Exam 3 notes FSHN 414 proteins
Overview of Proteins
Proteins are essential biological macromolecules that play a fundamental role in nearly every biological process within living organisms. They are comprised of long chains of amino acids, which are organic compounds made up of carbon, hydrogen, oxygen, and nitrogen. The specific sequence of amino acids in a protein determines its unique structure and function, making proteins crucial for numerous bodily processes, structures, and regulatory functions.
Definition of Proteins
Proteins are high molecular weight biopolymers formed by linked L-alpha amino acids through peptide bonds. Peptide bonds are covalent chemical bonds that link amino acids in a protein chain, and the specific arrangement of these amino acids is critical for the protein's structure and function. The sequence of amino acids dictates how a protein folds and interacts with other molecules.
Classification of Proteins
Simple Proteins
Albumins: These proteins are soluble in pure water and dilute salt solutions; common examples include egg white proteins and serum albumin, which helps maintain osmotic pressure in the blood.
Globulins: They are soluble in dilute salt solutions but not in pure water; serum globulin is a key example, playing an important role in immune responses.
Glutelins: Insoluble in both pure water and dilute salt solutions; they can be solubilized in acids or alkalis; wheat glutenin is a prominent example known for its elasticity in baking.
Prolamines: These proteins are soluble in moderate concentrations of ethanol; useful examples include wheat gliadin and maize zein, which contribute to the functional properties in food.
Histones: These low molecular weight proteins are rich in basic amino acids, highly soluble in water, and play an essential role in DNA packaging in cell nuclei.
Protamines: Among the smallest known proteins, they are rich in arginine, typically lack sulfur, and are soluble and highly basic, often found in sperm cells.
Conjugated Proteins
Conjugated proteins are complex structures consisting of amino acids plus other non-protein components. Notable examples include:
Nucleoproteins: These molecules consist of nucleic acids and proteins and are crucial in cell nuclei of both plants and animals for DNA packaging and regulation.
Lipoproteins: Found throughout the body, these proteins combine lipids with proteins, such as lecithovitellin in egg yolks, playing a pivotal role in lipid transport.
Glycoproteins: These proteins that combine carbohydrates with proteins are found in cells and tissues such as saliva and tendons, involved in cell-cell recognition and signaling.
Phosphoproteins: Found in milk and egg yolk, these proteins contain phosphoric acid residues essential for various biological functions, contributing to balanced nutrition for young organisms.
Importance of Proteins
Proteins are vital for various life processes and serve critical roles including:
Enzymes: They act as biological catalysts speeding up biochemical reactions essential for metabolism.
Carrier Proteins: Proteins like hemoglobin and myoglobin transport oxygen throughout the body, essential for cellular respiration.
Storage Proteins: Such as ferritin and casein, which store essential nutrients and minerals for the organism’s needs.
Contractile Proteins: Proteins like actin and myosin are involved in muscle contraction and movement.
Hormones: Many hormones, including insulin, are proteins that regulate vital physiological processes.
Structural Proteins: Proteins like collagen and elastin provide strength and support to tissues, contributing to the structural integrity of organisms.
Functional Roles in the Food Industry
In the food sector, proteins contribute to various functionalities:
Water Binding: Proteins enhance moisture retention in processed foods, improving texture and shelf life.
Viscosity: They affect the texture and consistency of food products, contributing to mouthfeel and sensory experience.
Emulsifying Agents: Proteins stabilize mixtures of oil and water, crucial in products like mayonnaise and salad dressings.
Nutritional Value: Proteins provide essential amino acids necessary for growth, maintenance, and overall nutrient balance in diets.
cohesiveness and elasticity
color
flavor
foams
provide body
buffers
enzymes
Free Amino Acids and Peptides
Significance
Free amino acids are easily absorbed by the body and can enhance flavor—monosodium glutamate (MSG) is a well-known example. Protein hydrolyzates—broken down proteins—contribute to flavor development in foods through enzymatic actions, improving palatability.
free amino acids are easily absorbed
MSG - monosodium glutamate - flavor enhancing
adds flavor w/o adding flavor itself
“chinese restaurant syndrome”
protein hydrolyzates - flavors
carbonyl-amine browning, flavors
Loss of Quality
Proteins can undergo changes leading to various forms of degradation:
Non-enzymatic Browning: Can affect color and flavor, often undesirable in food processing.
