The Periodic Table / Metals and non-metals
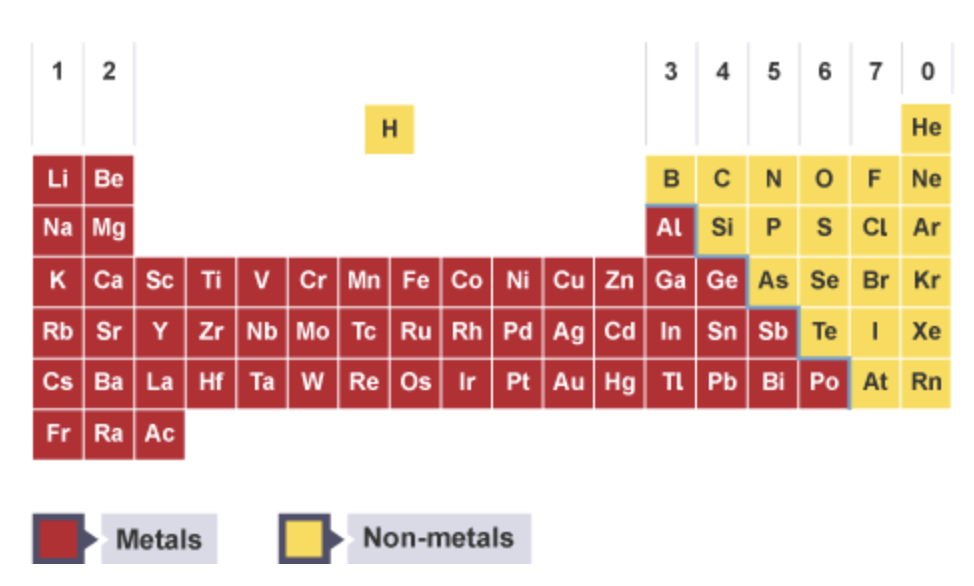
The periodic table is split into metals and non-metals
In the periodic table each letter represents a different element. These letters are called symbols.
An element is a pure substance made from one type of atom. Each element has a unique symbol made from sometimes one or two letters.
Chemically similar elements are put together in groups.
A chemical formula represent atoms and elemnts where more than one substance is bonded together.
An element is a substance that can’t be broken down. Each elemnt is made up of its won type of atom. Oxygen, Magnesium, Gold, Iron are all element.
A symbol is a letter that represents an element. Eg:
H = Hydrogen, C = Carbon, He = Helium, Mg = Magnesium
 Compounds
Compounds
A compound is a pure substance which is made from more than one element, and atoms, they chemically bond together to make a compound, and this means we can describe these compounds with a chemical formula.
Examples
Water is a compound of hydrogen and oxygen. The symbols H for hydrogen and O for oxygen are used to represent H₂O.
Oxygen gas is a molecule made of two oxygen (O) atoms represented as O₂.
Sulfuric acid is a combination of hydrogen (H), sulfur (S) and oxygen (O) represented as H₂SO₄.
H₂O | Two hydrogen atoms, one oxygen atom | 3 atoms in total |
CO₂ | One carbon atom, two oxygen atoms | 3 atoms in total |
Chemical properties
In physical changes, no new substances are formed
In Chemical reactions, new substances are formed.
During chemical reactions, colour changes may occur, a gas be formed or energy be released.
It is difficult to reverse chemical changes
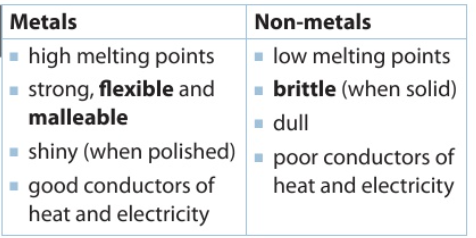
The chemical properties describe how a substance reacts with other substances
The chemical formula is the ratio of elements in a compound.
 Chemical trends
Chemical trends
Elements in the same group of the periodic table have similar physical properties and chemical properties
Conversion of mass is the formation of a new substance
Reactants are substances that react together to form the products
Product is the substances created in a chemical reaction
Compound is two or more elemnts chemically joined together
Oxidation is where a substance gains oxygen in a reaction
Salt is a a products formed when an acid reacts with a metal or a metal giving substances.
Reactivity series is a chart showing metals in order of decreasing reactivity
Metalloids have properties of both metals and non-metals