Oncogenic Viruses
Selman Ali
Viruses and Cancer
Rous’s protocol for inducing sarcomas in chickens
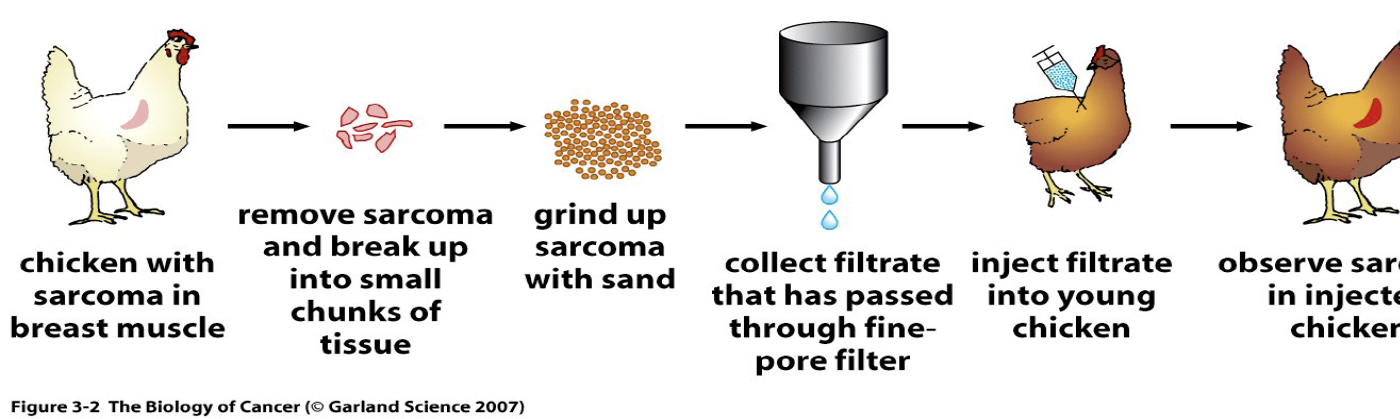
Sarcomas are rare cancers that develop in the supporting or connective tissues of the body such as muscles, bones, nerves, cartilages, blood vessels and fat.
Oncogenic viruses in human
Currently, there are seven recognized human oncoviruses:
DNA/Epstein-Barr Virus (EBV), also known as HHV-4
DNA/Human Papillomavirus (HPV),
DNA/Hepatitis B virus (HBV),
RNA/Hepatitis C virus (HCV),
RNA/Human T-cell lymphotropic virus-1 (HTLV-1),
DNA/Human Herpesvirus-8 (HHV-8), also known Kaposi sarcoma-associated herpesvirus, and KSHV
DNA/Merkel Cell Polyomavirus (MCPyV)
Viruses Associated With Human Cancers

Facts
Viral cancers do not normally arise acutely after infection, but instead develop 15–40 years later.
In cancers, viral replication is either diminished or absent
The virus exists intracellularly as naked nucleic acid in the form of a plasmid, episome, or cellular-integrated genome
DNA virus genomes can integrate directly into the host genome, while RNA virus genomes must undergo reverse transcription to DNA before integration can occur
Viral carcinogenesis
Events in infected cells that may lead to cancer
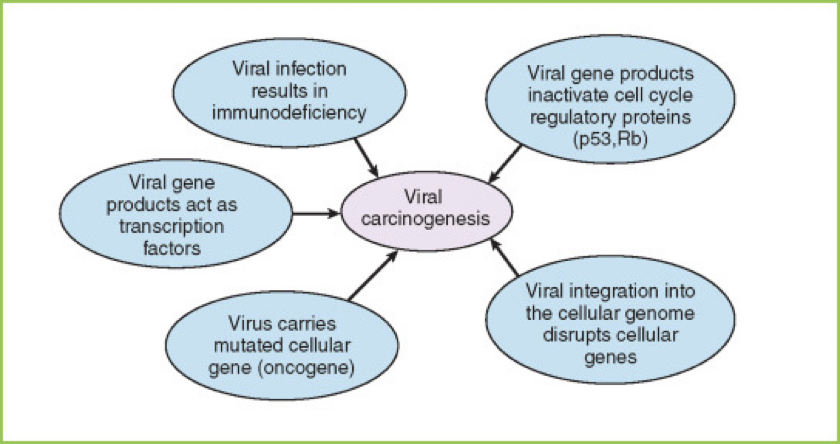
How do viruses contribute to cancer?