Enzymatic Oxidation: This can particularly affect phenolic substrates, such as tyrosine, leading to deteriorated quality.
Microbial Deterioration: Microorganisms can degrade proteins and amino acids, leading to spoilage and loss of nutritional value.
degradation of free glutamine - heat processing
Dietary Proteins
Nutritional Roles
Proteins supply essential nitrogen and amino acids critical for growth and maintenance.
All essential amino acids must be available simultaneously for effective protein synthesis.
Essential amino acids, which include histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine, cannot be synthesized by the body and must be obtained through dietary sources.
Protein requirements vary based on age, gender, physical activity, and health status, making personalized nutrition crucial.
Quality of Food Proteins
Protein quality plays a significant role in nutrition:
Animal Proteins: Generally considered high-quality with a complete amino acid profile, providing all essential amino acids in adequate amounts.
Plant Proteins: May lack certain essential amino acids such as lysine, tryptophan, and methionine. Combining different plant-based proteins can create a complete amino acid profile.
Cereals: Serve as a primary source of plant-based protein globally, often requiring complementary protein sources (e.g., legumes) to meet nutritional needs effectively.
Amino Acids and their Classification
Proteins are composed of approximately 20 different amino acids that can be classified based on their properties:
Hydrophobic Amino Acids: Nonpolar, hydrophobic amino acids include alanine, isoleucine, and valine, contributing to core structures of proteins by forming hydrophobic pockets.
aliphatic
Ala
Ile
Met
Pro
Val
aromatic
Phe
Tyr
Trp
Hydrophilic Amino Acids: Polar amino acids can be further divided into charged (like arginine and lysine) or uncharged (like serine and threonine), playing key roles in protein-protein and protein-ligand interactions.
charged
Arg
Asp
Glu
His
Lys
Acid/Base Properties of Amino Acids
Amino acids can exist in charged forms depending on the pH of their environment, which influences their behavior in biological systems:
Each amino acid has a specific isoelectric point (pI), where it carries no net charge, which affects its solubility and migration behaviors in electric fields during techniques like electrophoresis.
At a neutral pH, amino acids exist predominantly as zwitterions (dipolar ions) where the amino group is positively charged and the carboxyl group is negatively charged.
a-amino and a-carboxyl groups are charged
dipolar ion
pH of electric neutrality - pI - isoelectric point
add acid
COO- becomes COOH
pH at which [COO-] = [COOH] == pKa1
add base
NH3+ becomes deprotonated, NH2
pH at which [NH3+] = [NH2] == pKa2
amino acids with ionizable side chains
pKa3
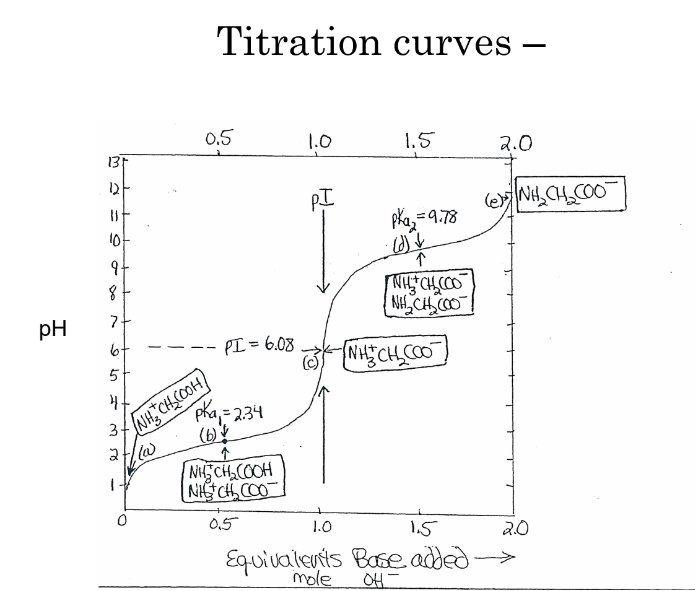
glycine
has 2 ionizable groups
a-carbonyl
a-amino group
Protein Structure
Amide (peptide) linkage
between a-amino group of one amino acid and a-carboxyl group of another amino acid
H-bonds
disulfide bonding
hydrophobic interactions between nonpolar residues
Proteins exhibit four distinct levels of structural organization:
Primary Structure: The specific sequence of amino acids connected by peptide bonds, forming the backbone of the protein.
