aromatic compounds
Importance of Aromatic Chemistry
Biological Activity of Aromatic Molecules
Examples:
Caffeine
MDMA
Chloroxylenol
Nicotine
Lidocaine
Ibuprofen
Why Is It Called "Aromatic"?
Term Origin: Introduced in 1855 by Hoffmann
Initial Association:
Early aromatic compounds associated with pleasant smells (e.g., thyme, vanilla, almonds, cinnamon)
Modern Definition:
Refers to benzene derivatives or compounds with one or more benzene-like rings
Not always pleasant fragrances
Benzene Structure and History
Discovered by Michael Faraday (1825)
Structure proposed by August Kekulé (1865)
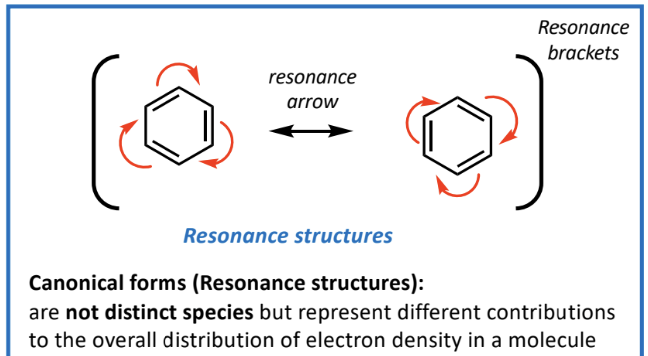
Representation of electron density with resonance structures
Each structure is a canonical form contributing to overall electron delocalization
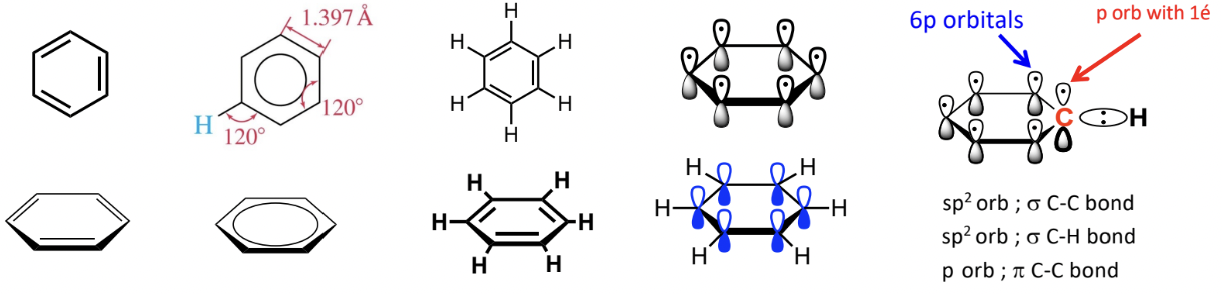
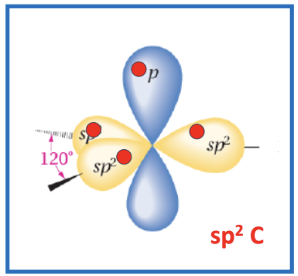
Hybridization and Aromaticity
In benzene, (c6h6) each carbon is are sp2 hybridized
