7MNT0003 - Week 4 - Appetite
Appetite: Biological Factors Involved in Food Preferences
Outline of the Lecture
Part 1
Definitions
The Satiety Cascade concept
Human eating behaviour
Classical (historical) theories of appetite regulation
Differential effects of macronutrients on appetite
Part 2
Central appetite regulation
Part 3
Peripheral appetite signals
Sleep
Is appetite genetic?
Part 4
Measurement of appetite (online video)
Learning Objectives
By the end of the lecture, students should be able to discuss:
The differential effects of macronutrients on satiety
Peripheral signals (short & long term) involved in appetite regulation and the pathways by which they communicate
Central mechanisms involved in appetite regulation
The role of genetics in appetite regulation
How sleep affects appetite
The main experimental techniques used to investigate effects on appetite
Definitions
Appetite
Defined by the Oxford Dictionary as: “a natural desire to satisfy a bodily need, especially for food.”
More broadly, appetite refers to the entire field of food intake, motivation to eat, and food preference.
De Graaf et al (2004) describe appetite as the internal driving force for the search, choice, and ingestion of food, encompassing aspects of psychobiology.
Hunger
Referred to as the DESIRE TO EAT, which combines hunger and sensory/cognitive inputs (sight/smell of food, time of day, habit, etc.).
Defined as “a feeling of discomfort or weakness caused by a lack of food, coupled with the desire to eat.”
The verb form states: “to have a strong desire or craving for” (Oxford Dictionary).
It acts as a biological drive to seek food and obtain nutrients, as noted in studies by Mattes & Friedman (1993) and others.
Hunger is not directly measurable but inferred from objective conditions and subjective sensations.
Satiety & Satiation
Both concepts involve processes during ingestion that decrease the drive to eat and promote the termination of an eating episode.
They control meal termination/duration and meal size, influenced by rapid signaling from taste, stomach, and upper intestine, and the role of learning and sensory-specific satiation.
Satiation (or intra-meal satiety) refers to postprandial events following an eating episode that inhibit hunger drive and further eating, influencing the time between meals and frequency, along with metabolic signals and gastric distension.
Satiety (or inter-meal satiety) deals with the sensations of fullness experienced between meals.
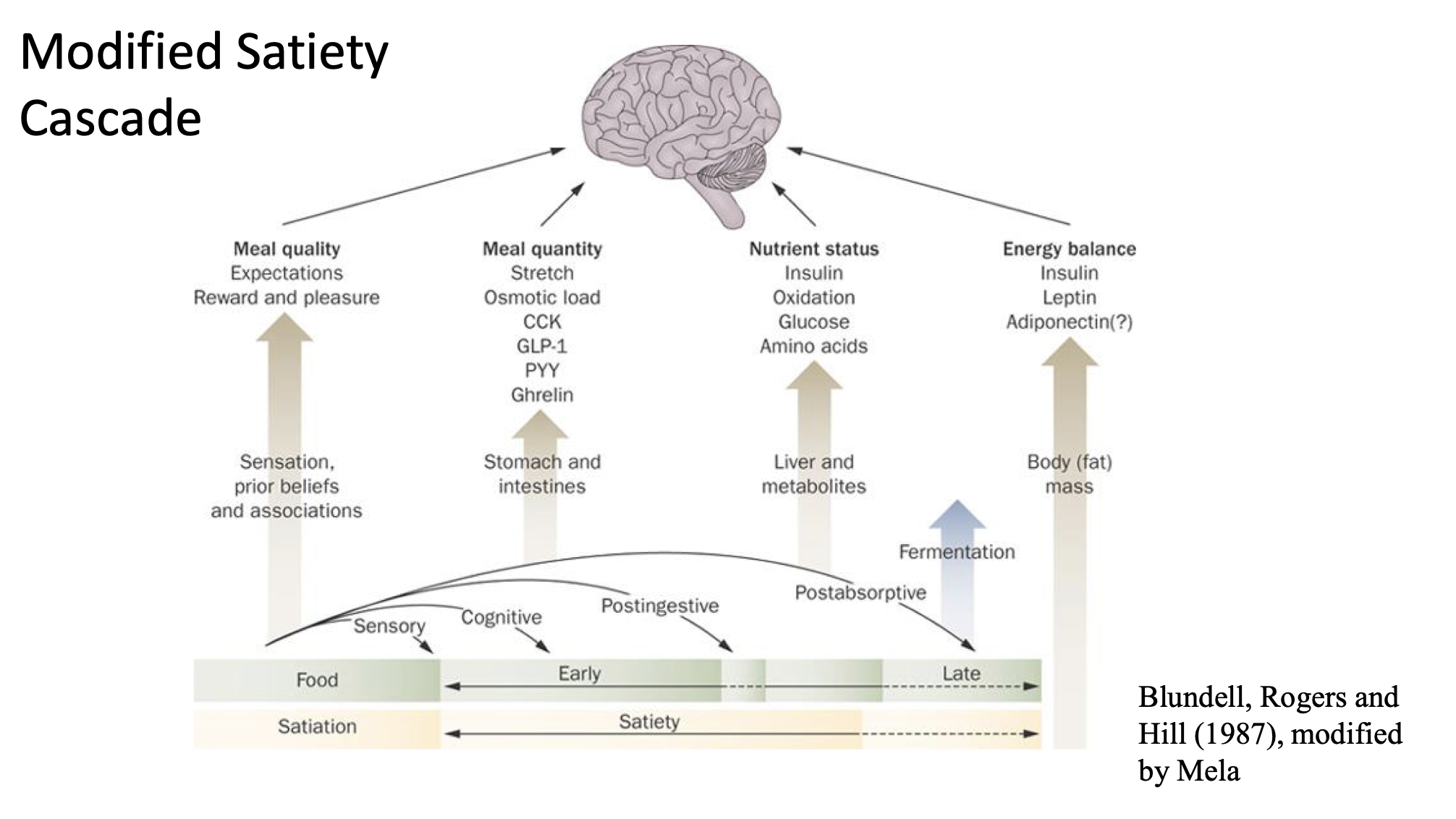
The Satiety Cascade Concept
The satiety cascade is a complex interplay involving various signals that influence hunger and satiety at different stages of eating.
Key components include:
Meal quality
Expectations of food intake
Sensory perceptions (reward and pleasure associated with food)
Meal quantity
Physiological responses such as stomach stretch and osmotic load
Presence of hormones: Cholecystokinin (CCK), Glucagon-like peptide-1 (GLP-1), Insulin, Adiponectin, and others.
The cascade is influenced by energy balance, nutrient status, and previous experiences with food.
Human Eating Behaviour
Food intake is a biological phenomenon that serves physiological needs to maintain homeostasis, but is also influenced by:
Psychological/Cognitive Factors: Learned habits (e.g., habitual meal times), health beliefs, dietary restraint.
Sensory Factors: Visual, olfactory, and gustatory stimuli play crucial roles.
Environmental Factors: Food availability, social interactions, cultural influences, and emotional states can override physiological mechanisms that typically regulate homeostasis (as discussed by de Graaf et al (2004) and others).
Classical Theories of Appetite Regulation
Early views suggested a feedback system maintained a ‘set point’ of food intake, which is now considered overly simplistic (Reviewed by Woods & Ramsay (2007)).
Energostatic Theory
Proposed by Booth & Toates (1974), asserting that energy supply is the primary factor controlling appetite, with each macronutrient having equal satiating effects.
Evidence against this theory includes:
Overconsumption of high-energy-density foods.
High-fat diets tend to be less satiating compared to carbohydrates (CHO) or proteins.
Glucostatic Theory
Original proponent: Mayer (1953), suggesting that blood glucose levels are monitored by glucosensitive receptors in the brain (hypothalamus).
Modified later to emphasize liver glycogen stores as well.
Neural activity has been recorded in specific hypothalamic regions upon glucose administration.
Blocking glucose metabolism stimulates feeding in various studies indicating its role as a hunger signal.
