CHEMISTRY: Ions Ionic and Molecular Compounds
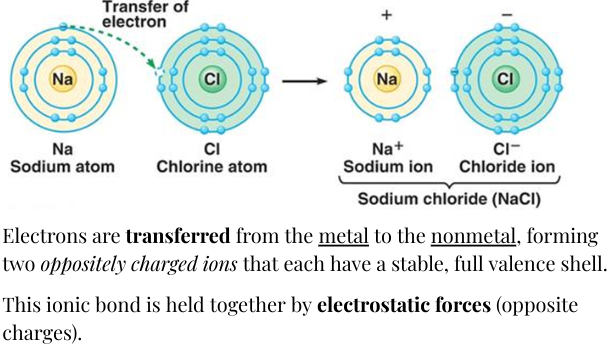
\ Ions: Transfer of Electrons (charged atoms)
- Noble gases are the most stable tehy eight valence electrons (octet, except for helium)
\
- In Groups 1A to 7A (skip transition), the atoms of elements achieve stability through loss, gain, or sharing in the formation of composites their valence electrons.
- Representative metals lose valence electrons to make positive ions (cations): Group 1, Group 2, and Group 3
- In response to metals, nonmetals gain electrons to form bytes that form negative ions (anions): Group 5, Group 3, Group 6, Group 2.

The charge of metals, is it’s group number (anions)
The charage of non-metals is it’s group number - 8
6.2 - Ionic Compounds
- Made up of one or more canion and anions bonded together (usally metals and non-metals)
\
Properties of Ionic compounds:
- Ionic compounds are always solid because they have very strong attraction and build (solid)
Therefore it takes a lot of energy to move them apart (high melting and boiling points)
- Brittle (easy to break) because pressing on it makes the like charges repeal
- Ionic compounds dissociate into their ions in solution, making them electrolytes (able to conduct electricity when dissolved in water)
\
6.4: Multivalent Ions:
Elements that can make multiple stable ions, so as Fe and Fe2 can both be iron oxide.
- Copper
- Cobalt
- Tin
- Lead
- Ect;
When you have roman numerals beside the symbol that is it’s charge and you cross it’s charges so that you can get the formula
6.4 - Polyatomic Ions
- A Polyatomic ion is a group of electrically charged covalent atoms, such as the formulation CO3 2- for the carbonate ion. The charge is shared all over the atom
- The names that end with ate or are mostly polyatomic ions.
1. A nonmetal and one or more atoms contain polyatomic ions.
- The NH4+ ammonium ion is a polyatomic positive ion
- If several polyatomic ions are used to balance the charge, parentheses include the polyatomic ion formula.
6.5 - Molecular Compounds: Sharing Electrons
- Non-metals share valence electrons in covalent bonds, which ensures that every atom has a stable electron set-up. * In a molecular compound, the first nonmetal uses its name; the second nonmetal uses the first syllable of its name, followed by the ideal element.
- The name of a two-atoms-molecular compound uses prefixes to indicate the subscriptions in the formulation.
6.6 - Lewis Structures for Molecules
- For all the atoms in the molecule, the total number of valence electrons is determined.
- In Lewis's structure, the central atom and each of the attached atoms is connected by a pair of electrons.
- All remaining valence electrons are used to complement the octets of the surrounding atoms and the central atom as solitary pairs.
- If octets are not completed, one or more lone electron pairs are placed as double or threefold connecting pairs
6.7 - Electronegativity and Bond Polarity
- Electronegativity is an atom's ability to attract electrons that it shares. * The electronegativity of metals is generally low, while nonmetals have high electronegativity.
- Atoms share electrons equally in a nonpolar covalent bond.
- The electrons are unequally divided into a polar covalent connection because they are attracted to the more electronegative atom.
- The atom is partly positive in a polar relationship (d+) and partly negative in the polar link with lower electronegativity (d+) (d-).
- Atoms forming ionic bonds differ greatly in electronegativity.

6.8 - Shapes of Molecules
- The shape of a molecule is based on the Lewis structure, the geometry of the electron group and the number of atoms connected. * The geometry of the electron group surrounding a central atom with 2 electron groupings is linear; the geometry is planar trigonal in three electron groupings, and geometry is tetrahedral in four electron groups.
- The shape is identical to the electron arrangement when all of the electron groups are attached to atoms.
- A central atom has a bending form of 120° with three groups of electrons and two bonded atoms. * There is a trigonal pyramid shape in a central atom with four different electron groups and three connected atoms.
- A central atom has a curved form at 109°, consisting of four electron groups and two bonded atoms.
6.9 - Polarity of Molecules and Intermolecular Forces
- Non-polar molecules have non-polar covalent links or a bonded atomic arrangement to waive dipole
- The dipoles do not cancel in polar molecules * Opposite loaded ions are held in ionic solids by ionic connections in a rigid structure.
- Intermolecular forces are known as dipole-dipole and hydrogen binding maintained together with the solid and liquid states of polar molecular compounds. * The weak attractions between temporary dipole known as dispersion forces form nonpolar compounds.
\