
Understanding the limits of idealized models (e.g., ideal gas law) reminds us that all models are approximations; real-world systems exhibit deviations that must be accounted for in precise engineering.