Counterarguments: Plasma glucose levels are tightly regulated, limiting its usefulness in appetite regulation. Normal meals occur before glucose levels get critically low; hence, glucoprivic eating may be an emergency response.*
Lipostatic Theory
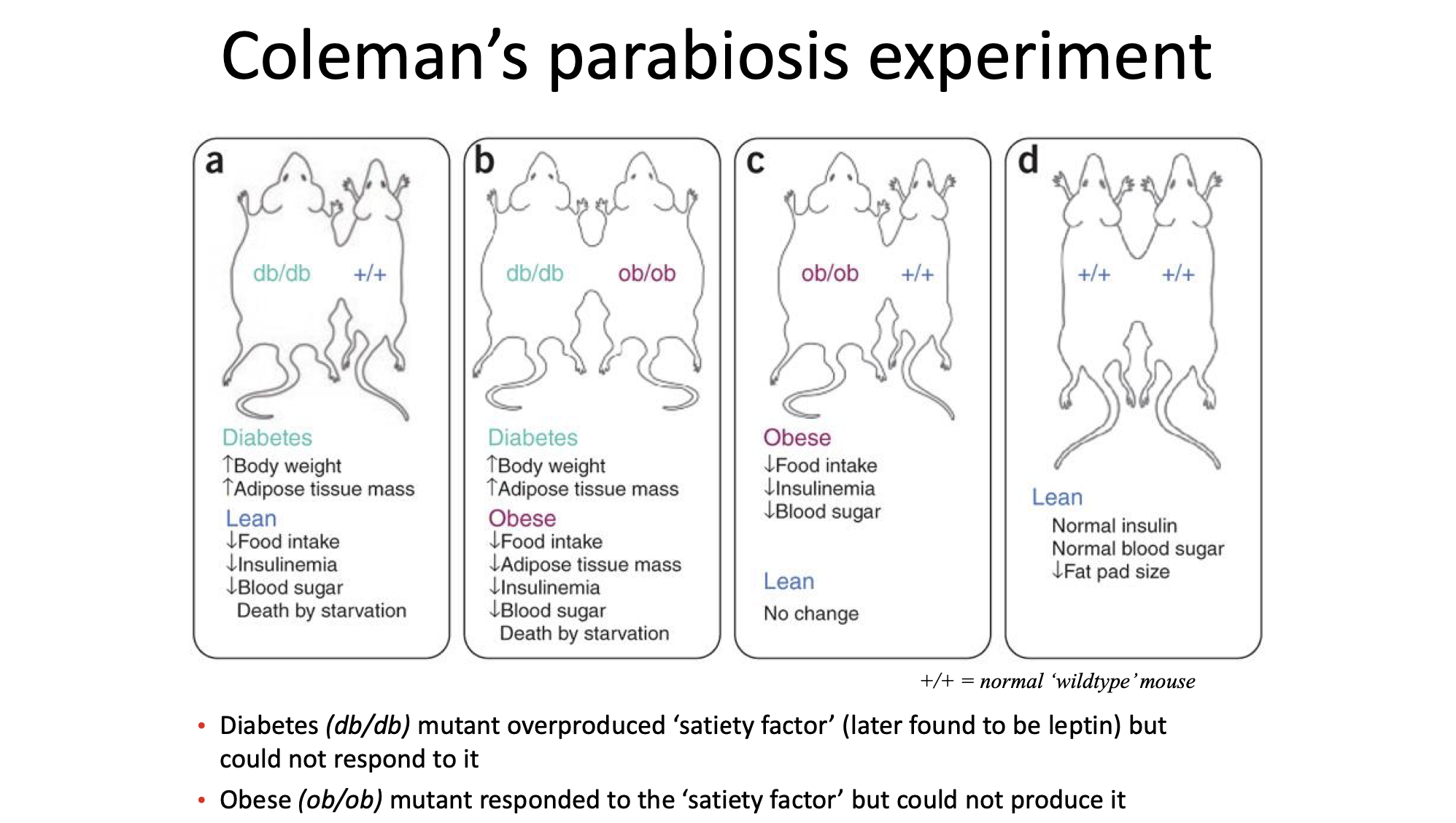
Introduced by Kennedy (1953), asserting that body fat levels are regulated by a ‘lipostatic factor’ secreted by adipose tissue, in proportion to fat stores.
Parabiosis experiments (joining two rats’ circulatory systems) supported this theory, demonstrating how one rat’s obesity affected the other's food intake.
Aminostatic Theory
Proposed by Mellinkoff (1956), suggesting that there's an inverse relationship between serum amino acids and hunger ratings.
Essentially, when serum amino acids increase, hunger decreases.
Supported by evidence indicating that animals strive to consume foods containing the appropriate balance of amino acids, and studies showing enhanced satiety from protein preloads in comparison to carbohydrates or fats.
Possible mechanisms for this theory? - Protein = delayed emtying, greater feeling of fullness.
Minnesota starvation study: Important to read for this theory
Differential Effects of Macronutrients on Appetite
Research indicates differing effects of macronutrients on satiety, showing a hierarchy where protein yields the highest satiety, followed by carbohydrates, and then fat.
For example, a study led by Poppitt et al (1998) demonstrated significantly lower ad libitum intake and hunger scores following a high protein preload.
Findings include specific dietary recommendations emphasizing the importance of protein in regulating food intake, highlighting that protein content, as well as its source, plays a substantial role.
A meta-analysis reported that increasing dietary protein can lead to significant reductions in body weight.
Central Appetite Regulation (Part 2 of Lecture)
Focused on the role of the hypothalamus and the importance of classical brain lesion studies in energy homeostasis.
Key findings include:
Lateral Hypothalamic Nucleus (LHA): Referred to as the “hunger center.” Lesions result in reduced food intake.
Ventromedial Hypothalamic Nucleus (VMH): Considered the “satiety center.” Lesions lead to obesity and reduced physical activity.
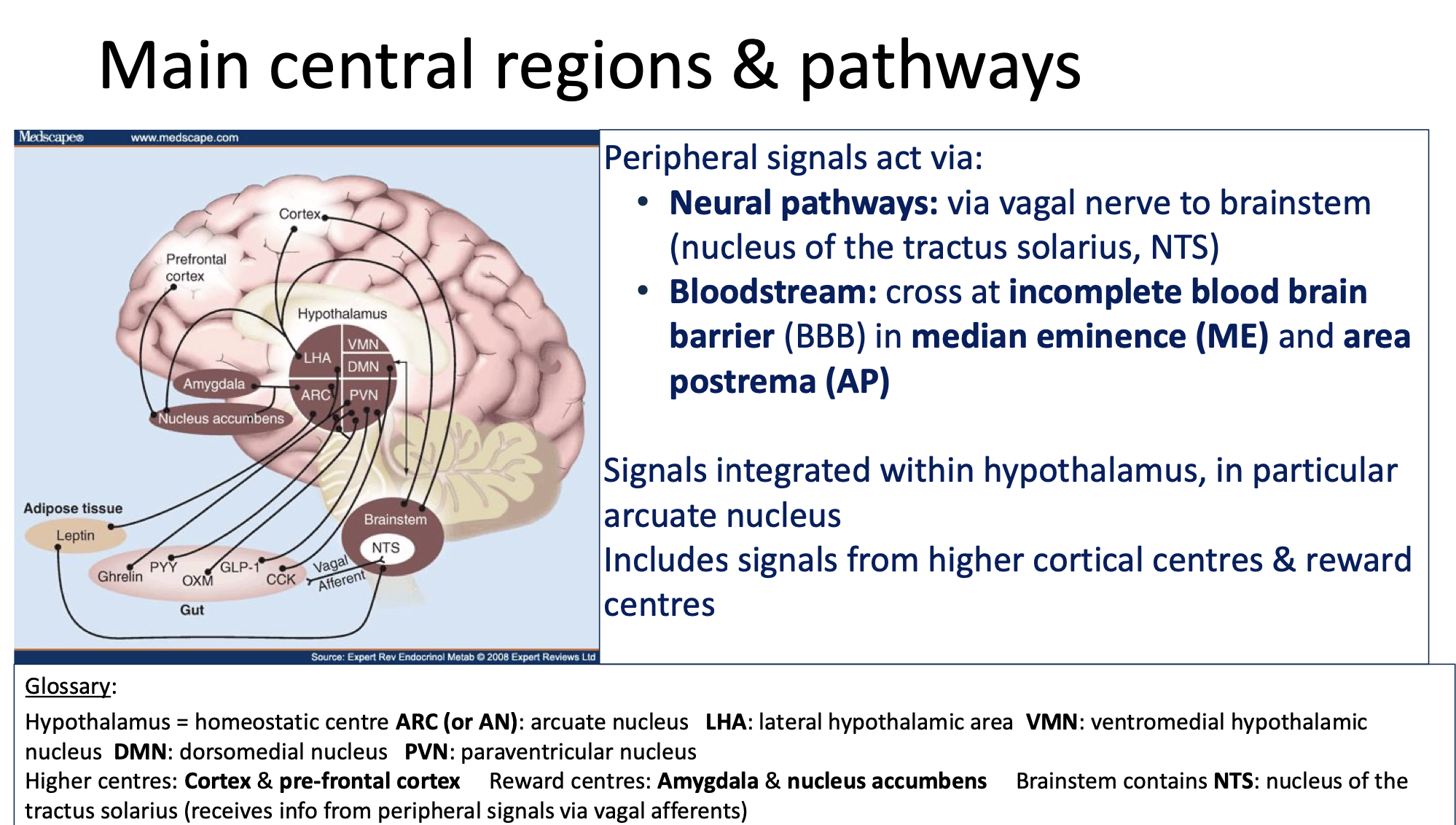
The regulation of appetite involves complex neuronal pathways with multiple reciprocal connections between the hypothalamus, brainstem, and higher cortical areas.
