Year 1 - Inorganic
Summary and key points - CH8 (Periodicity), CH 9 (the Alkaline Earth Metals), CH10 (the Halogens)
CH8:
Different areas of the periodic table has different names.
Alkali metals
Alkaline earth metals
Transition metals
Metaliods
The metals and non-metals ladder, the metaloids that touch this line, have a combination of metallic and non-metalic properties
The periodic table is split into s,p, d and f blocks
So, all the elements in their blocks determine which is their highest sub-shell
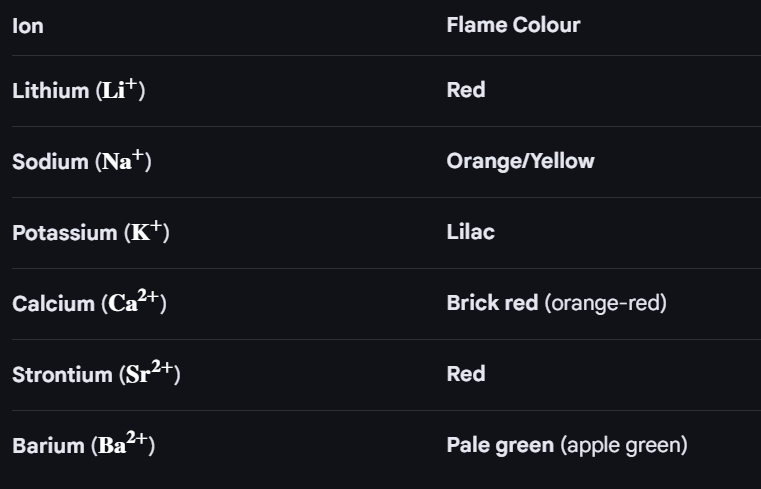
When elements are heated they give out energy, because electrons move to a lower energy level (electron shells closer to the nucleus is lower enegy level), additionally light wavelengths are released that appear on spectrum (s, p, d and f line)
Scandium and Zinc are not transition metals, beacuase they do not form compounds with partially filled d orbitals
Definition of Periodicity: A regular repeating pattern
Definition of Atomic radii: Half the distance between pair of atoms
It is difficult to measure radius of an isolated atom, because there is no clear point at which electron cloud density around it drops to zero (basically meaning there is no fixed edge of an atom)