Each sp2 carbon has one perpendicular p orbital
Bonding:
4 bonding electrons per carbon:
1 in each sp2 orbital forms sigma (s) bonds with neighbouring carbons with hydrogen
1 in p orbital forms a pi (p) bond
Delocalization:
6 p orbital electrons can hybridise to form a set of contiguous π bonds
these 6 π bond electrons are delocalised above and below the ring
this π system means that all six sides of the aromatic ring are identical due to this delocalization
Interactions of Benzene with Functional Groups
Benzene has delocalized p electrons which interact with functional groups
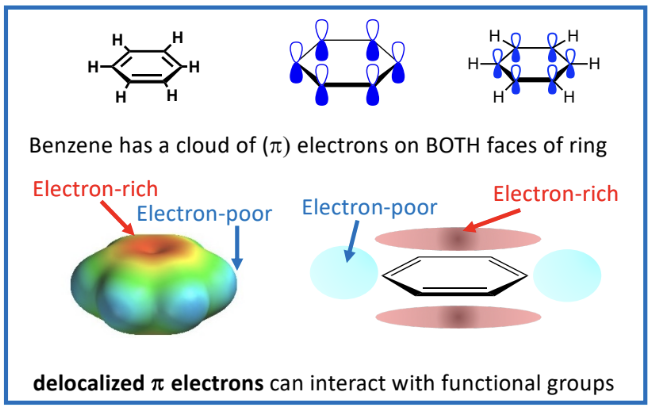
Electron Density Variations:
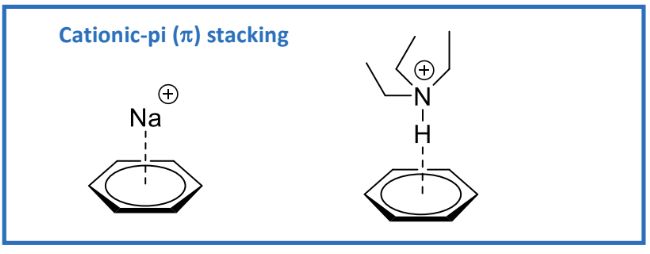
Areas in benzene can be electron-rich or electron-poor, leading to cationic-pi interactions
Aromatic Stacking:
Important in biological systems, such as stabilizing DNA
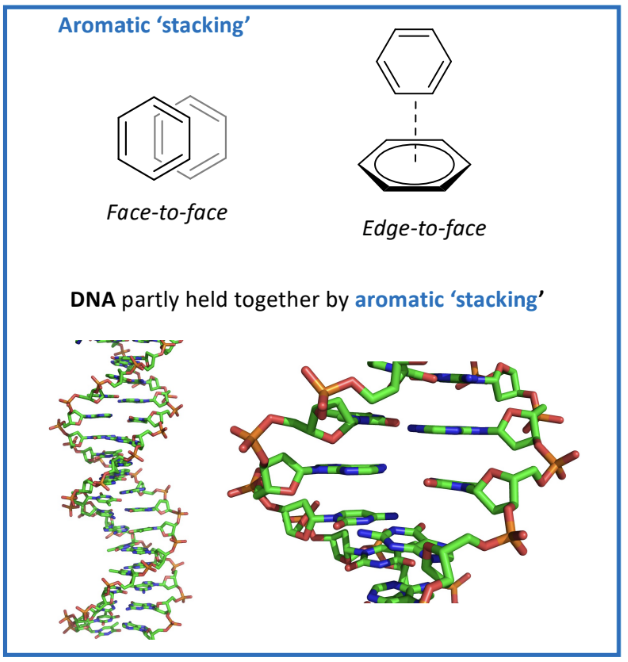
face to face means they’re slightly offset, positive and negative parts.