Glossary of Key Areas in the Hypothalamus
Hypothalamus: The primary homeostatic center in the brain.
ARC (Arcuate nucleus), LHA (Lateral hypothalamic area), VMH (Ventromedial hypothalamic nucleus), DMN (Dorsomedial nucleus), PVN (Paraventricular nucleus).
Higher brain centers include regions such as the cortex and prefrontal cortex, with reward centers including the amygdala and nucleus accumbens.
The NTS (Nucleus of the tractus solitarius) in the brainstem is crucial for receiving information from peripheral signals through vagal afferent connections.
Must understand process occurring in diagram below
ARC: 2 primary neuronal populations in ARC integrate signals of nutritional status and influence energy homeostasis
1: Inhibit food intake via expression of CART (cocaine and aemphatamine regulated transcript) and POMC (pro-opiomelanocortin)
2: Stimulate food intake via expression of NPY (neuropeptide Y) and AgRP (agouti-related peptide)
🧠 Step 1: The hypothalamus gathers information
The hypothalamic nuclei shown in the image (like VMN, DMN, PVN, and LHA) constantly receive information about what’s happening in your body — for example:
Blood glucose levels
Hormone levels (like leptin, ghrelin, insulin)
Nerve signals about hunger, stress, or body temperature
⚙ Step 2: The brain needs to act — but it must communicate with the body
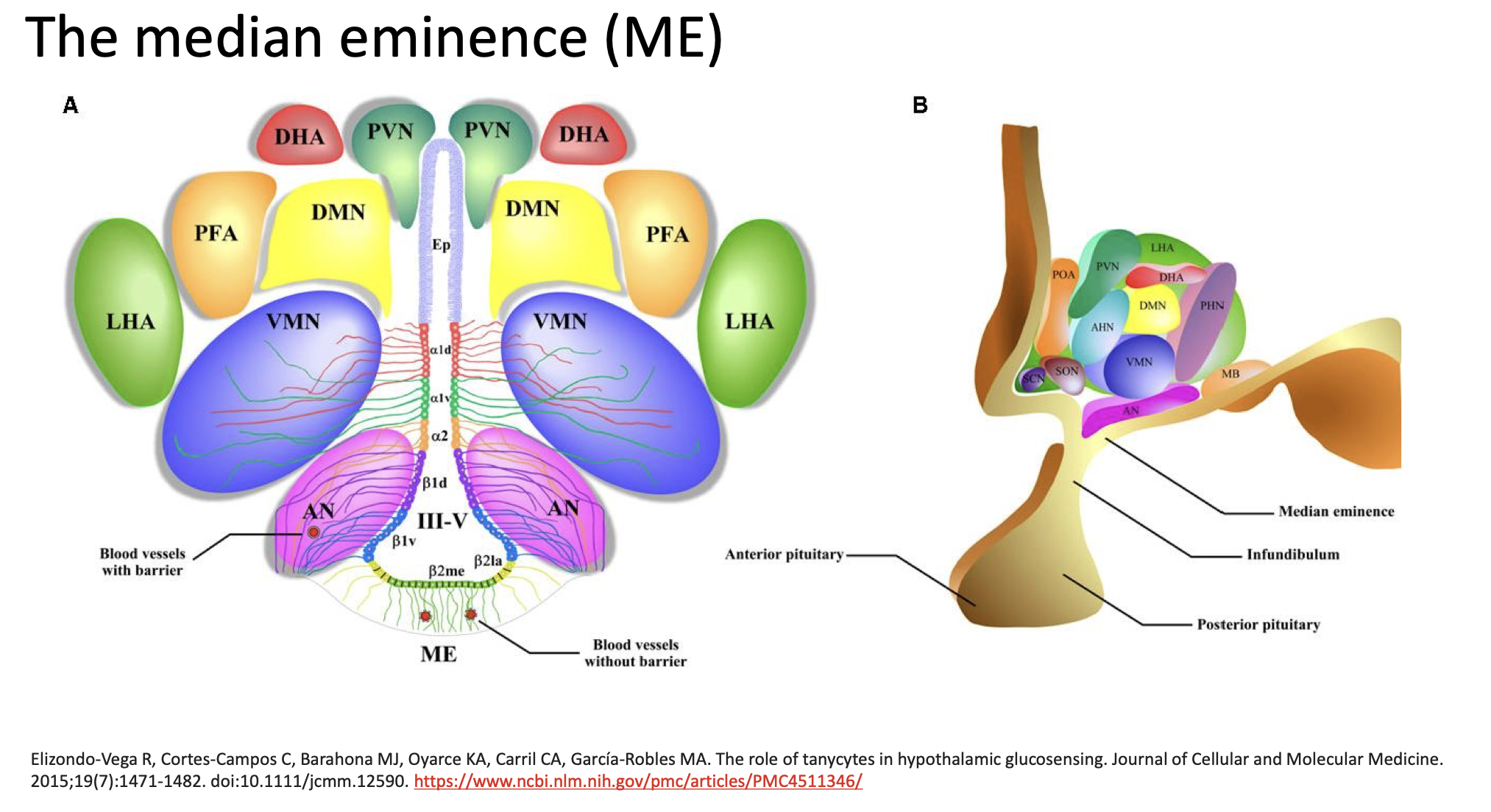
To send out hormonal signals, the hypothalamus uses the median eminence as a “gateway” between the brain and bloodstream.
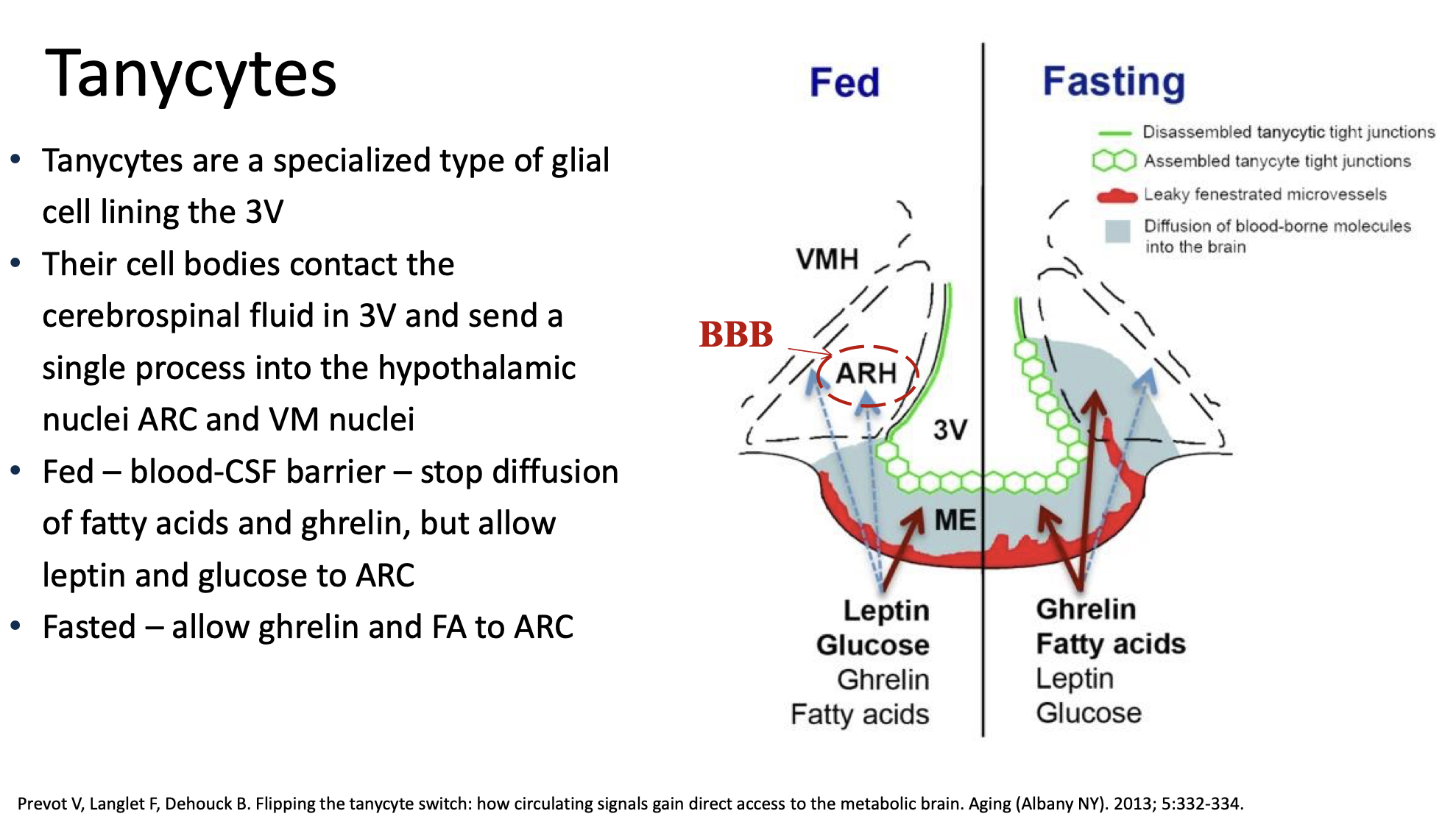
The ME is one of the few brain regions without a full blood-brain barrier, meaning substances in the blood can be detected by special cells called tanycytes.