General trends:
Across a period the atomic radius decreases → Nuclear charge increases → Shielding is roughly the same → increased attraction between the positive nucleus and electrons
The first ionisation energy increases across the period → Nuclear charge increases → Shielding is roughly the same → More energy to remove the electron on the outermost shell → increased electrostatic forces of attraction
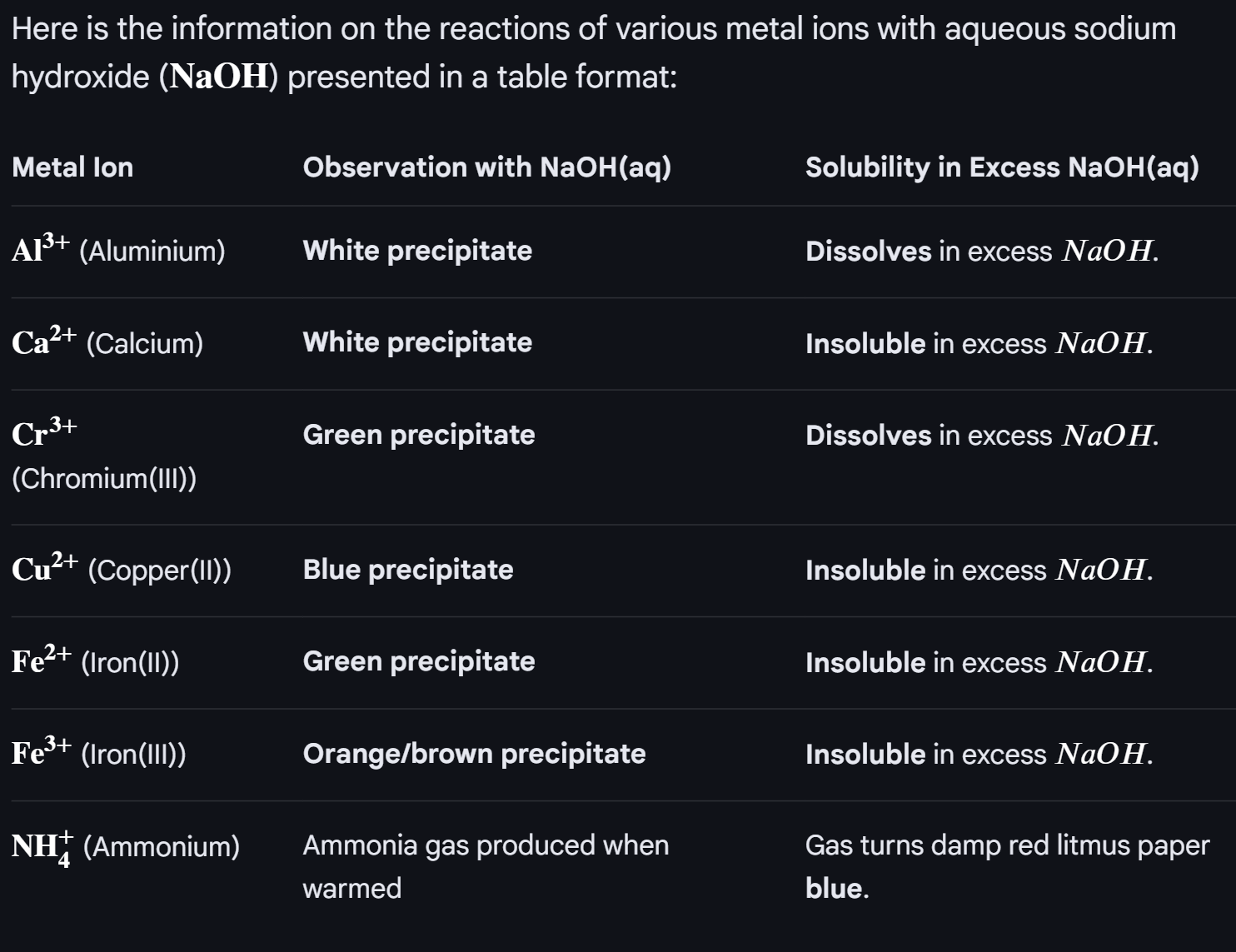
CH9:
As you go down group 2 metals, the atomic radius increases, so the forces of attraction between the delocalised electron and posiitve ions decreases, so more energy is required to overcome this force of attraction
Reaction of group 2 with liquid water:
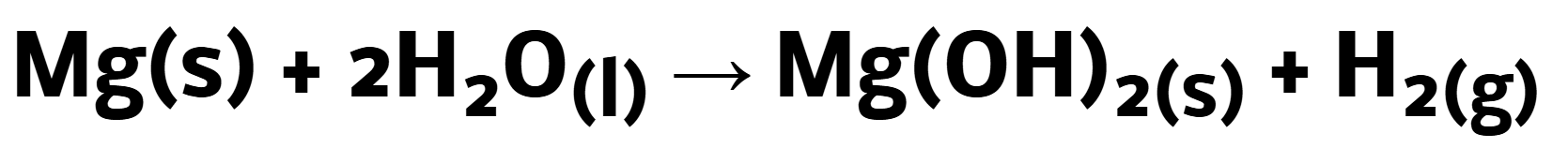
Reaction of group 2 with steam (gaseous water) => more rapid reaction
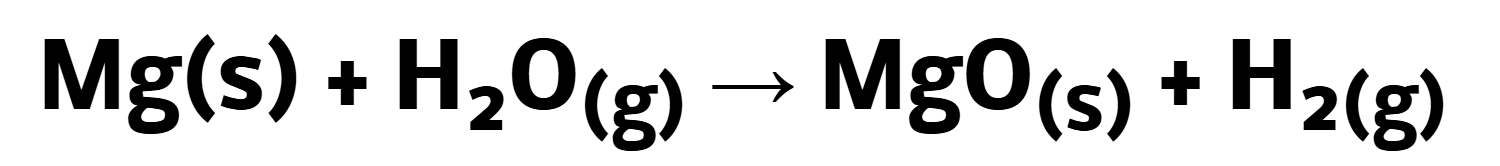
Reaction of group 2 with sulfate: (ionic equation) [insoluble x-ray imaging]
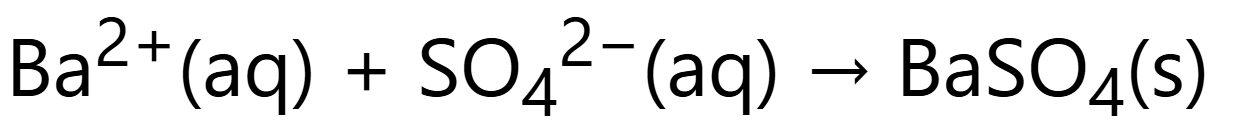
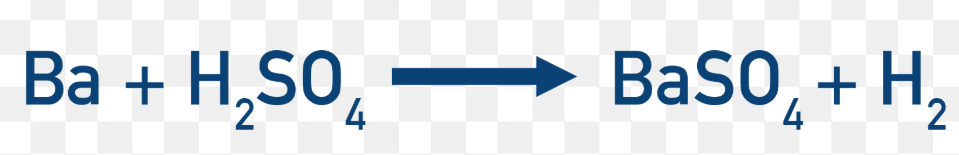
Full equation:
Properties:
As you go down the group the hydroxides become more soluble.
