Water
Water
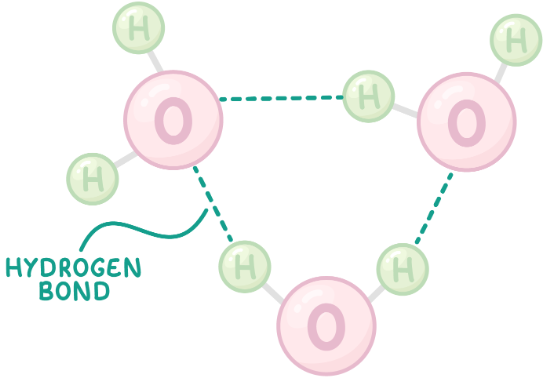
Water is a polar molecule
form hydrogen bonds
Roles of water
Solvent
many substances dissolve in water
Temperature control
water can buffer sudden temperature changes
Cooling mechanism
mammals use the evaporation of water as sweat to cool the skin
Habitat
many organisms can survive and reproduce in water
Metabolite
many chemical reactions involve water
Transport
organisms can use water to move substances
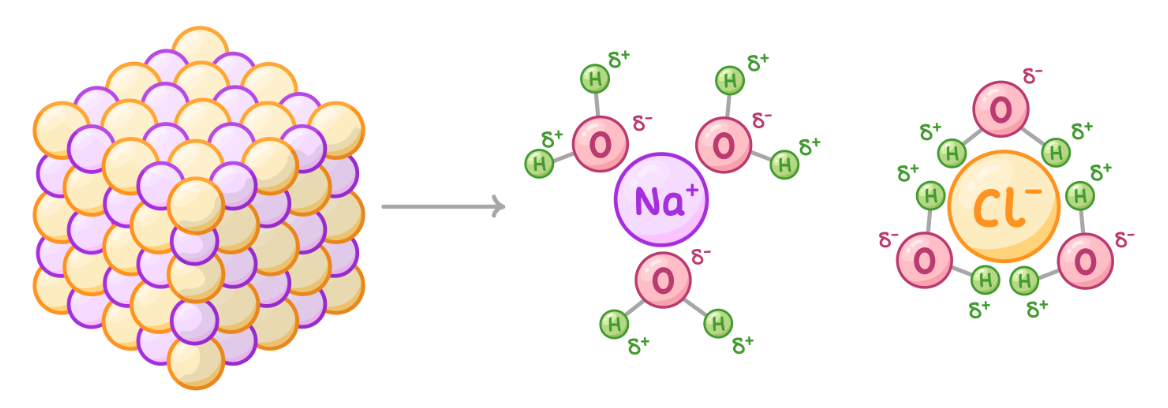
Water as a solvent
other polar molecules can dissolve in water
the positive and negative ends of water molecules attract ions and allow them to dissolve
Water as a temperature buffer
high specific heat capacity
hydrogen bonds can absorb a lot of energy before breaking so water takes a lot of energy to heat
high specific heat capacity means water is resistant to rapid changes in temperature
allows organisms to keep a steady body temp as mainly made up of water
Water as a cooling mechanism
high latent heat of vaporisation (high bp)
organisms can use evaporation of water as a cooling method without losing too much water
when water evaporates from the surface of the skin it takes heat energy away from the surface thus cooling the organism
Water as a habitat
high specific heat capacity and high latent heat gives a stable environment
ice is less dense than water as an open lattice structure is formed
floating ice forms an insulating layer on the surface of a water body
water below does not freeze allowing organisms underneath to go undisturbed
Water as a metabolite
Hydrolysis
use of water to break down complex molecules
Condensation reactions
release water to join molecules
photosynthesis
uses water as a raw material
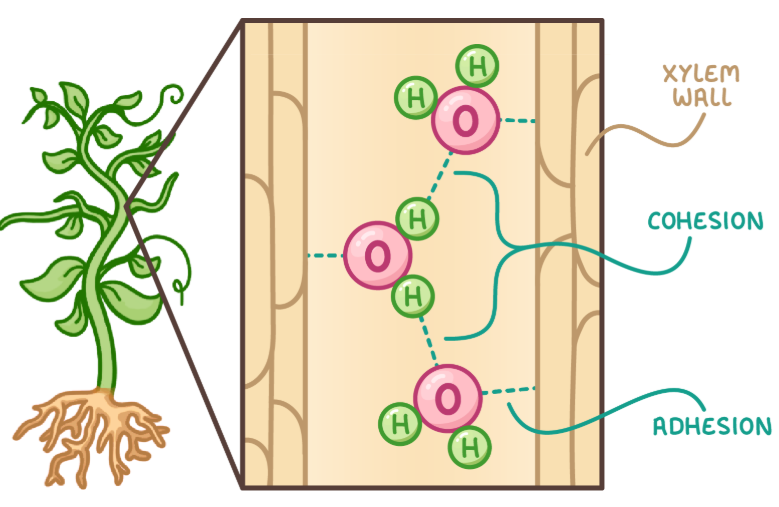
Water as a transport medium
The tendency of molecules to stick together via hydrogen bonds is cohesion
also has a tendency to stick to other molecules known as adhesion
strong cohesion and adhesion helps water flow through organisms and carry substances with it
when water molecules meet air they create high surface tension - forms a skin like structure on the surface of the water which is strong enough to support small organisms like pond skaters
xylem example
cohesion and adhesion allow plants to transport water through the xylem in a continuous column