🔬 Step 3: Tanycytes sense and transport signals
Tanycytes line the third ventricle and extend down to the ME.
They sense nutrients and hormones in the blood (like glucose).
When levels change (for example, blood sugar drops), tanycytes help transfer this information to hypothalamic neurons or regulate which hormones are released into the blood.
Think of tanycytes as “messengers” or “gatekeepers” that monitor what’s happening in the bloodstream and pass this info to brain cells.
🩸 Step 4: Neurosecretory neurons release hormones into blood vessels
Neurons from hypothalamic nuclei (like PVN or ARC/AN) send their axon terminals into the median eminence, where they release releasing or inhibiting hormones (tiny signaling molecules).
Examples:
TRH (thyrotropin-releasing hormone) → controls thyroid activity
CRH (corticotropin-releasing hormone) → controls stress hormone (cortisol) release
GnRH (gonadotropin-releasing hormone) → controls reproductive hormones
🧬 Step 5: Hormones travel to the pituitary gland
The hormones released in the ME enter tiny blood vessels (portal veins) that carry them down to the anterior pituitary.
The pituitary responds by secreting other hormones (like TSH, ACTH, LH, FSH) into the main bloodstream.
These hormones then act on organs like the thyroid, adrenal glands, or gonads — influencing metabolism, growth, stress, and reproduction.
🔁 Step 6: Feedback to maintain balance
When the body’s hormones reach proper levels, signals return to the hypothalamus — telling it to stop or adjust further hormone release.
This keeps everything in homeostasis (balance).
Higher brain centres: hedonics, reward and addiction
• Food intake activates higher brain centres involved in pleasure & reward
• Hedonic feedback integrates with internal signals from body to accentuate
activation AGRP/NPY neurons to induce stronger consummatory behaviour
• Palatable high fat/sugar “obesogenic” foods can motivate eating even when
satiated
• Role of learning: rewarding effect reinforces future behaviour; conditioning
Neurobiological mechanisms of food reward
• Main neurotransmitters involved in food reward: endogenous opioids,
dopamine, GABA (γ-aminobutyric acid)
• Reward pathways linked to differential eating behaviours in individuals with
obesity - greater activation of central food reward regions in participants with
obesity relative to normal weight controls
e.g. when looking at high kcal food images ++activation brain regions involved in
food reward (Stoeckal et al (2008) Neuroimage 41: 410-421)
• Hyper-reactive reward system to palatable food may have role in
pathophysiology of obesity
Hyperphagia: Weakened reward circuit and higher sensitivity to palatability (hedonic properties) - 1: Over eating blunts reward and thus,
2: Increased drive to compensate for weak reward circuit, metabolism increases in the somatosensory cortex, which suggests enhances sensitivity to hedonic properties of food
Peripheral Appetite Signals (Part 3 of Lecture)
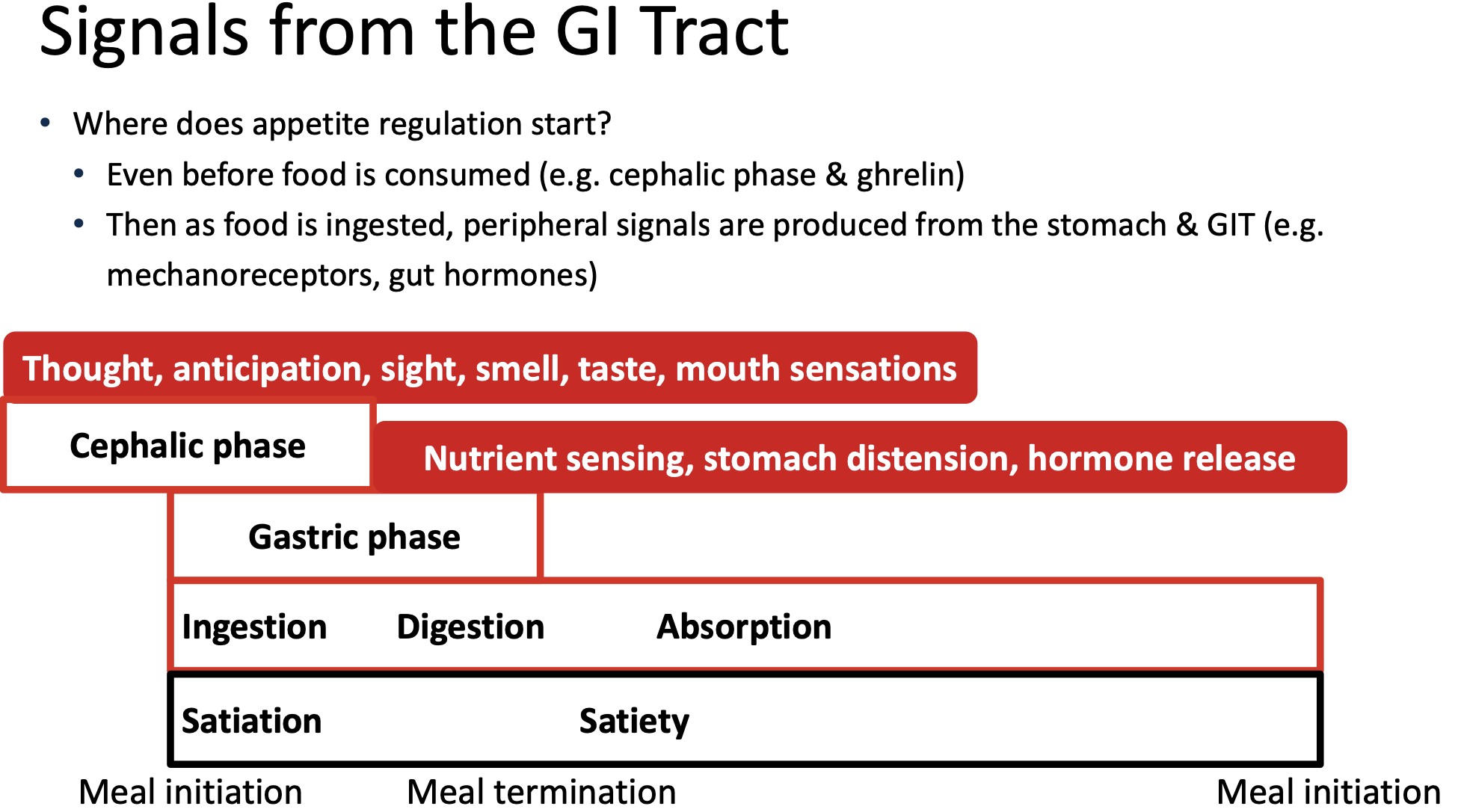
The gastrointestinal tract begins sending signals regulating appetite even before food is consumed, during the cephalic phase.