Secondary Structure: Localized folding into structures like alpha-helices and beta-pleated sheets, stabilized by hydrogen bonds between backbone atoms.
Tertiary Structure: The overall three-dimensional folding resulting from interactions among side chains, including hydrogen bonds, disulfide bridges, ionic interactions, and hydrophobic packing.
Quaternary Structure: The arrangement of two or more polypeptide chains or subunits into a single functional protein, as seen in hemoglobin and antibodies.
three dimensional structure of proteins
fibrous
strength - rigid
insoluble in water
globular
hormones
enzymes
a protein, such as any food protein, may have within itself both regular or disordered structures as well as random regions (structural domains)
myoglobin
primary meat pigment
stores oxygen in muscle
globin
heme
a-helix
silk
fibrous protein
produced by insects
b-pleated sheet
b-bleated sheets are more stable than a-helix
collagen
structural protein
rigid and inextensible
major constituent of tendons and connective tissues
characterized by repetitious tripeptide sequence
Gly-X-Pro
“Pro” → kinks
X → any amino acid
Gly → R => H
Acid/Base properties of Proteins
peptide linkage amino and carboxyl groups can not ionize
only a-amino group at N-terminal and a-carboxyl group at C-terminal retain charges
pKa values of ionizable side chains are similar to corresponding amino acids
Denaturation of Proteins
Denaturation refers to the alteration of a protein’s native structure, which can be triggered by various environmental changes:
Changes in pH, temperature extremes, or the presence of denaturing agents can lead to protein unfolding and loss of structure.
Denaturation may result in a complete loss of biological activity or functionality of the protein.
Effects include changes in solubility and water-binding capacity, often impacting food texture or dietary benefits.
Effects of Denaturation
Desirable Effects: Can enhance digestibility and emulsifying properties in food processing - viscosity, inability to crystallize, better foaming and emulsifying
Undesirable Effects: May lead to loss of functional properties, solubility, and altered biological activity, making it crucial to control conditions during food preparation.
reversible
Agents Causing Denaturation
Physical Agents: Such as heat (cooking), cold (freezing) endothermic hydrophobic interactions, and mechanical action (whipping), which physically disrupt protein structures.
destabilization of major noncovalent interactions
H bonding, electrostatic, and van der Waals interactions
exothermic
as temp increases, KE increases
protein susceptibility to heat depends on…
Water activity (Aw)
more Aw, increased denaturation w/ heat
protein concentration
pH
ionic strength
Chemical Agents: Include pH extremes, organic solvents, detergents like SDS (sodium dodecyl sulfate), and salts that can lead to protein unfolding and aggregation.
Conclusion
The study of proteins and their functionalities is crucial not only for understanding biological processes but also for the food industry and nutritional science. Gaining insights into the complexities of amino acids, the intricacies of protein structure, and the impacts of various environmental factors on proteins helps facilitate applications ranging from food processing to health and nutrition, leading to better dietary outcomes and innovations in food technology.
Functional Properties of Proteins
1. Definition of Functionality
Functionality refers to the physical and chemical properties of proteins that influence their performance in food systems. This encompasses various stages such as processing, storage, preparation, and consumption. The functionality of proteins is determined by how they interact with other food ingredients and systems, which can affect everything from texture to flavor and nutritional value.
2. Functional Roles of Food Proteins
2.1 Functional Properties Table 12
Solubility: The manner in which proteins interact with water and other food components significantly affects their functionality.
Whey Proteins: Known for their solubility in aqueous beverages, contributing to smooth textures and enhancing flavor profiles due to their hydrophilic nature.
Muscle Proteins and Egg Proteins: Essential for binding processes in various food products, including meats, sausages, cakes, and breads, contributing to both texture and stability.
