Ch. 4.8 Trends in Periodic Properties
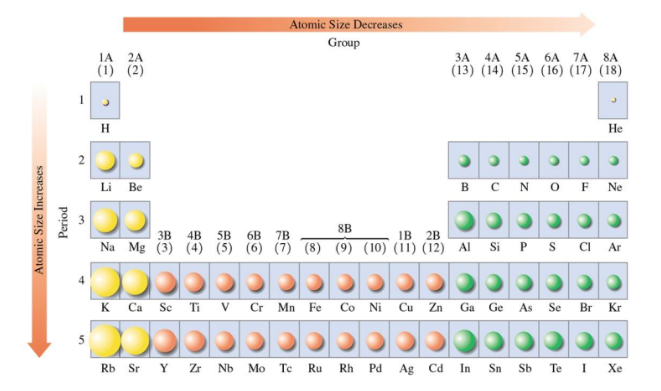
The atomic size of representative elements is affected by the attractive forces between the protons in the nucleus and the electrons in the outermost energy level
Group Numbers: Valence Electrons
For representative elements in Groups 1A (1)–8A (18), chemical properties are mostly due to the valence electrons.
Valence electrons are the electrons in the outermost energy level.
The group number gives the number of valence electrons for the representative elements.
Lewis Symbols
Lewis symbols represent the valence electrons as dots placed on the sides of the element symbol.
Rules:
One to four valence electrons are arranged as single dots.
Five to eight valence electrons are arranged with at least one pair of electrons around the symbol.
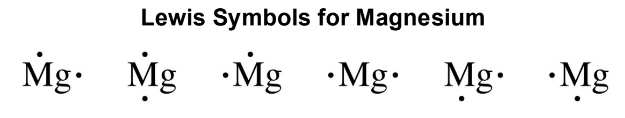
Lewis Symbols (2 of 2)
Lewis symbols for selected elements in periods 1 to 4 are shown in Table 4.11.
Helium (He) is stable with two valence electrons (often drawn with two dots).
Atomic Size
Atomic size is determined by the atom’s atomic radius, the distance between the nucleus and the outermost electrons.
Increases for representative elements from top to bottom of the periodic table.
Decreases within a period as the number of protons in the nucleus increases, pulling electrons closer.
For representative elements: the atomic size increases going down a group but decreases going from left to right across a period.
Ionization Energy
Ionization energy is the energy required to remove one of the outermost electrons.
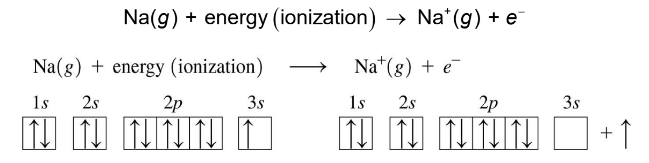
As the distance from the nucleus to the valence electrons increases, ionization energy decreases
Ionization energy is low for metals and high for nonmetals.
Ionization energy decreases down a group and increases going across a period from left to right.
Metallic Character (1 of 2)
An element with metallic character is one that loses valence electrons easily.
Metallic character:
is more prevalent in metals on the left side of the periodic table.
is less for nonmetals on the right side that do not easily lose electrons.
Metallic character of the representative elements increase going down a group and decreases going from left to right across a period
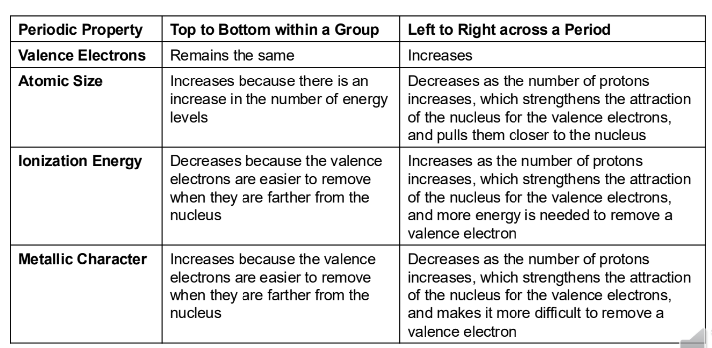
Summary:
Valence electrons: remains the same down a group; increases left to right across a period.
Atomic Size: increases down a group; decreases left to right across a period.
Ionization Energy: decreases down a group; increases left to right across a period.
Metallic Character: increases down a group; decreases left to right across a period (with noted inconsistencies in some slides).
Mathematical and symbolic notes
Valence electron configurations for representative elements (examples):
ext{Ca: } [\mathrm{Ar}]\ 4s^{2}
ext{Pb: } [\mathrm{Xe}]\ 4f^{14}\ 5d^{10}\ 6s^{2}\ 6p^{2}
Helium is stable with two valence electrons: ext{He: } 2
Electron-dot symbols illustrate valence electron counts (Cl: 7, C: 4, N: 5) as described above.