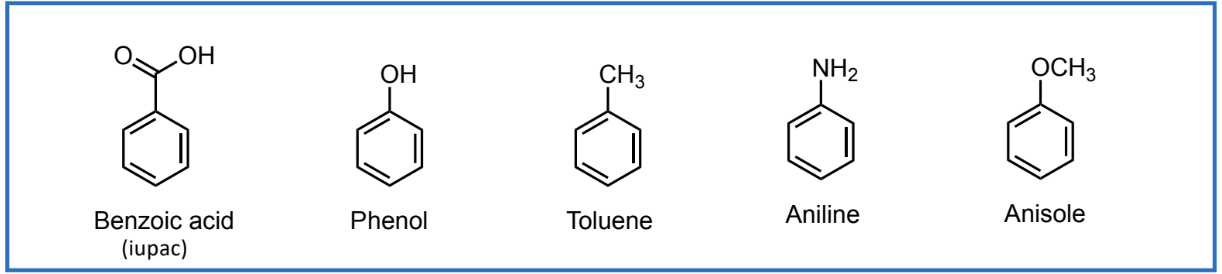
Aromatic Nomenclature
lots of non-systematic names in common use
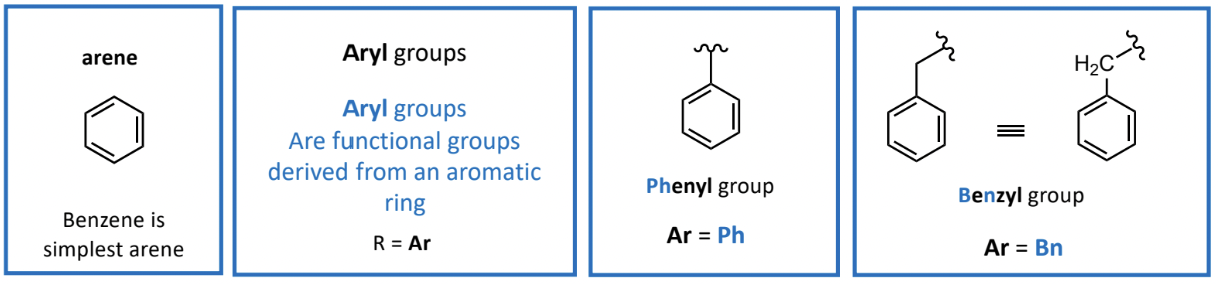
Functional Groups Derived from Aromatic Rings (Aryl group)
Aryl group denotations: R = Ar
Benzyl group and phenyl group defined as derivatives of benzene
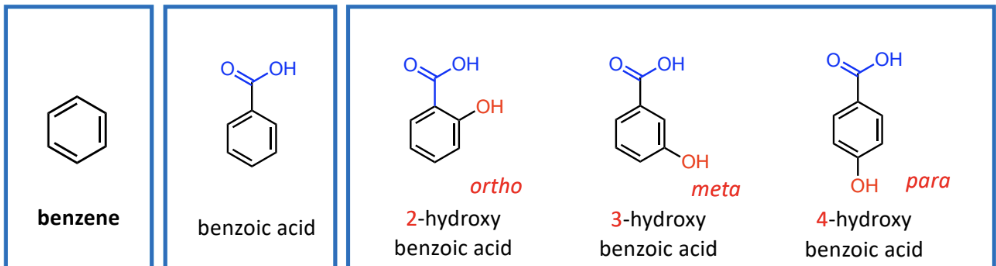
Substitution Patterns in Aromatic Nomenclature
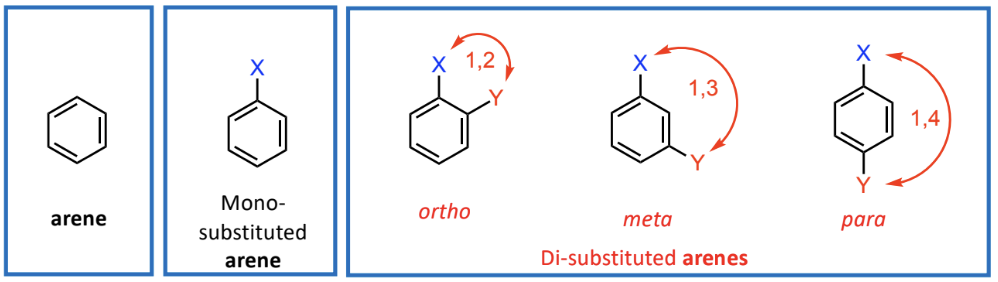
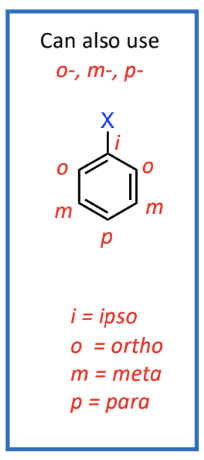
1,2 relationship (ortho), 1,3 relationship (meta), 1,4 relationship (para)
Stability of Benzene
Reactivity Studies:
