Enzymes
Homework 1
Biological catalysts
Production of ATP (energy) id a complex chain of reactions
Enzymes control the rate of chemical reactions, speeding them up in order to be able to maintain life functions (most organisms would otherwise conduct them too slowly) E.g Lactose is broken down into small bits to make it digestible for us by Lactase.
Metabolism = all the reactions that happen within the organism
Chemical reaction = A+B (reactants) → C+D (products)
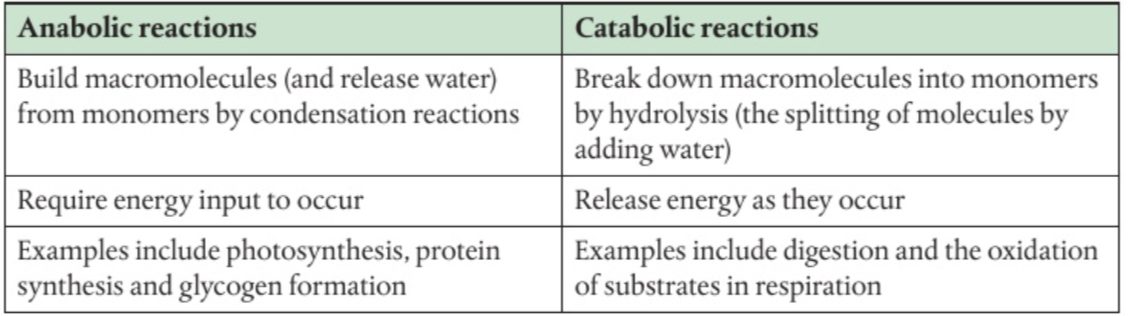
Anabolic reactions = metabolic reactions joining together complex molecules
Protein synthesis, DNA synthesis and replication, carbohydrate synthesis (complex)
Catabolic reactions = metabolic reactions breaking down complex molecules
Digestion, cellular respiration, digestion of complex carbon compounds
Energy is the capacity to cause change
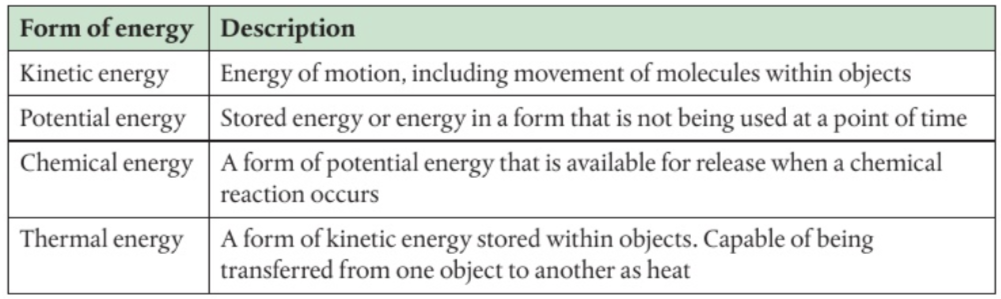
kcal = measurement of heat
Enzymes = specifically shaped globular proteins with an active site that perfectly fits the substrate it breaks down
The active site contains very few amino acids, reactions are catalysed by these
Fischers lock-and-key model now adapted to the induced-fit model
Enzymes change in shape when they come into contact with their substrates
Activation energy: energy required for a metabolic reaction, lowered by enzymes
Minimum rate of motion: kinetic energy provided by the substrate for the reaction
Energy barrier: there are fewer molecules with enough energy to pass the energy requirement for a reaction. There are two ways of solving this:
Increasing energy present in the molecule (addition of heat = more kinetic energy)
Lowering the energy required for the reaction
Reactions keep going until there is equilibrium between reactants and products
For reactions to occur, substrates and enzymes need to collide, which requires kinetic energy
Often, substrates or enzymes are anchored in structures like cell membranes
E+S ⇔ ES ⇔ E+P
E = Enzyme, S = Substrate, ES = Enzyme-Substrate Complex, P = Product
Class work 1
Protein Folding
Non—polar amino acids: not soluble in water
Amino acids can be positively or negatively charged
Amino acids are linked together by peptide bonds
Protein folding gives the protein its function, i.e channel proteins (aquaporin, sodium-potassium pump, etc) are folded to be hollow in the center, allowing for other molecules to pass through
Amino acid chains bend and form kinks, creating linkages and interactions
Denaturation = peptide bonds breaking down, protein loses structure and can no longer bind with the substrate at the active site
Exergonic reactions: energy is released, energy of reactants is higher than energy of products
Endergonic reactions: energy is consumed, energy of products is higher than energy of reactantser
Homework 2
Reactions depend on molecular collisions. The faster molecules move, the more they collide, and with greater energy
Reactions with no enzymes increase rate of reaction as temperature rises, but due to denaturation, reactions with enzymes have an upper limit
Intermolecular bonds become stressed and broken under high temperatures = denaturation
Denaturation can be temporary if the temperature returns to a suitable level, but if the temeprature is so high that it prevents the enzyme from returning to its native state
Denaturation from too high pH: when a solution becomes acidic, the amount of hydrogen ions rise, and these ions (positive) can bond with the negative charges in the amino acids in the active site (or subsrate) preventing them from bonding together by preventing a matching of charges
Denaturation from too low pH: when a solution becomes more basic, the amount of hydroxide ions rises, and these bond with positive charges in the amino acids, once again preventing matching of charges and thereby any form of bonding
Most enzymes operate in areas of the body with somewhat neutral pH levels, but there are exceptions
Pepsin is an enzyme active in the stomach
Collision theory: if the amount of reactant molecules increases, the rate of reaction will speed up, since there will be more molecules to collide with, generating more energy. This has an upper limit, since enzymes have a maximum speed they can operate at and only one active site
Class Work 2
Glucolysis
Part of respiration, step one of converting sugar into ATP, involves 10 enzymes
Higher molecular motion = higher chance of collision
Thing that accelerate molecular motion:
Increased substrate concentration (to a limit)
Increased temeprature (to a limit)
Increased pressure
Decreased area