unit 1 and 2
Section 1 | The Scientific Method
The scientific method is a logical approach to solving problems by observing and collecting data, formulating hypotheses, testing hypotheses, and formulating theories that are supported by data.
\ Observing and collecting data
- Observing is useing the five senses in order ot get information
- data may be * qualitive (ex. the color of a cake) * quantative (ex. counting the amount of birds in a flock)
- A system is an amount of matter in a space that is chosen to be studied
*

Formulating Hypotheses
- Scientists make generalizations based on data
- They use this to make a hypothesis or a testable statement * ex. If I add phosphorus fertilizer then the plant will grow faster over the course of 25 days
Testing Hypotheses
- Testing a hypothesis requires an experiment in order to support or refute a hypothesis
- Controls are the experimental conditions that remain constant
- Variables are any experimental conditions that change
Theorizing
- A model is more than a physical object in science and can be an explanation of how phenomena occur and how data or events are related * visual verbal or mathmatical * ex. atomic model of matter
- A theory is a broad generlization that explains a body of facts of phenomena * ex. atomic theory
\ Section 2 | Units of Measurement
- Measurement represent qualities
- A quantitiy is something that had magnitude, size, or amount
- Measurement does not equal quantity * a teaspoon is a unit of measurement * volume is a quantity
- The choice of unit depends on the quantity being measured
SI Measurement
- Scientists have created a system called SI
- SI has 7 units * most other units are deried from the 7
- Mass * Mass is the quantity of matter * The SI unit for mass is kilogram * Weight is the measure of gravitational pull on matter * Mass does not rely on gravity
- Length * Length is a measure of distance * The SI standard for length is the meter * Kilometers or km is used to express longer distances * Centimeters or cm is used to express shorter distances
\ \ Derived SI units
- Volume * The amount of space occupied by an object * The derived SI unit is cubic meters * The cubic centimeter is used * Liters is a non-SI unit * 1 L = 100 cm^3 * 1 mL = 1 cm^3
- Density * density is the ratio of mass to colume or mass divided by colume * density = mass/volume * the derives SI unit is kilograms bper cubic meter * Density is a characteristic physical property of a substance * \
SI units are added to units to express larger and smaller unit values
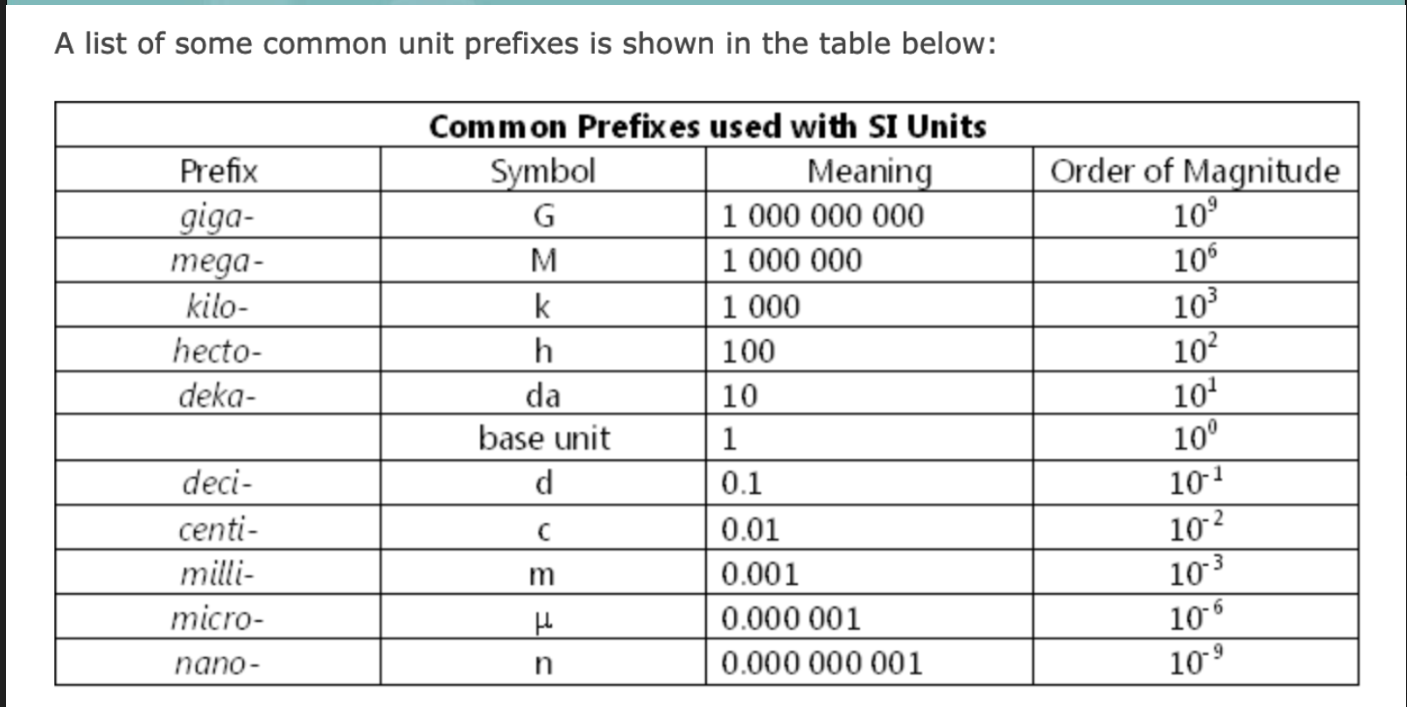
\