Integrations that cause activation or inactivation of oncogenes or tumor suppressors (e.g. RNA viruses)
Expression of genes that alter key signal transduction pathways
Chronic activation of inflammatory responses
Cell Transformation by Viruses
Transformation is a change in the morphological, biochemical, or growth parameters of a cell.
Transformation may or may not result in cells able to produce tumours in experimental animals (transformed cells do not automatically result in the development of 'cancer‘).
Carcinogenesis is a complex, multi-step process in which cellular transformation may be only the first
Transformed cells have an altered phenotype, which is displayed as one (or more) of the following characteristics:
Loss of anchorage dependence
Loss of contact inhibition
Colony formation in semi-solid media
Decreased requirements for growth factors
Changes in transformed cells by oncogenic viruses
increased growth rate and life span in vitro
chromosomal abnormalities
synthesis of new antigen
potential of forming tumours in animals
virus-specific macromolecules may be detected
Disturbance of the cell cycle by oncoviruses
A proto-oncogene is a gene whose protein product has the capacity to induce cellular transformation given it sustains some genetic insult.
An oncogene is a gene that has sustained some genetic damage and, therefore, produces a protein capable of cellular transformation
The process of activation of proto-oncogenes to oncogenes can include retroviral transduction or retroviral integration point mutations, insertion mutations, gene amplification, chromosomal translocation and/or protein-protein interactions
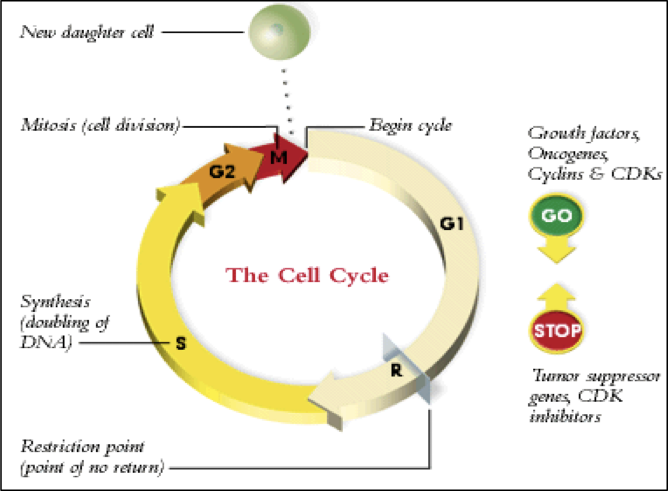
Tumour suppressor genes represent the opposite side of cell growth control, normally acting to inhibit cell proliferation and tumour development
How do viruses transform cells?
Viruses cause the abnormal expression of normal cellular genes.
Contain viral oncogenes (v-onc) derived from a normal cellular gene (c-onc).
v-onc can differ from c-onc in number of ways
Transformation mechanism of oncogenic retroviruses
Viruses produce proteins that interfere with normal cell processes (cell cycle).
EBV- Epstein Barr Virus- human herpesvirus 4 (HHV-4)
Most potent transforming agent,
Widespread in all human populations (over 90%)
Usually carried as an asymptomatic persistent infection (latent infection, latency).
The virus is associated with the pathogenesis of certain types of lymphoid and epithelial cancers, including
Burkitt lymphoma (BL),
Hodgkin disease and
nasopharyngeal carcinoma (NPC).
EBV
Isolated in 1964 from tumour samples (Burkitt’s lymphoma).
The virus infect B cells through CD21 and epithelial cells
Envelope and tegument proteins differ in size from other herpesviruses.
