Topic 4 Inorganic Chemistry
Group 1 The alkali metals
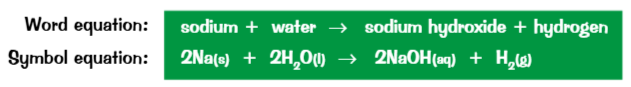
Group 1 elements all react in a similar way with water
Lithium sodium and potassium react vigorously in water
The reaction produces metal hydroxide solution. This solution is alkaline.
The reaction of the alkali metals with water also produce hydrogen this is why you can see fizzing.
They also react with oxygen to produce metal oxides.
Group 1 elements become more reactive as you go down the group
You can see the rate of reaction with the water
Lithium takes longer to react becuase it is the least reactive whilst francium is the most reactive
Atoms lose electrons more easily down the group
Group 1 metals have 1 electron in their outer shell
as you go down the group the outermost electron is in a shell thats further from the nucleus
Which means the attraction between the outermost electron and the nucleus become less.
as you go down the group the atoms get bigger the outer electron is more easily lost and the metals become more reactive due to this
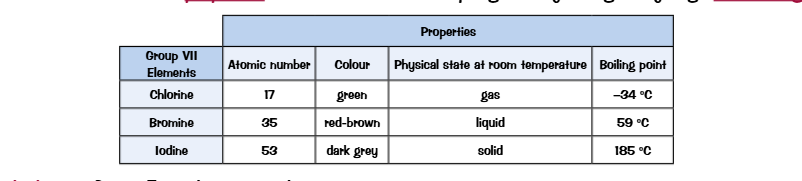
Group 7 The halogens
A the atomic number of the halogens increases, the elements have a darker color and a higher boiling point. This means at room temperature the higher up you go on the group the more reactive.
For example
Reactivity decreases going down the group 7
All group 7 elements have electrons in their outer shell, they can gain one electron from a 1- ion
The easier it is for a halogen atom to attract an electron the more reactive the halogen will me
As you go down group 7 the halogens become less reactive, it gets harder to attract the extra electron to fill the outer shell when its further away from the nucleus. (The atomic radius is larger)
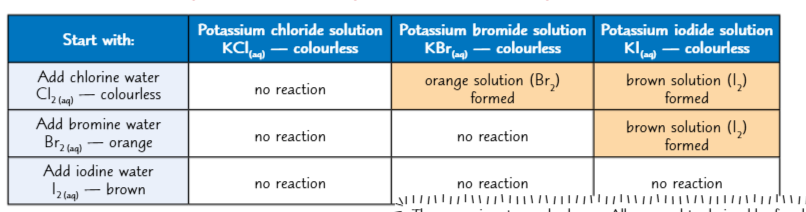
Displacement reactions
More reactive halogens will displace less reactive ones
The elements in group 7 take part in displacement reactions
A displacement reaction is where a more reactive element pushes out (Displaces) A less reactive element from a compound
So id you ass chlorine water to potassium iodide solution the chlorine will react with the potassium in the potassium iodide to form potassium chloride
The iodine is displaced from the salt and gets left in the solution turning it brown.
Halogen displacement reactions involve the transfer of electrons
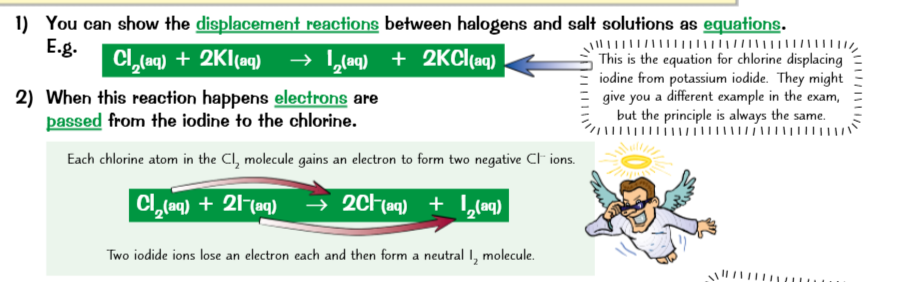
A loss of an electron is called oxidation. A gain in electron is called reduction.
Gases in the atompsphere
78% Nitrogen
21% Oxygen
1% Argon
0.04% carbon dioxide
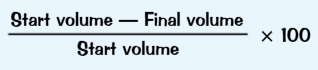
Iron can be used to determine the percentage of oxygen in the atmosphere
This is because iron reacts with oxygen in the air from rust, so iron will remove oxygen from the air.