Viscosity: A crucial determinant of the thickness and overall texture in culinary preparations like soups, gravies, and dressings.
Mechanism: Achieved through water binding capabilities and hydro-dynamic size of proteins.
Proteins involved: Includes gelatin, muscle proteins, and egg proteins which aid in creating the desired mouthfeel.
Gelation: A fundamental property for forming edible structures that can trap water, resulting in desirable textures in foods such as gelled desserts and processed meats.
Mechanism: Involves the entrapment of water through network formation among proteins.
Relevant proteins: Primarily muscle proteins and egg proteins used in various applications, including meats, gels, cakes, bakeries, and dairy products like cheese.
Cohesion: Vital for adhesion and structural integrity in baked goods and pasta, affecting how these foods maintain their shape and texture.
Bonding Types: Involves hydrophobic bonds, ionic interactions, and hydrogen bonding which contribute to the overall structural properties.
Elasticity: Influences the texture and mouthfeel of food products, making them pleasant to consume.
Mechanism: Resulting from hydrophobic bonds and disulfide cross-links which provide structural support during food processing.
Emulsification: Crucial for forming stable emulsions, where proteins help blend immiscible liquids, ensuring a smooth and homogenous product.
Mechanism: Involves the adsorption of proteins to interfaces and the subsequent film formation that stabilizes the emulsion.
Food examples: An essential feature in items like sausages, bologna, cakes, soups, and salad dressings.
Foaming: Critical for creating stable air bubbles in products such as whipped toppings and ice cream, which is essential for fine texture and lightness.
Mechanism: Involves interfacial adsorption where proteins stabilize air pockets within the food matrix.
Proteins involved: Specifically egg proteins and milk proteins are key in this property.
3. Properties Influencing Functionality
Key properties that influence the functionality of proteins include:
Size: Protein molecular size can affect solubility, emulsification, and texture.
Shape: The three-dimensional structure contributes to how proteins interact with each other and with other molecules in food systems.
Amino Acids Composition: The specific arrangement of amino acids influences the chemical behavior and nutritional quality of proteins.
Sequence and Net Charge: The order of amino acids and their resultant charge can affect how proteins behave in different environments.
Hydrophobicity/Hydrophilicity Ratio: This balance plays a crucial role in solubility and interactions with fats and water.
Protein Structure: Includes secondary, tertiary, and quaternary structures, which all contribute fundamentally to protein function in food systems.
Molecular Flexibility: The flexibility of protein structures can influence their ability to interact with other components, thereby impacting mouthfeel and sensory perceptions.
4. Protein Hydration
Importance: Water-protein interactions are critical in determining the rheological (flow and deformation) and textural properties of food products.
Functional properties reliant on water/protein interactions include:
Dispersibility: How well proteins can be spread out in a solution or food matrix.
Wettability: The ability of proteins to absorb water, which is vital for various culinary applications.
Solubility: The capacity of proteins to dissolve in water, affecting everything from taste to nutritional profile.
Swelling, Gelation, Coagulation: All these processes critically depend on the hydration of proteins and the interactions that ensue.
emulsification
foaming
5. Water Binding Capacity
binds to…
charged groups
peptide groups (backbone_
amine groups (Asn, Gln)
OH groups ( e.g., Ser)
non polar residues - hydrophobic hydration
Measurement: Expressed as grams of water bound per gram of protein, which indicates protein’s effectiveness in retaining moisture.
Charged amino acids can bind approximately 6 mol of water per mol.
Uncharged amino acids bind about 2 mol of water per mol.
Nonpolar amino acids bind roughly 1 mol of water per mol.
6. Water Holding Capacity
Definition: Refers to the protein's ability to absorb and retain water against gravity within the protein matrix, influencing texture and juiciness in food products.
Relevance: More significant than mere water binding, as it directly affects moisture retention and overall quality in food products.
7. Solubility Factors
Influencing Factors:
protein-protein + solvent-solvent = protein-solvent
Thickening, Foaming, Emulsification, Gelation: These processes heavily rely on the solubility of proteins.
The balance between protein-protein interactions and protein-solvent interactions is crucial in this respect.
Hydrophobic interactions generally decrease solubility, while ionic interactions can enhance it, highlighting the complex nature of protein behavior in food systems.
