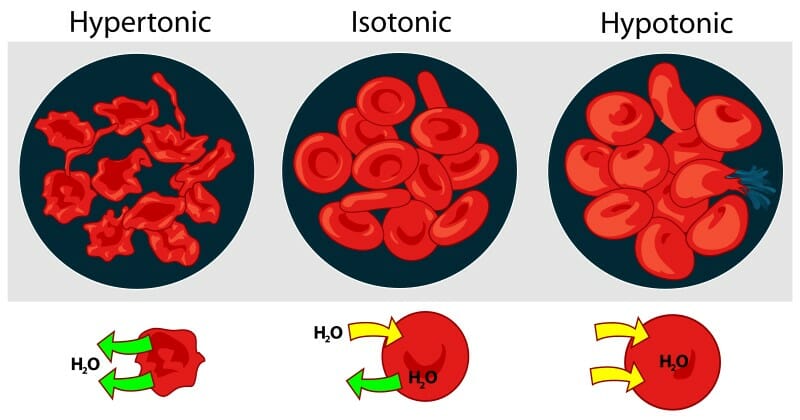
bio s1
intro
3 features of living things
- requirment for energy and nutrients
- homeostasis
- Use of DNA as herieditary material
\
nutrients- anything an organism need for growth and survival and cant make itself
Homeostasis- maintaining a stable internal enviroment
consumers- organisms that get energy from eating
producers- male own food from the enviroment
development-process by which 1st cell of an organism gives rise to an adult
growth- getting more cells/cells growing
inheritance- transmission to offspring of DNA
Organizations of Life
- Atom- non-living building blocks of all substances
- Molecule- non-living, 2+ atoms together
- Cell-smallest unit of life
- Tissue-cells organized to serve a collective function
- organ- 2+ tissues working together to serve a function
- organ system- set of organs that work together to keep a body working
- organism-an individual of 1+ cells
- population- group of individuals of the same species in an area
- community- all populations of all species in an area
- ecosystem- a community of all species in an area
- biosphere-all regions of earth where organisms live
\
biochem
Sub-Atomic Particles
| Protons | neutrons | electrons |
|---|---|---|
| + (+1) | neutral | - (-1) |
| found in nucleus | found in nucleus | surrounding nucleus |
| p+ | n^0 | e- |
| determines idenity of an atom | isotopes- #p+/=/#n^0 | bond and react electron cloud>>>>>nucleus |
Most atoms are neutral: p+=n0; when p+ doesnt= n0= ion
Anion - - charge
cation - + charge
Element- pure substance made of all same atoms
atomic #- the number of protons an atom has
mass #- n0+p+ in an atom
Orbitals- the area electrons fill- fill orbitals closed to nucleus first
Chemical bonds- attractive force between 2 atoms exists because electrons interact with eachother
ionic bonds
- strong mutual attractoin between ions of opposite charges
- each ion has its own charge (polar)
- ex: NaCl- table salt
covalent bonds
- can be polar or non-polar
- depends on electronegativity of an atoms in bond
- sharing electrons * when shared- electrons count in both orbitals
- can be shown as lines between atoms * number of lines is the number of bonds * extra dots are e-
Hydrogen Bonds
- an attraction between a covalently bonded hydrogen atom and another taking part in another speraerate polar covalent bond
- indivally weak; collectly strong
- 5% strength of covalent bond
- extensive hydrogen bonds give water its important properties
electronegativity- measure of the ability of an atom to pull electrons away from other atoms
Polarity- seperations of - and + into different regions
- caused by unequal sharing of electrons in a bond
Water/Solutions
water has 2 covalent bonds per molecule
oxygen=slightly negative
hydrogen= slightly positive
polarity leads to attraction between water molecules
hydrophilic- loves water
hydrophobic- hates water
Solutions
pH scale
- used to measure acidity
- acid h+
- basic OH-
- ph=-log[h+]
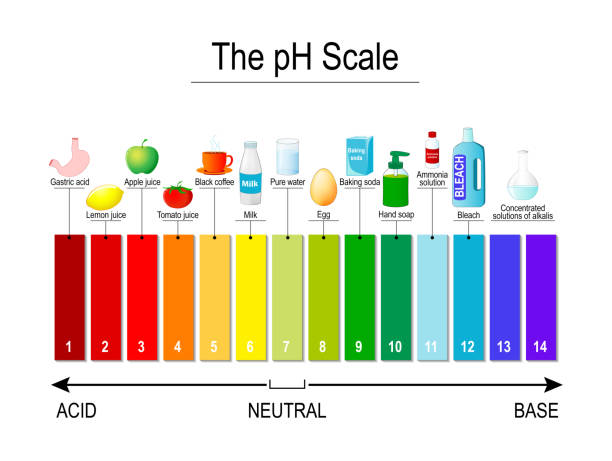
Water - Universal solvent
- dissolves anything polar or ionic
Solute- gets dissolved
solvent- does dissolving
Water Properties
- Cohesion- water sticking to itself
- adhesion- water sticking to other things
- water is less dense as a solid
- water has a high specific heat * takes more energy to heat water
