NMR - CARBON:
The position of the peaks in the NMR shows the different groups bonded to the carbon.
The number of peaks shows the number of environments present.
Protons and neutrons also have spin up and spin down.
When carbon 13, placed between magnets the gap between the spin up and spin down is shown.
And, if you remove electrons from the carbon 13, the gap increases, and this shows that electrons provide a shielding effect, around the magnetic field. The less electron density, the greater the larger the gap.
Same environment of the carbon = the same electron densities = the same gap of the energy
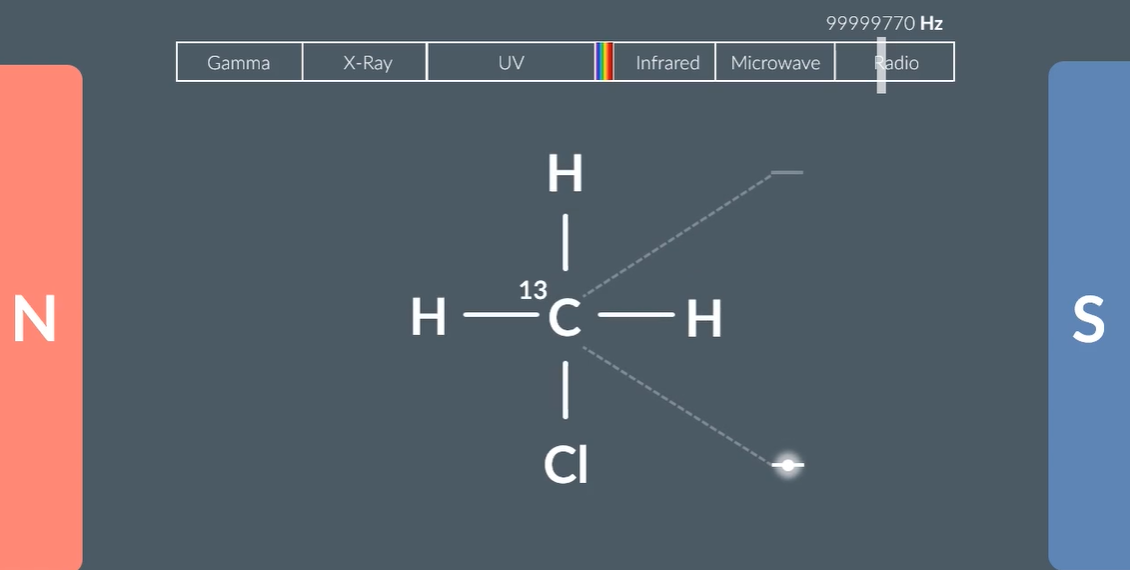
To change from a low energy state to a higher energy state, have to shine wavelength that is equivalent to the gap.
Finding the isotope C13, is very rare in environment, so a sample of it is placed in the NMR
Stronger magnets: higher frequencies interact.

The standardised model.