8. Classification Based on Solubility
Types:
Albumins: proteins that are soluble in water; found in egg whites and blood plasma.
Globulins: soluble in dilute salt solutions; commonly found in legumes and meat.
Glutelins: soluble in acidic and alkaline solutions; found in cereals like wheat.
Prolamines: soluble in 70% alcohol; examples include gliadin in wheat.
9. pH Influence on Solubility
Net Charge: The solubility of proteins changes above and below their isoelectric point (pI).
At the pI, proteins exhibit minimal solubility due to electrical neutrality, often resulting in precipitation.
Electrostatic repulsion occurs when the net charge is positive or negative, enhancing solubility significantly.
10. Ionic Strength Impact on Solubility
Low Concentration: Low ionic strength neutralizes charges on proteins, which can enhance their solubility.
High Concentration: Excess ions present in high concentrations compete with proteins for water, potentially reducing solubility.
11. Temperature Influence on Solubility
Range: Typically, solubility increases from 0 to 40°C due to increased molecular motion, but may lead to protein aggregation above 40°C as proteins unfold and hydrophobic residues become exposed.
12. Organic Solvents
Effect: The presence of organic solvents can cause proteins to unfold due to the repulsion of hydrophilic regions, which affects solubility and functional behavior.
13. Gelation
Definition: The process of forming a solid-like network of proteins that effectively holds water within the structure, resulting in desired textures in food.
Mechanism: Occurs under conditions of protein denaturation through heat, pH changes, or pressure which influences water retention and textural properties.
14. Types of Gels
Thermally Irreversible Gels: Formed by heat; these gels remain stable upon cooling and can include products like egg gels which do not revert.
most common
“thermoset”
Thermally Reversible Gels: Formulated by cooling and can melt upon reheating, a common example being gelatin-based products.
15. Characteristics of Thermally Irreversible Gels
Development Mechanism: Involves multiple interactions, including hydrophobic interactions, hydrogen bonding, ionic interactions, and sometimes disulfide bridges that contribute to network stability.
Influence Factors: Determined by the type of protein used, concentration of solid ingredients, and the environmental conditions during processing.
heat process
protein unfolds, exposing functional groups
aggregation and formation of gel network
non-covalent bonding interactions between functional groups
as protein form gel — viscosity increases
cooling — subtle changes
16. Conditions Influencing Gel Properties
Optimal Gel Strength: It typically exhibits an opaque appearance with significant water binding capacity.
pH Extremes: Can cause repulsion among molecules, leading to more transparent gels which may not be desirable in all applications.
High Salt Concentration: Foster excess attraction forces, leading to the formation of cloudy, turbid gels that can affect the aesthetic quality of food products.
SCALE
sol (low-no salt)
transparent gel
opaque gel
turbid gel (high salt)
Between transparent gel and opaque gel is highest gel strength
17. Thermal Reversible Gels
Example: Gelatin, which is derived from collagen, forms reversible gels when heated in moist conditions, becoming liquid and then solidifying upon cooling.
PROCESS
native collagen is heated under moist conditions for long time — random coil
gelatin is made
cooling causes gelatin to refold making gel
heating causes refolded gelatin to go back to unfolded form
NEVER GOES BACK TO COLLAGEN
18. Emulsification
Definition: The process by which two immiscible liquids, such as oil and water, are mixed together, resulting in one liquid being dispersed in the other.
19. Types of Emulsions
Oil in Water (o/w): This type has oil as the dispersed phase, with water as the continuous phase; examples include salad dressings.
Water in Oil (w/o): In this case, water is the dispersed phase and oil is the continuous phase; commonly seen in butter and margarine.
20. Natural vs Intentional Emulsions
Natural Emulsions: Such as milk, where proteins (casein and whey) help stabilize the mixture naturally.
Intentional Emulsions: Products like ice cream and mayonnaise are formulated to be emulsified, highlighting the role of proteins in maintaining texture and stability.
21. Role of Surfactants
Function: Surfactants (emulsifiers) help lower interfacial tension between the two phases, migrating to the interfaces and stabilizing the emulsions by exhibiting both hydrophilic and hydrophobic properties.
