Y10 Chemistry
Separation Techniques
Independant variable - A variable that the chemist changes
Dependant variable - A variable that the chemist measures
Control variable - A variable the chemist has to make sure to keep the same
Solute - a substance dissolved in the solvent
Solvent - the substance the solute is dissolved in
Mixture - the end product of both mixtures
Soluble - able to dissolve
Insoluble - unable to dissolve
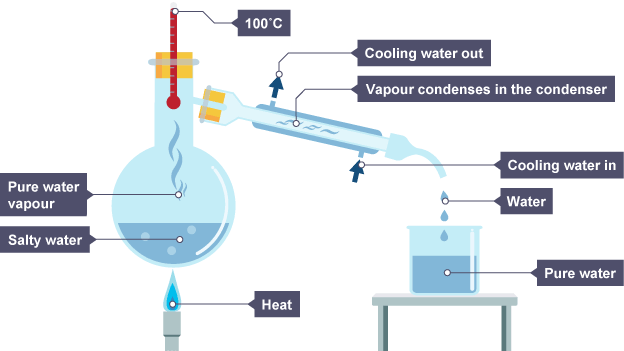
Simple distillation - Used to separate the solvent from a solution.
How simple distillation is used - As the dissolved solute has a much higher boiling point than the solvent, the heated solvent evaporates leaving the solute behind.
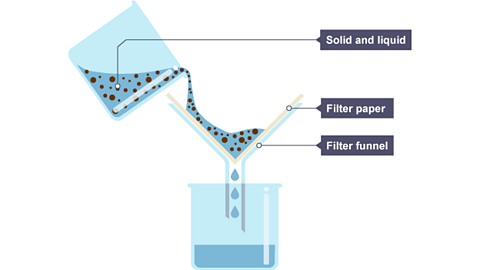
Filtration - used when the solute is insoluble
How filtration works - Filtration works by passing the mixture through a filter
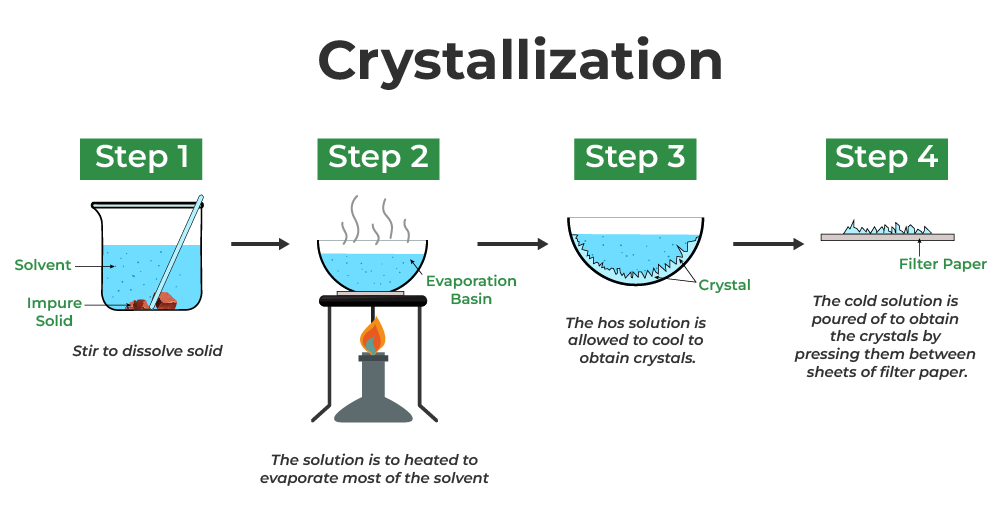
Crystallization - used when in need of purifying a solid such as soluble salt
How crystallization works - It works by dissolving the mixture/solid, and slowly evaporating it. As the two solutes and solvents have different boiling temperatures, they separate forming crystals
Paper chromatography - used to separate mixtures and to help identify the substances
How chromatography is used - Different colors are spread among a sheet of paper. The paper is later hanged above water. As the water moves, it pushes the color further helping identify it’s Rf value
Solubility of a substance - Defined on how far the substance travels on the chromotography sheet.
How the Rf value is defined - the Rf value is defined by dividing the travel of the substance and the length of the paper.

Atomic structure
Atoms - A thing all matter is made up of
Molecule - Formed when atoms chemically join and form bonds
Element - A substance made up of a single type of atoms
Compound - A substance made up of different types of atoms
Mixture - Two or more atoms NOT chemically bonded together
Pure substance - an element that boils/melts at a specific temperature
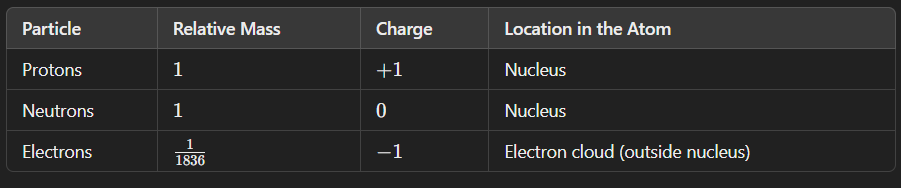
Atomic Mass - number of protons in atom
Atomic mass - number of both protons and neutrons in an atom
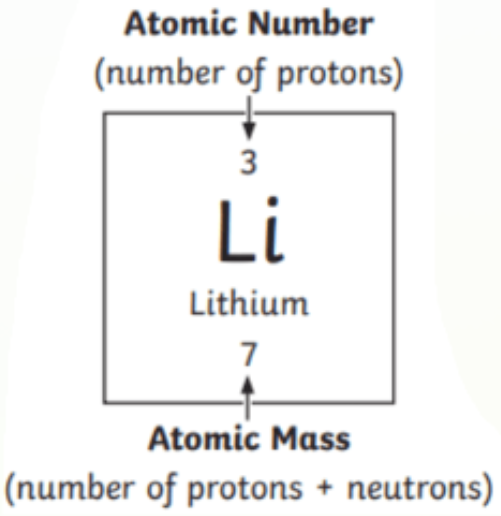
Isotope - an element that has the same amount of protons and neutrons
Relative atomic mass - weighted average of an atom
Period group - atoms with the same number of electrons in the outer shell. This makes the elements have the same chemical properties
Metals - Conductive elements that react to form positive ions
Non-metals - Non-conductive elements that react to produce negative ions
Chemical Reactions
The Law of conservation of mass - States that no matter is gained or lost in a chemical reaction