As you go down the group the sulfates become more insoluble.
CH10
The halogens are diatomic molecules, Fluorine has an unusual characteristic (having a weak F-F bond), due to the small size of fluorine that leads to repulsion between non-bonding electrons. [g,g,l,s] => the states at room temp F to iodine (due to increased size more electrons, greater van der waals effect)
Displacement reactions occur, where more reactive halogen displaces a less reactive halogen.
Reducing power (oxidised => loss of electrons) increases down the group
Chlorine:


No redox reaction, only acid base reaction
HCl (misty fumes)
Sodium thiosulfate
Bromine:
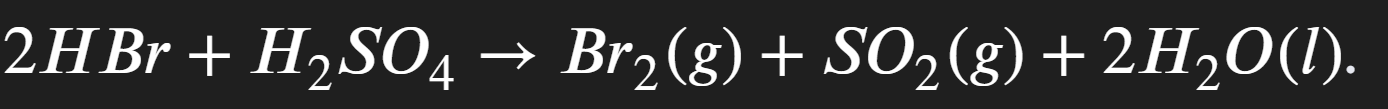

Inital misty fume
Red-brown fumes of bromine gas
Sulfur dioxide turns acidified potassium dichromate orange to green
Iodine:
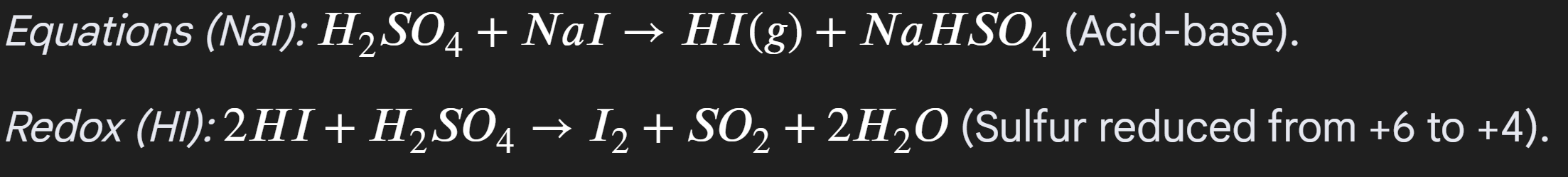
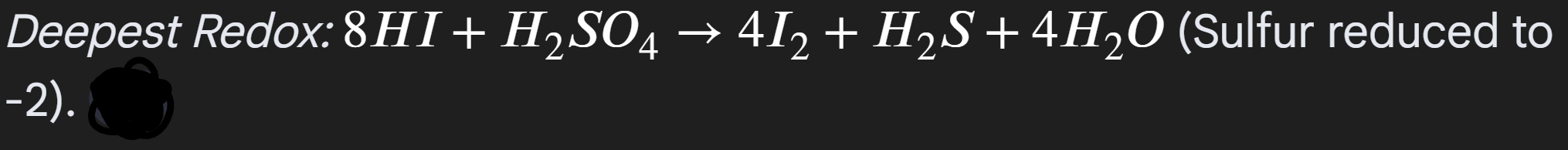
Initial misty fumes
Black solid Iodine
yellow solid sulfur
rotten egg smell H2S
The reason for many observations, because Iodine is powerful reducing agent, enabling multiple products to be oberved

Disproportionation reaction: (Chlorine and water without sun)
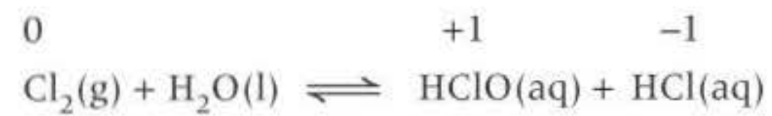
Forms two acids
Chloirc acid (HClO)
hydrochloric acid (HCl)
Chlorine and water with sun
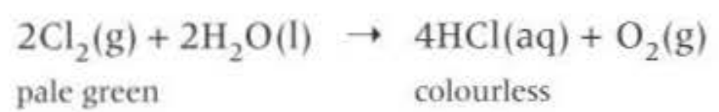
Reaction with alkali: (another disproportionation reaction)
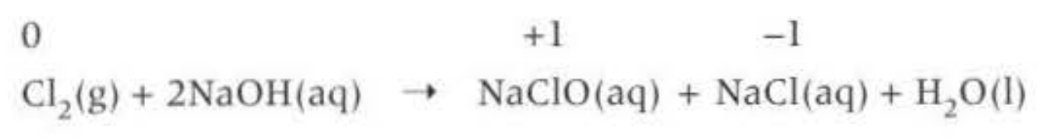
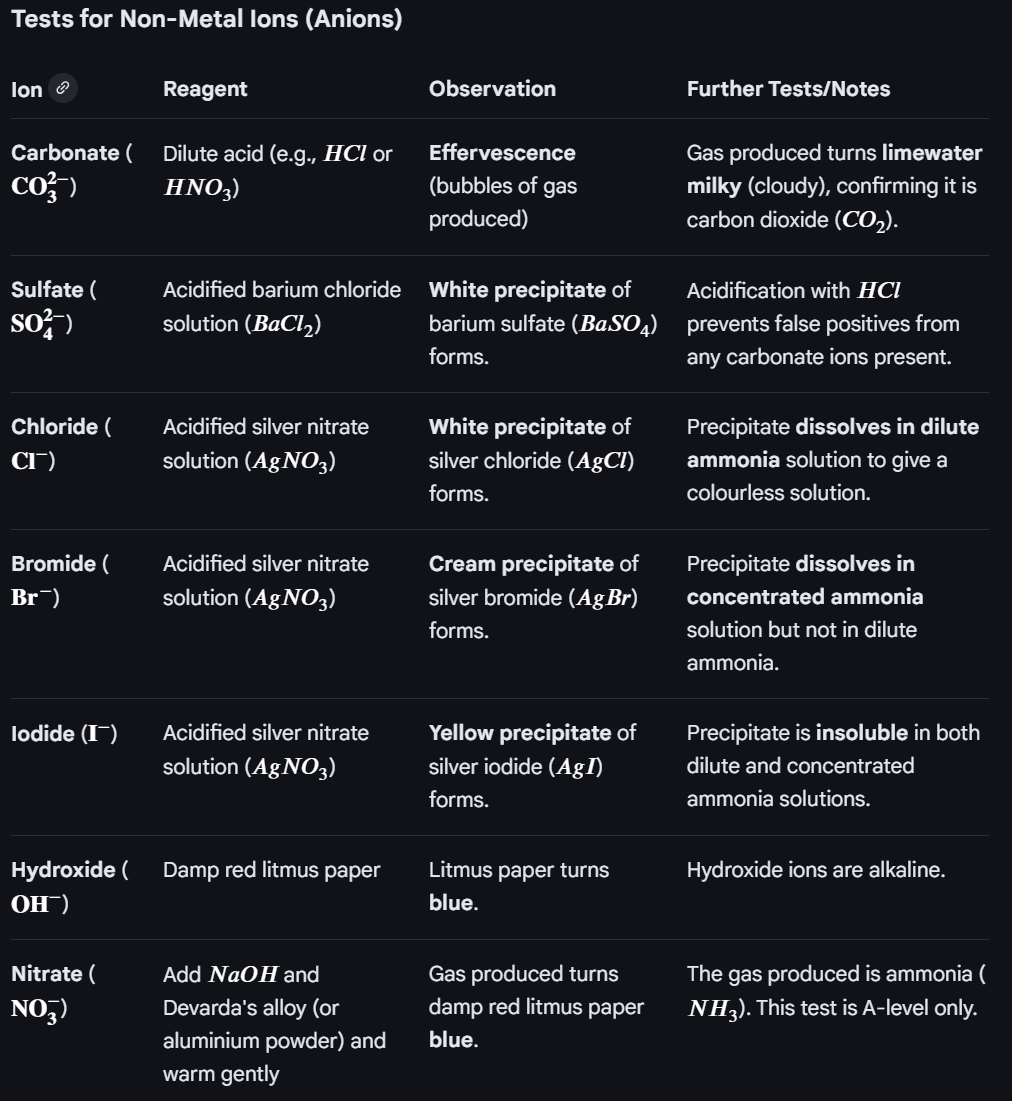
Important tests: