Compound of an Atom
People to Note
- Eugen Goldstein: Discovered Protons, Positively charged subatomic particles
- JJ Thompson- discovered electrons
- Experiment: Cathode Ray Tube and the Plum Pudding Model
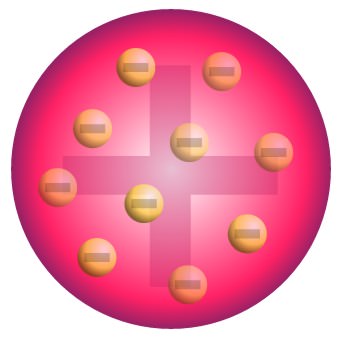
- Plum Pudding Model: Uniformly distributed throughout a positively charged material
- James Chadwick: Discovered neutron, no charge but with mass nearly equal to that of the proton
- Rutherford: Created the Nuclear theory of the atom by the gold foil experiment
Electrons: 1 unite of negative charge and its mass is 1/1840
Electrically Neutral Particle: Number of Electrons + Equal number of positive charge
Electrical Charge: a fundamental property of protons and electrons
Nuclear Theory Of The Atom
- Most of the atoms mass and all of its positive charge are contained in small cores called nucleus
- Most of the volume of the atom is empty space through which the tiny, negatively charged electrons are dispersed.
Properties of a Subatomic Particles
| Particles | Symbol | Relative Charge | Relative Mass |
|---|---|---|---|
| Electron | e- | 1- | 1/1840 |
| Proton | p+ | 1+ | 1 |
| Neutron | n^o | 0` | 1 |
quarks- Smaller subnuclear particles