Lab Techniques- Distillations
5.3 Distillations
Distillation: the process of raising the temperature of a liquid until it can overcome the intermolecular forces that hold it together in the liquid phase. the vapor is then condensed back into the liquid phase and subsequently collected in another container
Distillation is used to separate and purify chemicals based on their respective boiling points. The process uses a series of vaporizations and condensations.
Simple distillation: used when compounds have BPs that are 25-30°C or more apart
Fractional distillation: used when compounds have Bps within a range of 25-30°C of each other
Simple Distillation
Used when trace impurities need to be removed from a relatively pure compound or when a mixture of compounds with significantly different boiling points needs to be separated.
Equipment used:
Oil bath: oil doesn’t evaporate when you heat it, so it is used to keep a constant temperature throughout the process
Distilling flask: contains mix of compounds to be separated
Thermometer: used to measure what temperature the compounds are boiling out at
Condenser: kept cool by water cycling in and out. The cool temperature allows vampirized compounds to condense back into a liquid phase
Vacuum adaptor: allows the pressure of the entire system to be lowered, making vaporization easier
Receiving flask: collects the condensed liquid
Ice bath: keeps the receiving flask cold so that the liquid can readily condense back into its pure form
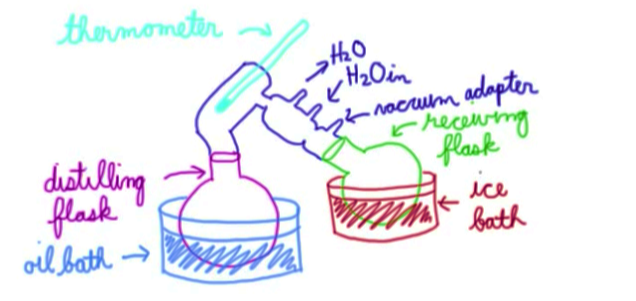
Process:
Liquid in the distilling flask gets heated to its boiling point, at which it vaporizes into a gas