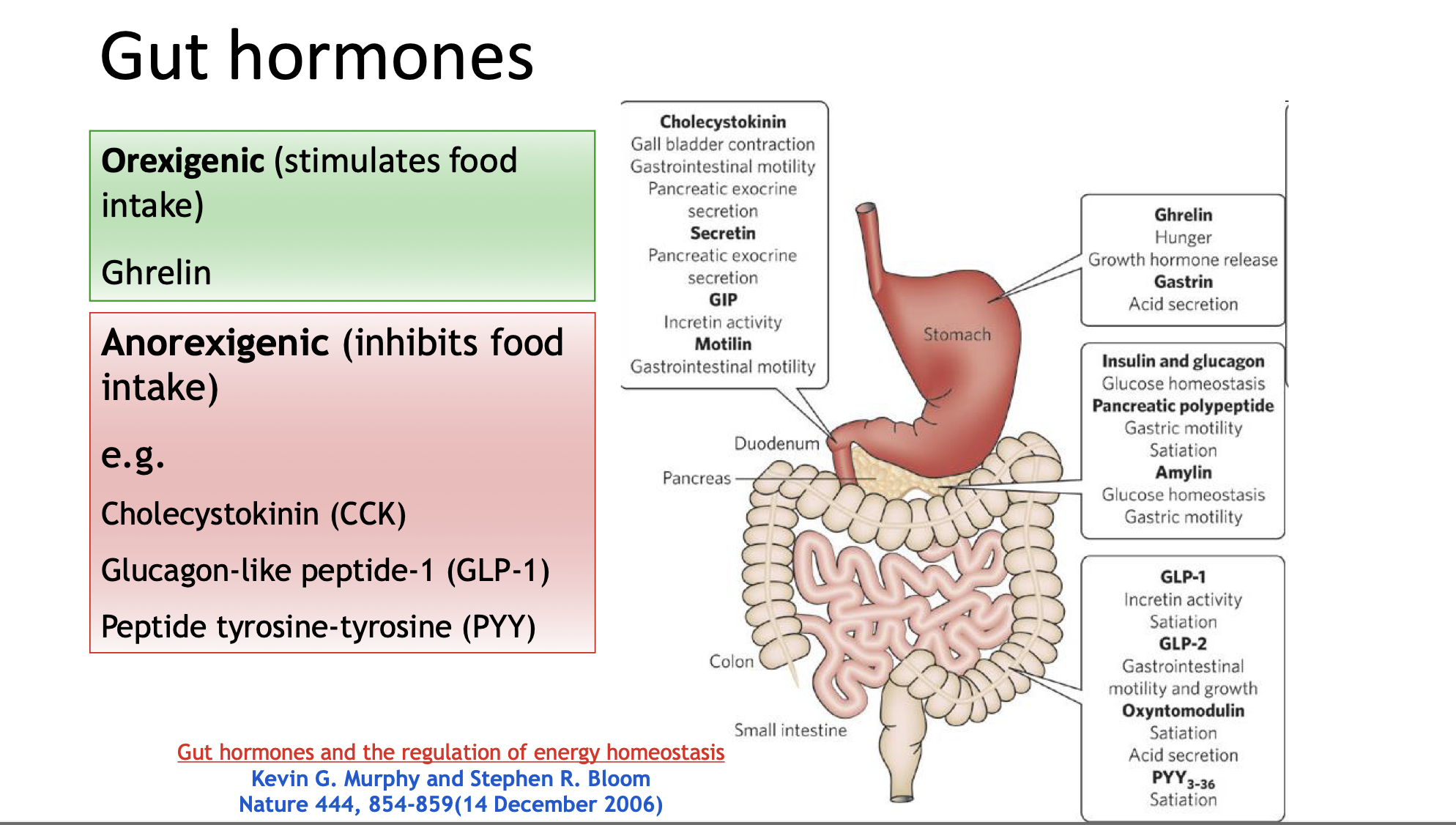
Notable hormones include:
Leptin:
Secreted by adipocytes proportional to fat mass, with profound effects on appetite regulation when administered centrally.
Central and peripheral leptin administration has been found to induce profound decrease in food intake and body weight especially through central administration.
in 1994, ob gene was deemed a gene that encodes leptin: a 146 aminco acid protein
Leptin receptor was cloned a year later.
Ob-Rb variant (involved in appetite) expressed primarily in hypothalamus
Circulating leptin actively transported across incomplete blood brain barrier into accurate nucleus via saturable process
Activates anorexigenic POMC/CART neurons
Inhibits orexigenic NPY/AgRP neurons
Majority of obese animals and humans have high ciruclating leptin levels
Suggestive of relative resistance to leptin action
Selective leptin resistance where certain actions are perserved
Hyperleptinaemia leads to leptin resistance
Mechanisms: Inability to cross BBB, tancytes prevent leptin crossing to arcruate nucleus if high glucose (insulin resistant)
Inhibition of leptin signalling pathway
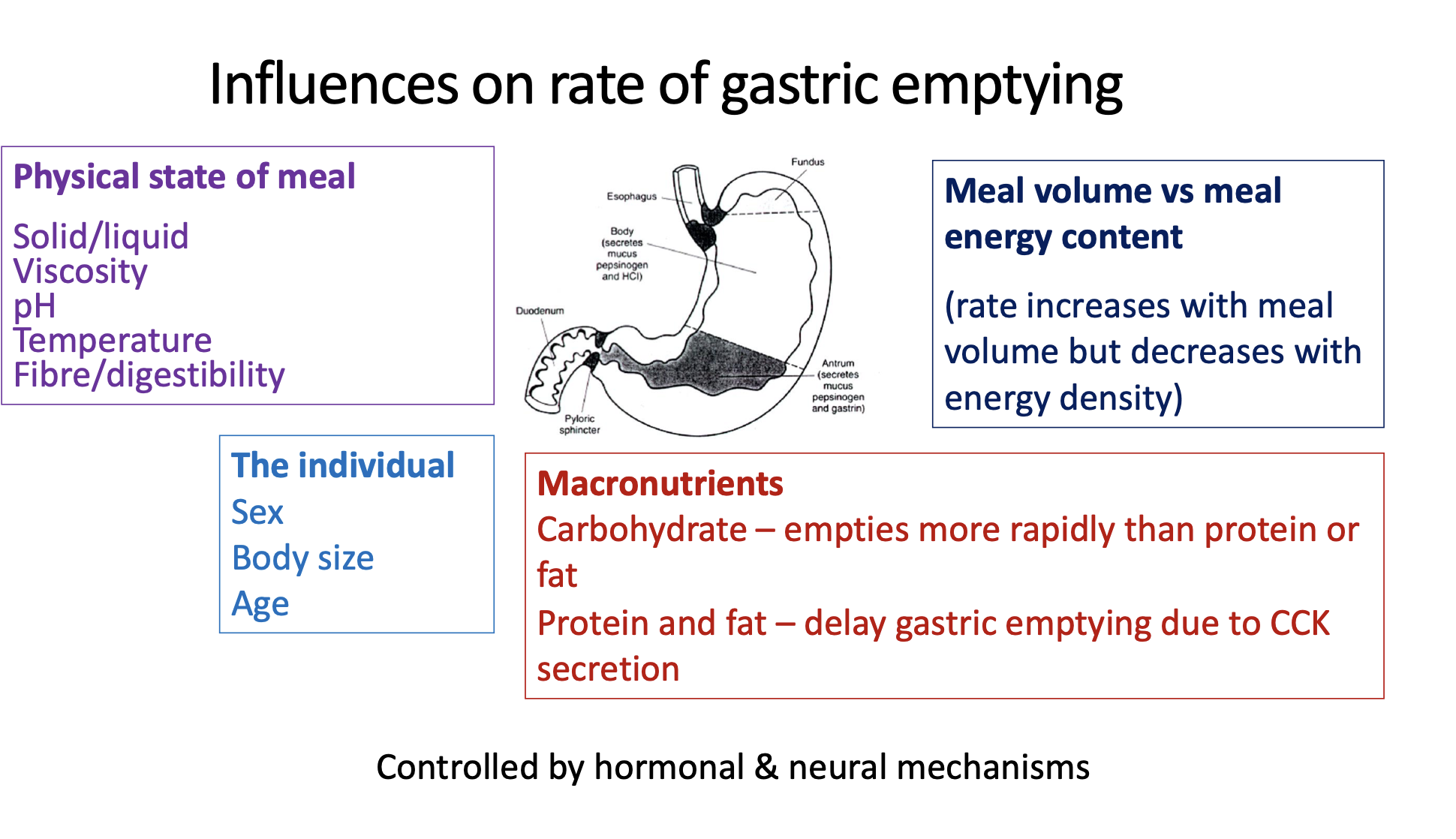
Gastric distension
• As food arrives into stomach
• Stomach muscle wall distended (stretched)
• Activate stretch receptors (mechanoreceptors) → signal centrally via vagal nerve
• Important part of satiation process, particularly if large volume
• Distension induced signals enhanced by intestinal derived gut signals (e.g. CCK) to enhance effects on satiety
• Influenced by rate of gastric emptying (rate by which stomach contents emptied into intestine)
Ghrelin: Produced primarily in the stomach, known as the “hunger hormone,” that stimulates food intake. As ghrelin increases, activation of NPY/AgRP increases = increased hunger
Effect of obesity on ghrelin
• Ghrelin levels are inversely correlated with bodyweight
• Secretion from stomach cells is reduced in obesity
• Ghrelin secretory cells in the stomach no longer respond to secretory stimuli in obesity (norepinephrine and glucose)
• Prevents ghrelin action in the brain, causing a state of central ghrelin resistance
• Ghrelin levels rise after weight loss – this may contribute to weight regain after weight
loss intervention
• Ghrelin resistance is also reversed following weight loss
Cholecystokinin (CCK): Released by L cells in the duodenum and jejunum, responding to protein and fat intake. This hormone plays a key role in signaling satiety and delaying gastric emptying.