No reaction occurs when Br2 is added to benzene, demonstrating its stability compared to unsaturated compounds
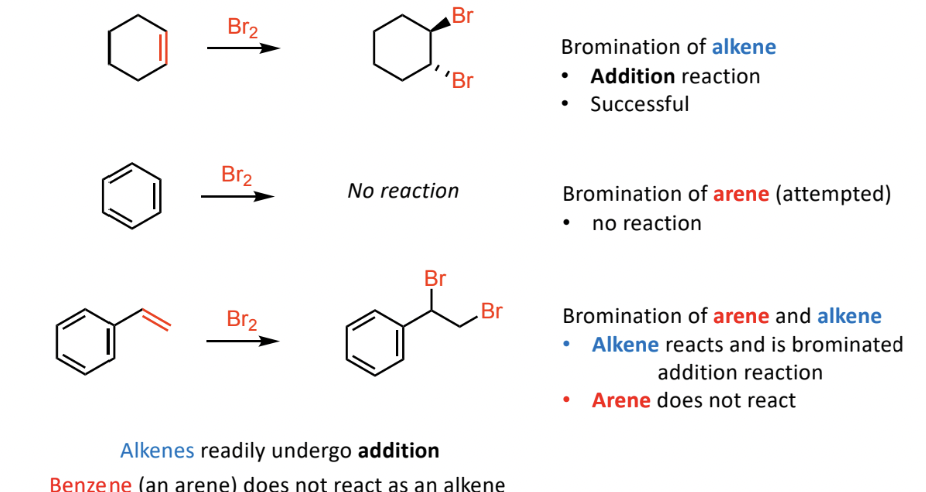
Alkenes readily undergo addition reactions while benzene (arenes) remains unchanged
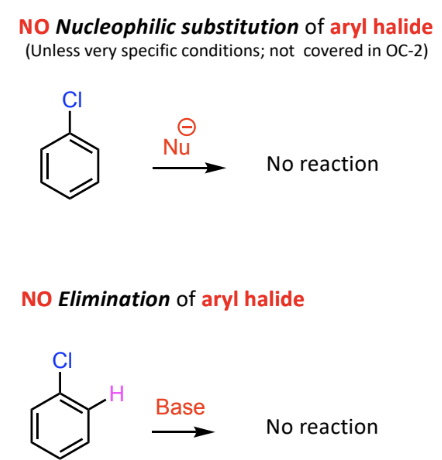
Reactivity Comparison: Alkyl Halides vs Aryl Halides
Alkyl halides undergo nucleophilic substitution easily
aryl halides do not
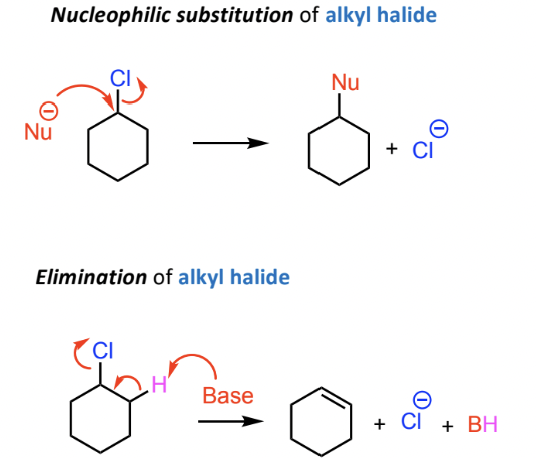
Aryl halides showcase different behavior:
No typical nucleophilic substitution or elimination under normal conditions
Specific conditions required for reactivity, no elimination of aryl halide.