22. Factors Affecting Emulsification
Effective Emulsification: For successful emulsification, proteins must orient and migrate to the oil-water interface, forming a stable film around droplets to prevent coalescence.
Influencing Factors: Include temperature, pH, ionic strength, and the degree of protein denaturation, all of which play a crucial role in the stability of emulsified products.
23. Foaming Process
Similar to Emulsification: The foaming process also involves two phases: an aqueous continuous phase and a dispersed air phase to create foam.
Examples: Commonly seen in whipped cream, ice cream, cakes, and meringues, where proteins trap air.
24. Factors Influencing Foaming
Key Aspects: The foaming capacity is affected by protein solubility and the degree of partial denaturation, which enhances exposure of hydrophobic regions that stabilize bubbles.
Egg Whites: Comprised of proteins such as ovalbumin, globulin, and ovomucoid that collectively contribute to the formation of stable foams. Best foams
Aging Foams: Over time, liquid may drain, leading to bubble rupture and coalescence, which compromise the integrity of foams in culinary products.
25. Inhibition of Egg White Foams
Causes: The presence of lipids can destabilize the foam structure, inhibiting the foam from achieving its desired volume and stability (EGG YOLK)
Mechanical Factors: The speed and duration of whipping have critical implications for foam stability, as improper techniques can lead to weak foam structures.
High Protein Foods
Top 10 Foods Highest in Protein
Lean Chicken Breast
Protein Content: 54.5g protein (109% DV) in a 6oz breast
Calories: 267 calories
Benefits: Lean chicken breast is an excellent source of lean protein, low in fat, and contains essential vitamins and minerals such as B vitamins and phosphorus.
Lean Pork Chops
Protein Content: 52.7g protein (105% DV) in a 6oz chop
Calories: 332 calories
Benefits: High in protein and a significant source of important minerals like zinc and selenium, also provides essential B vitamins.
Tuna
Protein Content: 50.8g protein (102% DV) in a 6oz fillet
Calories: 313 calories
Benefits: Rich in omega-3 fatty acids which support heart health, as well as high-quality protein for muscle repair.
Beef (Skirt Steak)
Protein Content: 48.7g protein (97% DV) in a 6oz steak
Calories: 456 calories
Benefits: Packed with iron and vitamin B12, important for energy metabolism and oxygen transport in the blood.
Firm Tofu
Protein Content: 43.5g protein (87% DV) per cup
Calories: 363 calories
Benefits: A versatile plant-based protein source rich in calcium and iron, making it an excellent choice for vegans and vegetarians.
Lentils
Protein Content: 17.9g protein (36% DV) per cup
Calories: 230 calories
Benefits: High in fiber, providing digestive health benefits, and low in calories; great for heart health and blood sugar management.
Low-Fat Yogurt
Protein Content: 14g protein (28% DV) per cup
Calories: 137 calories
Benefits: Contains probiotics for digestive health and is a rich source of calcium and B vitamins.
Grated Parmesan
Protein Content: 10.2g protein (20% DV) per oz
Calories: 111 calories
Benefits: Nutrient-dense cheese providing flavor and an excellent source of calcium and protein.
Squash and Pumpkin Seeds
Protein Content: 8.5g protein (17% DV) per 1oz handful
Calories: 163 calories
Benefits: High in magnesium and zinc, beneficial for immunity and muscle function while providing healthy fats.
Eggs
Protein Content: 6.3g protein (13% DV) in a large egg
Calories: 78 calories
Benefits: A complete protein source containing all essential amino acids, also rich in choline and vitamin D.
Soy Proteins
Overview of Soy Proteins
Average plant proteins contain approximately 16% protein, whereas soy has about 38% protein content.
Soy proteins have enhanced solubility in water compared to corn and wheat, which tend to be hydrophobic.
Types of Soy Proteins
Soy Flours and Grits
Contain approximately 40-60% protein.
Soy Protein Concentrates (SPC)
Retains around 75% protein content.
Soy Protein Isolates (SPI)
High purity soy protein with approximately 90% protein content.
Characteristics of Soy Proteins
Comprised of a mixture of proteins with molecular weights ranging from ~8000 to 600,000 daltons.