Hormone & neuropeptide released by L cells in duodenum & jejunum in response to protein & fat
➢ Rapidly released within 15 min of meal initiation
• CCK-A receptors found in vagal afferent neurons & circular muscle cells of pyloric region of
stomach and activated by locally released CCK:
➢ Acts on vagal afferents → activate NTS → signal to hypothalamus → anorectic effects
➢ In pyloric region of stomach → contract pyloric sphincter → delay gastric emptying
• CCK may also act directly on satiety via receptors in brain (not clear if crosses BBB)
• Peripheral administration inhibits FI
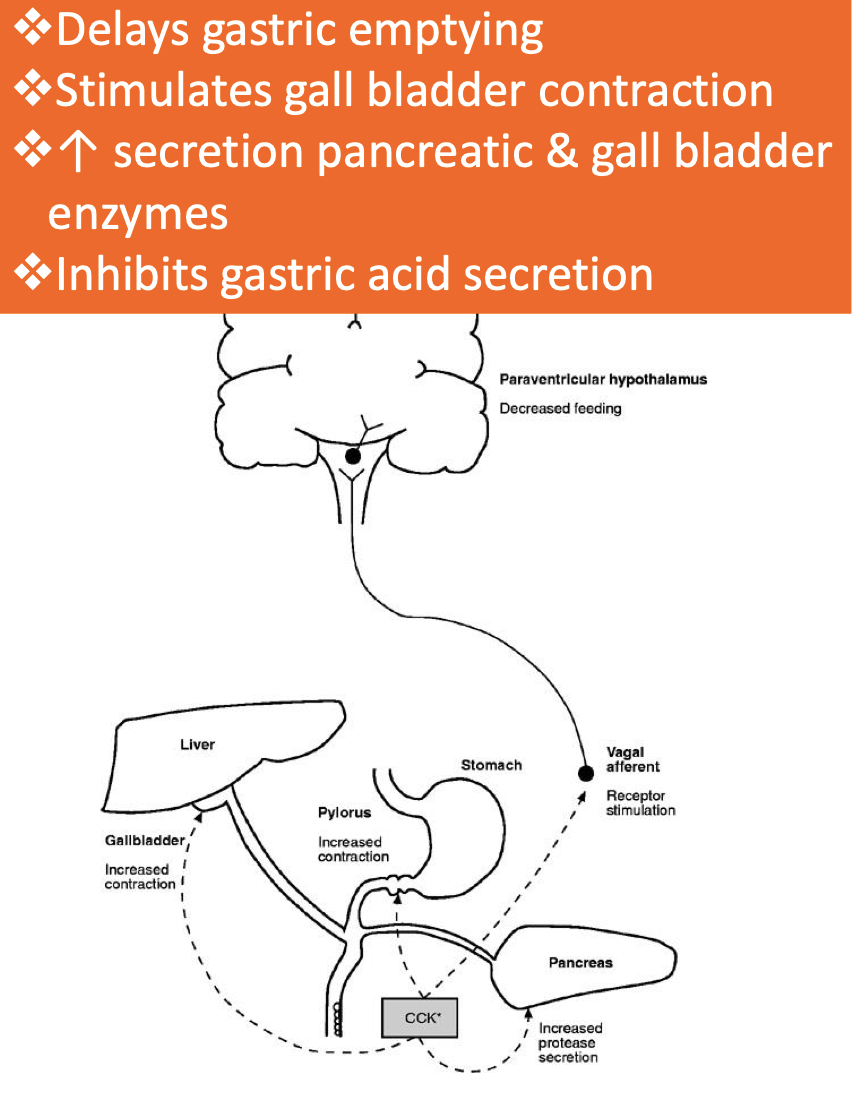
Effect of obesity on CCK
• Short-term signal to increase fullness – therefore role in satiation (might shorten an
eating episode)
• Not much is known about effects of obesity in human subjects on CCK release or
sensitivity
• Fasting levels are lower in severe obesity and this is sustained postprandially (however
just 1 study)
• Postprandial CCK release reduced following weight loss – survival mechanism?
Peptide YY (PYY): Similar to CCK, it is released from L-cells in the ileum and colon in response to food intake, particularly fat and fermentable carbohydrates.
Released into circulation by L-cells (co-secreted with GLP-1) in ileum & colon in response
to food intake, esp of fat & fermentable CHO
• Conc released proportional to EI (fat>protein>CHO)
• Biphasic – first peak within 15 min, second peak after 1-2h
• Appetite effects: Anorectic
• Peripheral administration → ↓ food intake (acute) & ↓ BW & adiposity (chronic)
• Elevated up to 6h after a meal
• Signals centrally by crossing via BBB to act directly via non-saturable mechanism & may
also activate vagal nerves
• Mode of action:
• Ileal brake → Delay gastric emptying & ↓ pancreatic & gastric juice secretions, ↑ ileal
fluid absorption
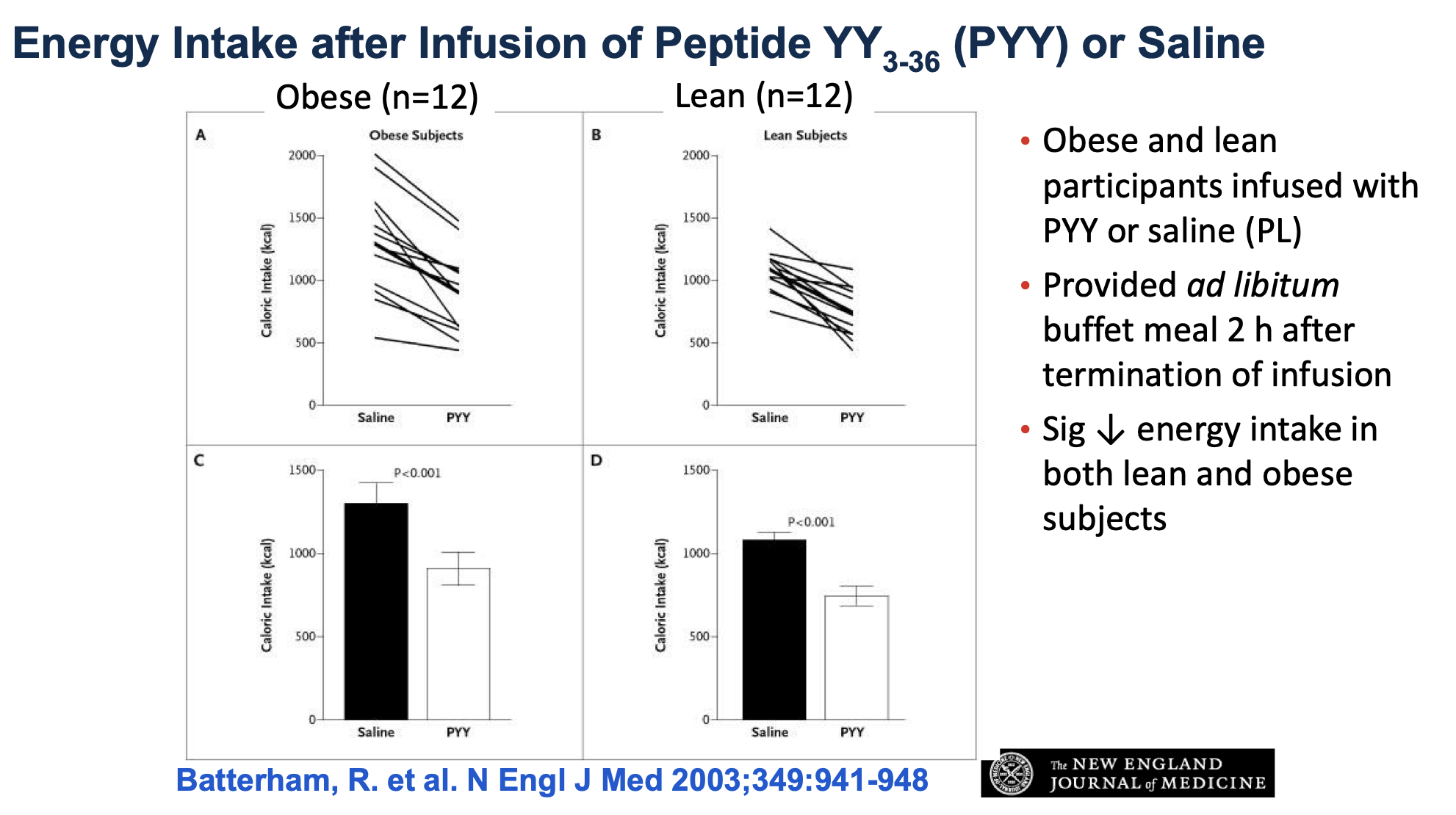
PYY led to significantly reduced energy intake in both lean and obese subjects^
Effect of obesity on PYY
• Reduced postprandial responses in PYY levels in obesity
• Figure: the PYY response in obese subjects was significantly lower than in
normal-weight controls 90 min after each of six test meals of increasing caloric content.