As a gas, it travels up and enters the condenser, whereby the cool temperature causes it to condense back into a liquid
The liquid now collects in the cool receiving flask
Receiving flask is switched out for each compound present (or else you will end up with a mixed solution)
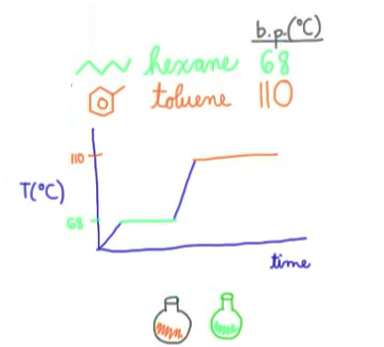
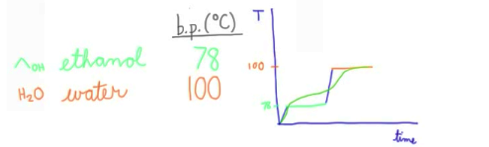
Distillation could be graphically shown like the image above. Plateaus present within the graph represent phase changes (liquid → gas), the temperature remains constant
Distillations can be used to also increase the concentration of a certain compound (by making it more pure)
however, this may prove difficult if compounds have similar boiling points and may require multiple distillations in order to achieve wanted product… which can be time consuming
If distillation graph lacks clear cut lines and plateaus like above (if curves are present), the products are not completely separated and need to be distilled again… again very time consuming
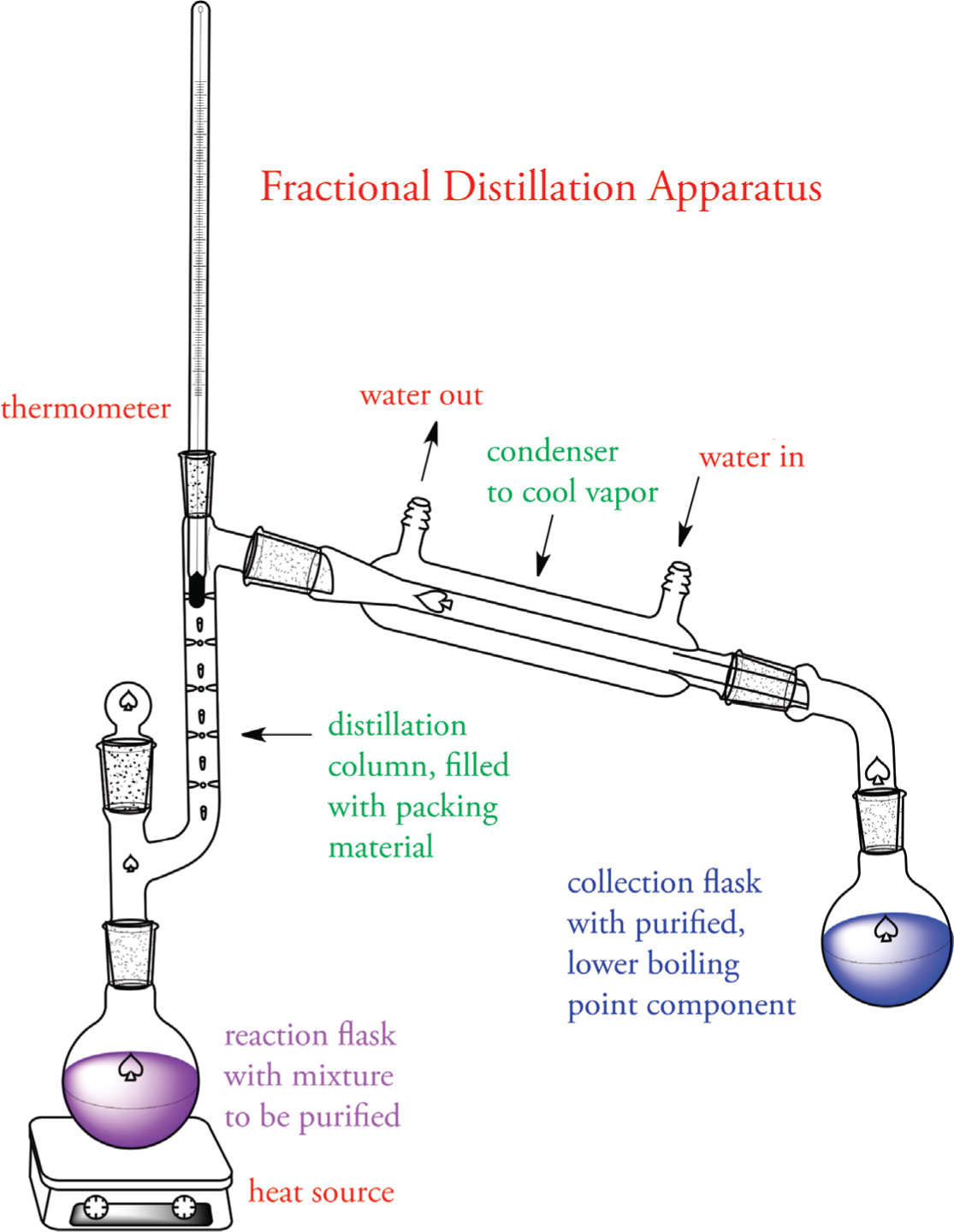
Fractional Distillation
Used when the difference in boiling points of the components within the liquid mixture are is not large
Equipment:
similar equipment but with the addition of a fractionating column
- the column contains beads or stainless steel sponge, which results in the liquid mixture being subjected to many vaporization-condensation cycles
- as cycles progress, the composition of the vapor gradually becomes enriched in the lower boiling component
- at the top of the column, the nearly pure vapor reaches the condenser and condenses back into the liquid phase where it is collected in the receiving flask
- yields a purer final collection