Their solubility allows for fractionation by centrifugation into four fractions: 2 S, 15 S, 7 S, and 11 S.
Functional Properties of Soy Proteins
Soy proteins are crucial in food processing for their gelling, viscosity, emulsification, and flavor-binding capabilities.
Gelation in tofu occurs through specific disulfide linkages and the presence of divalent cations like calcium and magnesium.
Cereal Proteins
Protein Content in Cereals
The average protein content in cereals is approximately 10%.
Notably, wheat is deficient in lysine, while corn is deficient in both lysine and tryptophan.
Classification of Wheat Proteins
Albumins: Water-soluble proteins.
Globulins: Soluble in salt solutions.
Gliadins: Soluble in 70-90% alcohol.
Glutenins: Soluble in acid or alkali.
Insoluble proteins (~80-85%) contribute to gluten formation when mixed.
Dough Formation Process
During mixing, shear forces unfold proteins, enhancing hydrophobic interactions.
Hydrogen bonding and disulfide cross-linking occur, contributing to gas retention capabilities essential for bread rising.
Milk Proteins
Composition and Types of Milk Proteins
Milk contains approximately 30-36g protein/L, predominantly categorized into two major types:
Associated with Milk Fat Globules: Including minor enzymes.
In Skim Milk: Includes casein and whey fractions, which are major components.
Major Casein Proteins Concentration
Caseins constitute about 24-28g/L, making up 80% of the total protein content in milk, featuring various subtypes.
Casein Micelles and Structuring
Caseins form spherical complexes called micelles, crucial for cheese curd formation, affecting texture and flavor.
Whey Proteins
Soluble proteins such as alpha-lactalbumin and beta-lactoglobulin play important roles in food processing and enhance nutritional value.
Meat Proteins
Classification of Meat Proteins
Contractile Proteins: Major proteins in skeletal muscle (myosin ~37%, actin ~12%).
Sarcoplasmic Proteins: Comprising ~20% of total protein, primarily enzymes and pigments.
Connective Tissue Proteins: Provide structural toughness (e.g., collagen).
Muscle Structure
The basic unit is the muscle fiber cell, which is composed of long, cylindrical groups arranged into bundles, contributing to muscle function and strength.
Roles of Connective Tissue
Connective tissue proteins significantly impact meat tenderness, affecting cooking methods, and are crucial for various culinary applications.
Post Mortem changes affecting meat
Lean meat
~70% water
retention is important
economics
water loss
unattractive
loss of soluble nutrients
altered texture
increased water content — decreased mechanical strength
most tissue water in myofibrils
freezing influences water loss
cooking — increases water loss
shrinkage of filaments
water holding capacity lowest at pI
~pH 5.5
during heating of meat proteins
shrinkage of tissue (changes in myofibrillar proteins)
hardening of muscle, releases juices
meat is a 3-D network
water trapped in protein matrix (like a gel)
changes in proteins affect WHC
as muscle fibers shrink
meat becomes tough and hard
denaturation of contractile proteins
collagen shrinks and pulls on muscle
forces out some water from matrix
extended cooking
decreases toughness
conversion of collagen to gelatin
degradation of myofibrillar protein
Collagen + heat + water —> denaturation —> gelatin (more water soluble)
Egg Proteins
Composition of Egg Proteins
The egg yolk contains various glycoproteins, while the egg white is rich in globular glycoproteins, contributing to its functional properties in cooking.
egg shell 4%
white 11.5%
at least 40 types of globular glycoproteins
ovalbumin ~70%
yolk 17.4%
glycoproteins
lipoproteins
phosphoglycoproteins
phopholipoproteins
Functional Roles of Eggs in Foods
Eggs play vital roles in coagulation, gel formation, emulsification, and providing structural integrity in many recipes, enhancing taste and texture.
egg white proteins
important as best food foaming proteins
egg yolk proteins
emulsification
Unconventional Protein Sources
Definition and Examples
Unconventional protein sources refer to novel plants or animals, as well as single-cell proteins derived from bacteria or algae.
There is an increasing focus on utilizing food processing waste for sustainable protein production, capitalizing on new technologies to combat food waste.