• Reduced response corresponded with lower subjective ratings of fullness in
obese subjects
Glucagon-Like Peptide 1 (GLP-1): A crucial anorectic hormone produced in response to food intake, known to inhibit food intake and promote satiety through its signaling pathways.
Produced by L-cells in small intestine
• Rapidly released into circulation in response to food intake, particularly CHO and fat
o Conc released proportional to EI
o Short half life – rapidly deactivated following release
• Appetite effects: Anorectic
o Peripheral admin →↓ meal size (acute) & ↓ BW (chronic)
• Signals centrally via activation of vagal nerves & may cross BBB to act directly – GLP-1 receptors found throughout the brain, including PVN, ARC, LH of hypothalamus
o Stimulates POMC/CART neurons, inhibits NPY/AgRP neurons
• Other modes of action:
o Ileal brake → Delay gastric emptying & ↓ gastric juice secretions
o May ↑EE & appears to ↓ circulating ghrelin levels
o Powerful incretin → enhances insulin biosynthesis
Effect of obesity on GLP-1
• Attenuated effect on postprandial GLP-1 response to test meals
• Weight loss associated with lower GLP-1 levels (and greater appetite)
• Exercise increases postprandial GLP- release so may help weight loss maintenance this
way?

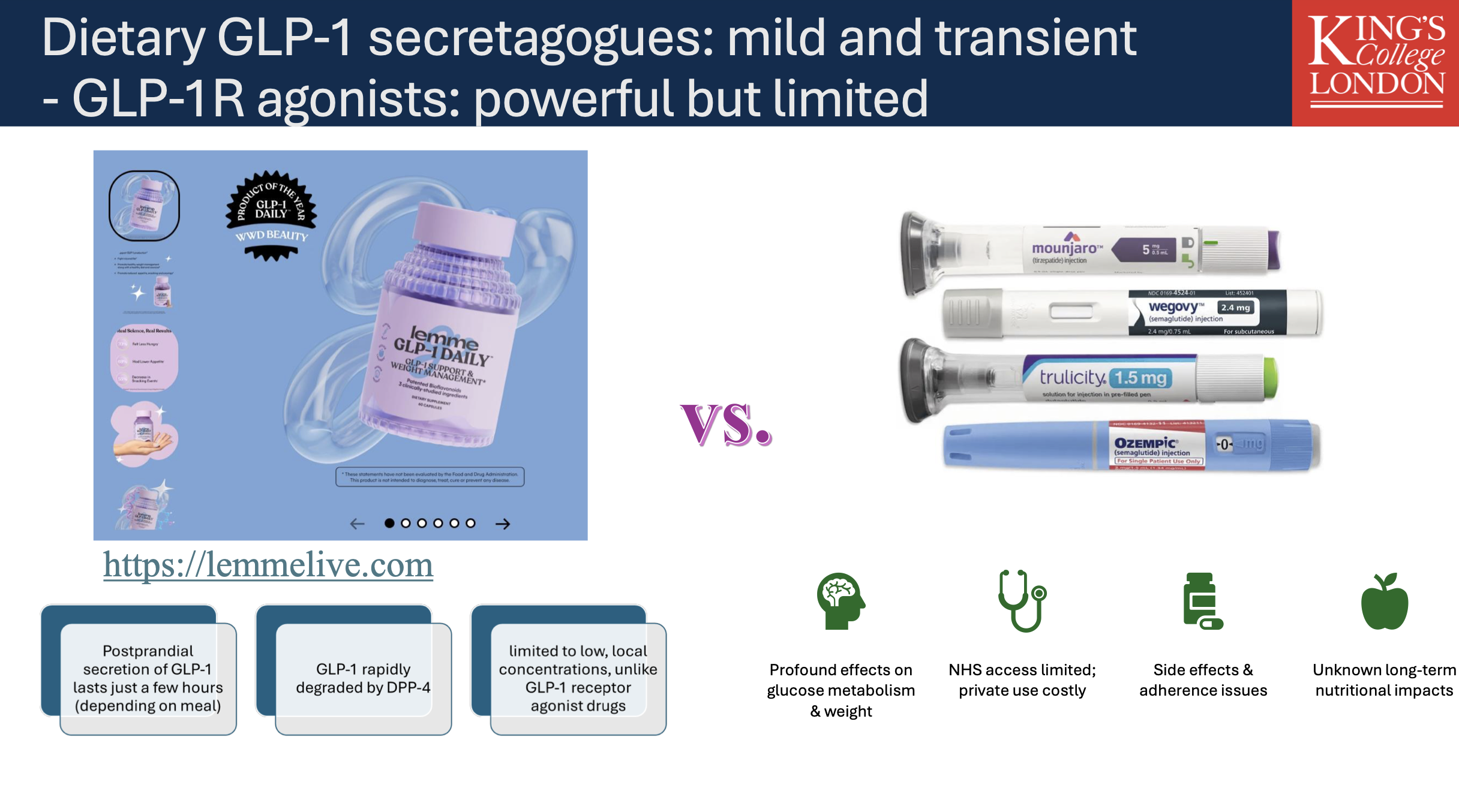
GPL-1 is rapidly degraded and cannot be “naturally” increased as advertised by these medications. Whilst they have profound effects on appetite control, they are limited and costly. We are also unaware of long-term health impacts.
Results of Appetite Hormones in Obesity
Ghrelin levels are generally inversely correlated with body weight; individuals who are obese have a blunted response to ghrelin and may exhibit ghrelin resistance.
Leptin resistance is prevalent among obese populations where high levels of circulating leptin do not produce expected reductions in food intake, possibly due to impaired signaling pathways or difficulties crossing the blood-brain barrier.
Effects of PYY and GLP-1 in regulating appetite can diminish in the obesity state, emphasizing the complications in the management of weight and appetite.
Genetics of Appetite (Part 4 of Lecture)
Genetic factors significantly influence appetite, with the Behavioral Susceptibility Theory positing that appetite mediates genetic risk of obesity.
The impact of genetics on appetite has been shown to be substantial in twin studies identifying over 70% (73%) heritability for appetite traits like satiety responsiveness.
Research into single nucleotide polymorphisms (SNPs) indicates that specific genetic variations may enhance food cravings or affect satiety signaling mechanisms.
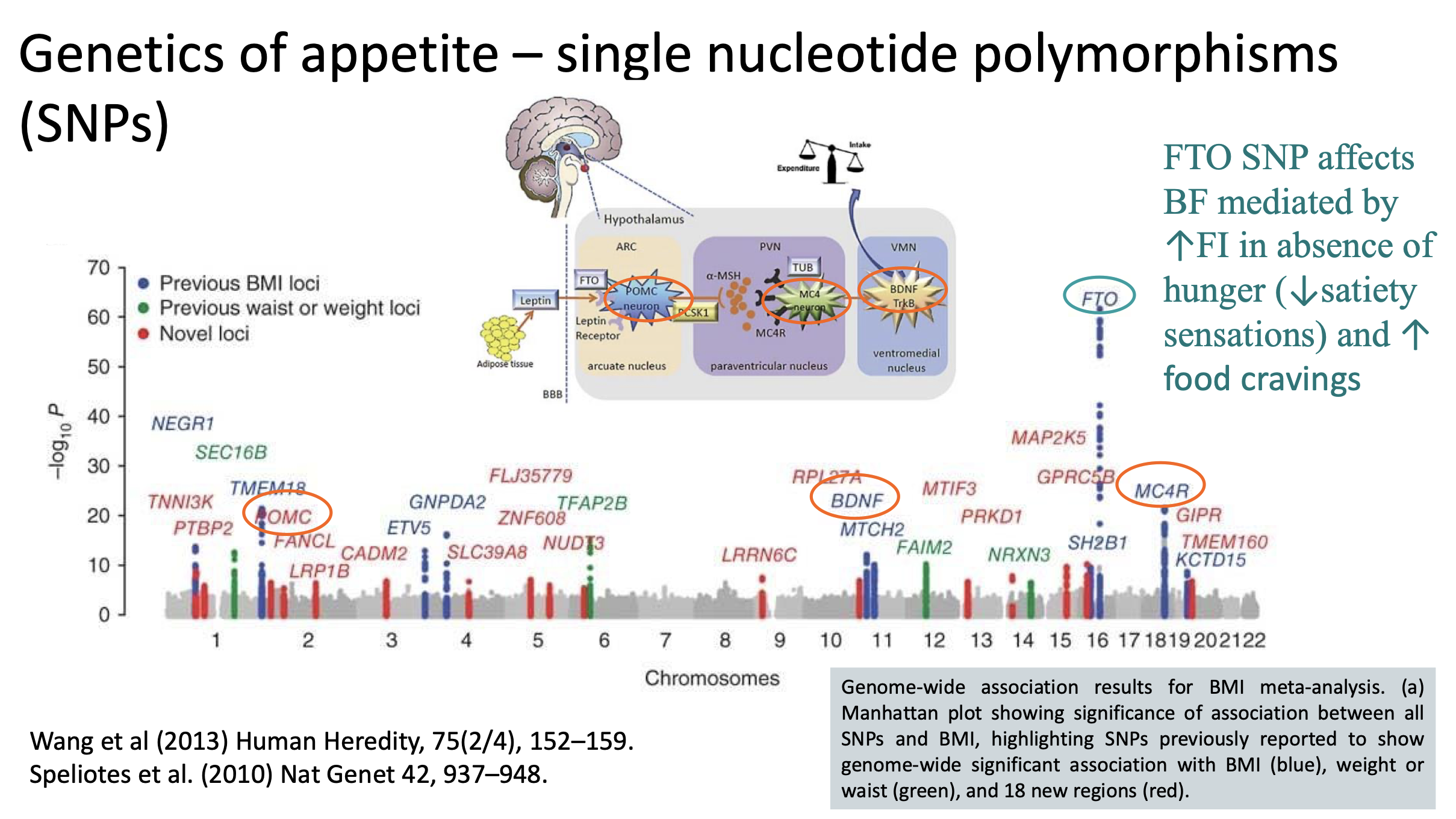
Key take home messages on genetics of appetite
• A substantial share of obesity-related SNPs operate via neuronal regulation of food intake,
satiety, and reward.
• Because the effect of any single SNP is small, aggregating many into scores (Polygenic Risk
Scores) offers more predictive utility — but still far from perfect.
• Many appetite-related SNPs are regulatory (non-coding) variants that affect when, where, and how much a gene is expressed, often in the brain.
• Key challenge is moving from SNP association studies to mechanistic insights into how genetic variation alters appetite circuits.
Sleep and Appetite
Sleep is a significant factor, with short sleep duration linked to obesity increases, with studies indicating a 38% higher incidence of obesity in those reporting short sleep periods.
Poor sleep affects appetite hormones like ghrelin and leptin, leading to increased hunger, weakened reward responses in the brain, and potential preferences for high-calorie foods.
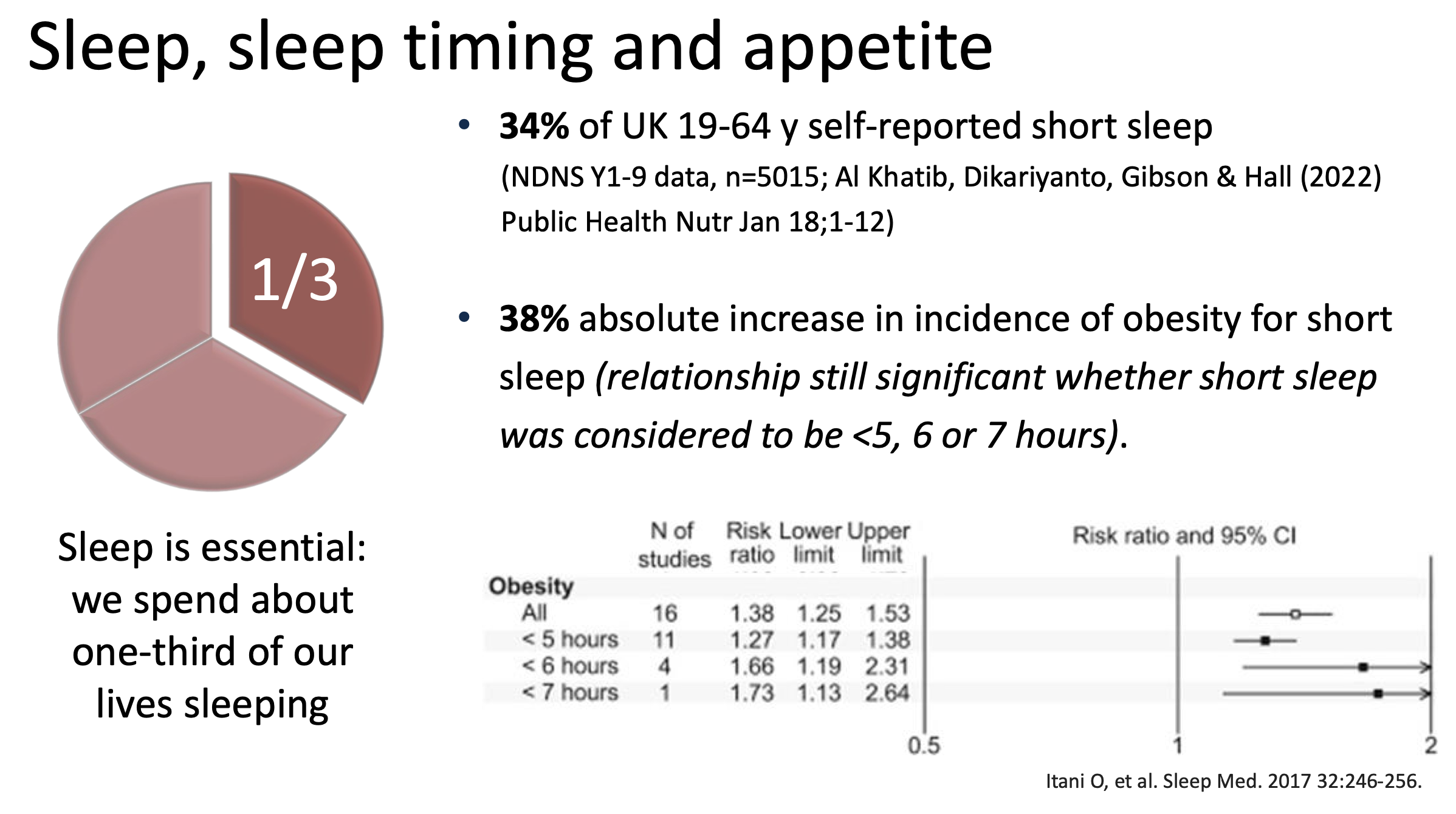
Poorer diet and greater adiposity in UK short sleepers
National Diet and Nutrition Survey, Years 1-9 (5015 adults 19-64 y)
Short-sleepers (<7 h): n=1711.
• Compared to 7-8 h sleep group:
higher waist circumference and BMI*
• Lower fruit+veg, fibre, protein, n-3 fatty acids, folate, vit C, potassium, iron intakes.
• Higher sugar intakes
WC increased 2.3 cm BMI increased 1.3 kg/m2
Randomised controlled trials show that short term
sleep deprivation cause increased energy intake
• Short term sleep deprivation led to an average net increase in energy intake of 385 kcal/d (no difference in energy expenditure)
• Short-term effects of dramatic sleep deprivation rather than chronic exposure to modest sleep deprivation
Experimental Techniques for Measuring Appetite
Measuring appetite is complex due to various influencing factors, and methods include:
Visual Analog Scale (VAS) for hunger assessment.
Controlled feeding studies to assess post-meal satiety and energy intake variations.
Free-living food intake measurement techniques, though challenging in terms of reliability and practicality.