- water has a high heat of evaporation * animals use this to cool down
\
biomolecules
carbs
organic compunds that consist of carbon , hydrogen, oxygen, 1-2-1 ratio
gluclose→ C6H12O6
energy source for animals
building blocks for plants
monomer → monosaccharide
polymer → polysaccharide
Starch
- plants store energy as starch * stems, roots, leaves, fruits
Cellulose
main strucutral component of plants
tough and maleable
glycogen
energy researved for animals
stored in muscles and liver
lipids
organic compounds also known as fat
Functions
- energy storage - harder to break down
- insulation
- membrane function
- coushioning
- hormone regulation
lipids are hydrophobic
1 true monomer
most incorprate fatty acids
phosphlipid
- make a cell membrane * phospholipid bilayer
- phosphate head
- two fatty acid tails (hydrophobic part)
cholestoral
- plays several important roles in the body
- maintains fulidity in animal cells membranes
- used to make important lipid hormones
triglycorides
- typical dietary fat
- glycerol head
- 3 fatty acid tails * full of C and H bonds that store energy
steroids
- 20 C 4 ring structures plus functional groups
- functional groups-
| saturated fats | trans fats | unsaturated fats(plant products [olive oil]) |
|---|---|---|
| all single bonds in tails | double bonds in tails | |
| straight shape | bent shape | |
| soild at room temp. | liquid at room temp | |
| more calories/less healthy | healthier |
proteins
most diverse molecule
shape determines function
structure
- primary- sequence of amino acids in a polypeptide
- secondary- side groups interact with one another; twists and folds
- tertiary- more folding to form a full protein (some proteins stop at this level)
- (word for 4th???)- multiple stage 3s combined to form a protein complex
functions
- transport
- defense (immune system)
- structure
- enzymes
- movement
amino acids
- 20 kinds of amino acids
- different amino acids have different side groups
- what proteins are made of
polypeptides - a chain of amino acids
- unique 3D shape
- can be joined together to form a large complex molecule
- peptide bonds * holds one amino acid to another * is a covalent bond
enzymes
- speeds chem reactions by lowering activation energy by reducing amount of energy without being changed itself
- metabolism- the sum of all reactions in an organism
- substrate- the one specific molecule an enzyme targets
- inhibitors- molecules that prevent enzymes from working
nucleic acids
DNA
- blueprint of life
- contains instructions on how to make proteins in the body
- cant leave nucleus
- double helix
- “rails” of ladder are made from alternating sugars and phosphates
- bases: A T C G
RNA
- copy of DNA
- carrys instructions out of nuclues
- \
\
Cells
cells
eukaryotic
- larger complex cells
- membrane bond organelles
- uni or multicellular
- 2.1BYA
- plants, animals, protist, fungi
prokaryotic
- small simple cells
- no membrane based orangelles
- unicellular
- 1st appeared 3.5BYA
- bacteria and archera
parts of a cell
- nucleus
- golgi apparatus
- lysosomes
- cholorplast
- mitochandria
- vacule
- flagella
- cell wall
- cilia
- cytoskeleton
- smooth endopalsmic rectilium
- nucleous
- rough endoplasmic rectiloium
- ribosome
- perozisome
- cytosol
- centrosoome
\ needed/skipped: / when use passive / active tranport./have all cell parts??
- \ \