To calculate the percentage of oxygen just put the volume recorded into this formula
The anser you get should be about 20%
Gases in reactions
Oxygen : When you burn something it reacts with oxygen in air.
Magnesium : Magnesium burns with a bright white flame in air and the white powder that is formed is magnesium oxide, magnesium oxide is slightly alkaline when it dissolved in water.
Hydrogen : Hydrogen burns very easily in oxygen, it can even be explosive. It has an almost invisible pale blue flame and the only product is water vapour. The combustion of hydrogen is often used a test for hydrogen gas, in small amounts the resulting explosion gives the characteristic of a squeaky pop.
Sulfur : Sulfur burns in air or oxygen with a pale blue flame and produces sulfur dioxide. Sulfur dioxide is acidic when it’s dissolved in water.
Carbon dioxide
carbon dioxide is a green house gas
The temperature of the earth is a balance between the heat it gets from the sun and the heat it radiates back out into space
Gases in the atmosphere like carbon dioxide , methane and water vapor natrual act like an insulating layer. They are often called green house gases. They absorb most of the heat that would normally be radiate out into space and re=radiate it in all directions including back towords earth.
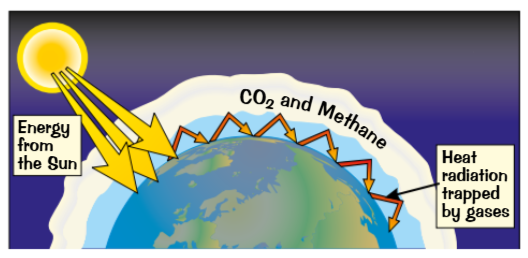
Human activity affects the amount of carbon dioxide in the atmosphere
Increasing carbon dioxide is linked t climate change.
The reactivity series
The reactivity series is how well a metal reacts.
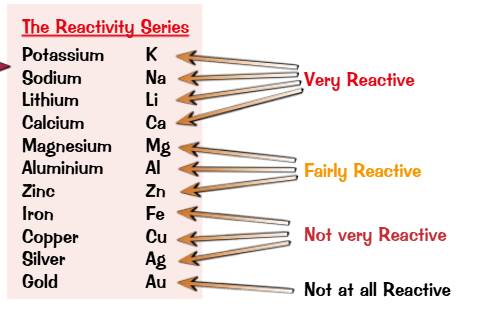
A more reactive metal displaces a less reactive metal
More reactive metals displace less reactive ones from compounds.
These are redox reactions: the metal is oxidised, and the displaced ion is reduced.
A reactive metal replaces a less reactive metal in a salt solution.
No reaction happens if the metal is less reactive than the one in the salt.
Displacement often causes a temperature change.
These reactions help identify a metal’s place in the reactivity series.
Iron
Iron and steel corrode to make rust
Iron corrodes (rusts) when exposed to oxygen and water.
Rusting is an oxidation reaction where iron gains oxygen to form iron(III) oxide.
Water binds to it, forming hydrated iron(III) oxide (rust).
Rust is soft and flakes off, exposing more iron to rust again.
Word equation:
iron + oxygen + water → hydrated iron(III) oxide (rust)
The 2 main ways to prevent rusting is with painting or coating with plastic or oiling it.

Metals and redox
Oxidation is the addition of oxygen , reduction is the removal of oxygen

Most metals are found in ores and need to be separated.
Unreactive metals (like gold) are found as pure elements.
Reactive metals form compounds in the Earth's crust, often as metal oxides.
More reactive metals are harder to extract.
Carbon is commonly used in reduction reactions to separate metals from oxides.
But carbon can't extract all metals — only some.
Uses of metals
. Aluminium
Properties: Low density, corrosion-resistant, strong (when alloyed)
Uses:
Aircraft (light and strong)
Food cans (doesn't rust)
Overhead power cables (low density and conducts electricity)
2. Copper
Properties: Excellent conductor, malleable, doesn't react with water
Uses:
Electrical wiring
Water pipes
Cooking utensils
3. Iron & Steel
Iron: Easily corrodes, but strong
Steel: Alloy of iron; stronger and resists rusting better
Uses:
Construction (bridges, buildings)
Car bodies
Tools and machinery
4. Gold
Properties: Very unreactive, shiny, malleable
Uses:
Jewellery
Electrical contacts (doesn’t corrode)
5. Titanium
Properties: Strong, low density, corrosion-resistant
Uses:
Aerospace
Artificial joints
Military equipment
Acids and Alkalis
An indicator is just a dye that changes color
Universal indicator : Is very useful for a combination of dyes high gives the colors shown to find the ph. of an Aqueous solution.
Litmus paper | Purple in neutral Red acidic Blue alkaline |
Phenolphthalein | Colorless in acidic Bright pink in alkaline |
Methyl orange | red acidic yellow alkaline |
Acids can be neutralized by bases or alkaline
A base is a substance that can neutralize an acid they are proton acceptors. Alkalis are soluble bases. An alkali is a source of hydroxide ions and has a ph greater than 7.
Reactions of acids
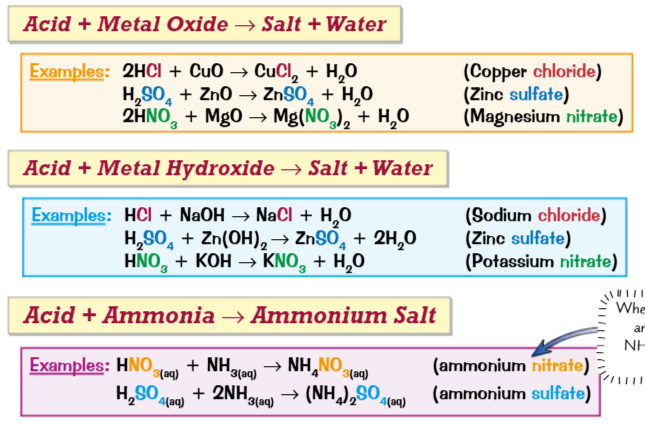
A salt, an ionic compound is formed during neutralisation reaction. This is a reaction between an acid and a base.
The type if salt depends on the acid used.
Acid + Base = Salt + Water

Titrations
A titration is an experiment used to find out how much acid is needed to neutralise a given amount of alkali, or vice versa.
Burette – contains the acid or alkali added drop by drop
Pipette – measures a fixed volume of the other solution (usually alkali)
Conical flask – holds the solution being titrated
Indicator – shows when neutralization happens (common: phenolphthalein or methyl orange)
Steps
Use pipette to add a measured volume of alkali into the conical flask.
Add a few drops of indicator to the alkali.
Fill the burette with acid.
Slowly add acid to the flask while swirling, until the indicator changes colour (shows neutralisation).
Record the volume of acid used (called the titre).
Repeat to get accurate, concordant results (within 0.10 cm³ of each other).
Helps calculate concentrations of acids or alkalis.
Can be used to make pure salts (in neutralization reactions).
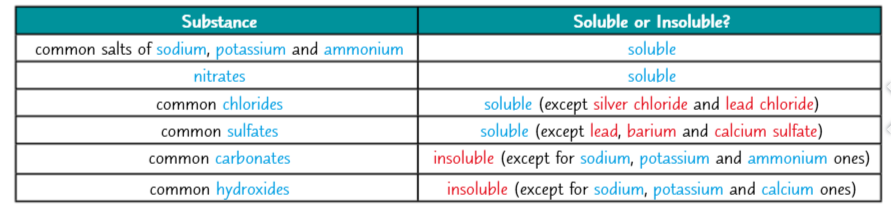
Making insoluble salts
The rules of solubility
How you make a salt depends on whether its soluble or insouluble
You may need to work out if, when wo solutions are mixrd a salt will form as a precipitate
This table is a pretty fail-safe way of working out whether a substance is soluble in water or not.
Tests for cations
Flame tests identify metal ions
Compounds of some metals burn with a characteristic color.

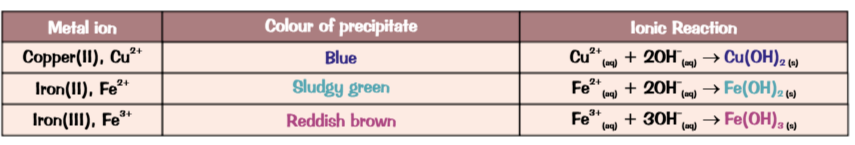
Some metals form a coloured precipitate with NaOH
Tests for anions



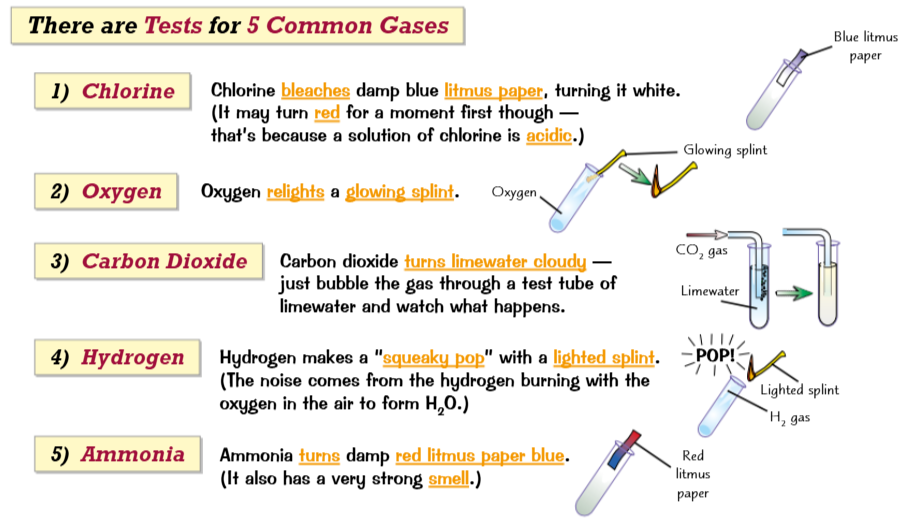
Tests for gases and water
Wet copper sulfate is blue - Dry copper sulfate is white
Color Differences:
Wet Copper(II) sulfate is blue due to its hydrated form.
Dry Copper(II) sulfate is white as it lacks water.
Water Interaction:
When copper(II) sulfate crystallizes, it absorbs water to form blue crystals.
Heating these hydrated crystals removes water, turning them white.
Adding Water:
If you add water to white copper(II) sulfate, it rehydrates and turns blue.
Water Purity:
Pure water has a defined boiling point (100°C) and freezing point (0°C).
Impurities can affect these points.