Two EBV types (A & B) circulate in human
Causing infectious mononucleosis in adolescents
Coding potential for around 80 proteins
EBV gene expression during latent infection in host cells
Six EBNAs: 1, 2, 3A, 3B, 3C and LP
Three Latent membrane protein (LMP): LMP-1-2A, and -2B
EBV Latency Phase
EBNAs Gene expression in host cells
Latent membrane protein (LMP)
Latency III
All EBNAs
LMP-1-2A, and -2B
Latency II
EBNA-1 (only)
LMP-1-2A, and -2B
Latency I
EBNA-1 (only)
Latency 0
No expression
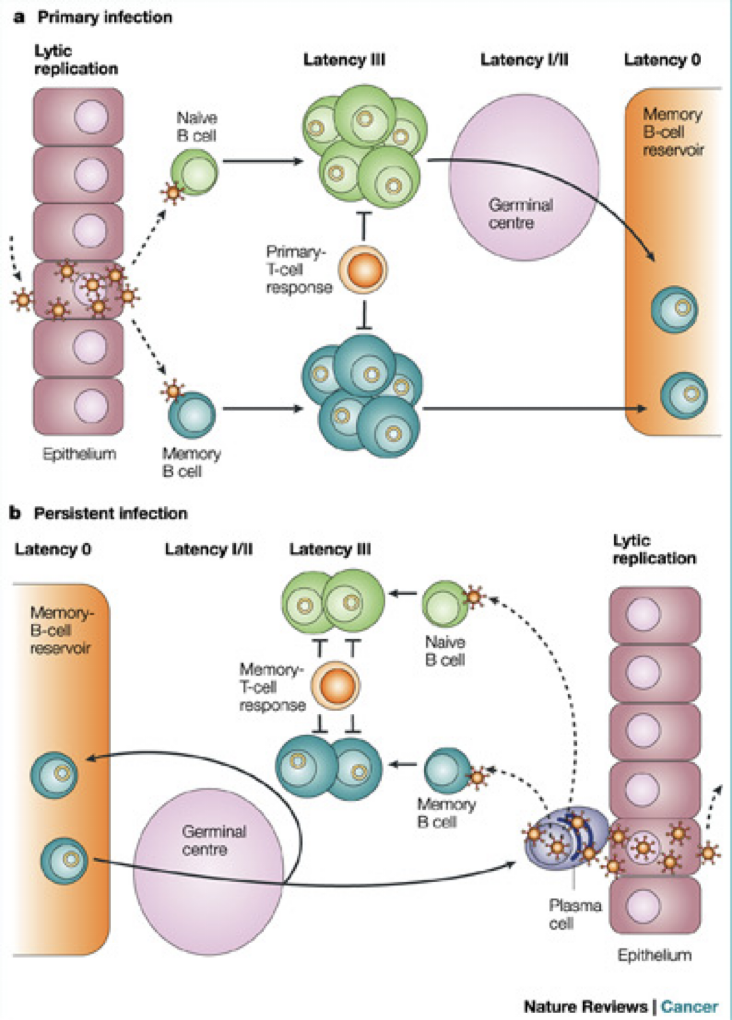
Virus latent infection:
The full genome is retained, but its expression is dramatically restricted, such that few viral antigens and no viral particles are produced.
To qualify as latency, this cryptic form of infection must display two additional properties: persistence and reversibility. Reversibility – i.e. the capacity of the genome to, under the appropriate circumstances, reactivate full viral gene expression, with production of infectious progeny (so-called productive or lytic replication) - is the key requirement of latency.
Transformation of B lymphocytes and Epithelia cells into malignant cells by Epstein Barr virus (EBV)
Epithelia and B lymphocytes are transformed by EBV into malignant cells as a result of expression of EBV latency gene products.
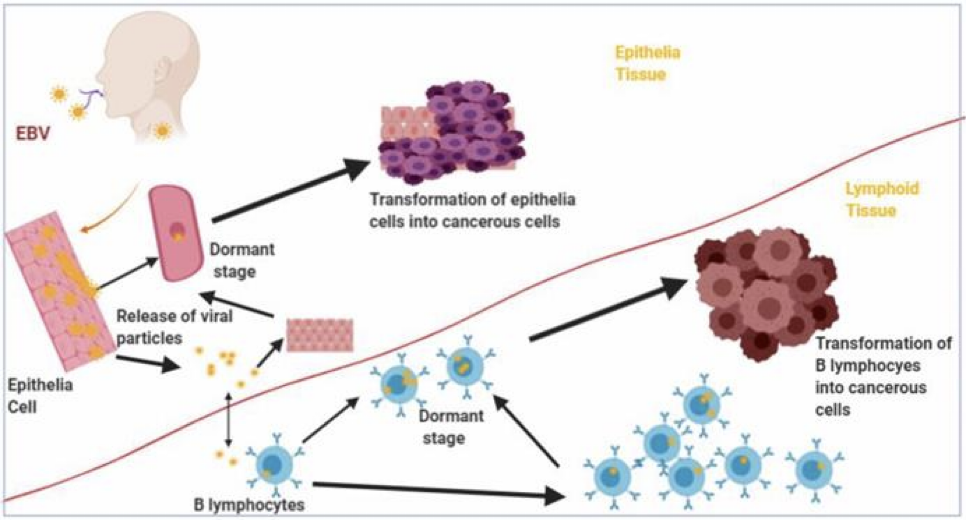
In vivo Interactions Between EBV and Host Cells
Biological Activities of EBV nuclear antigens (EBNAs) in tumorigensis
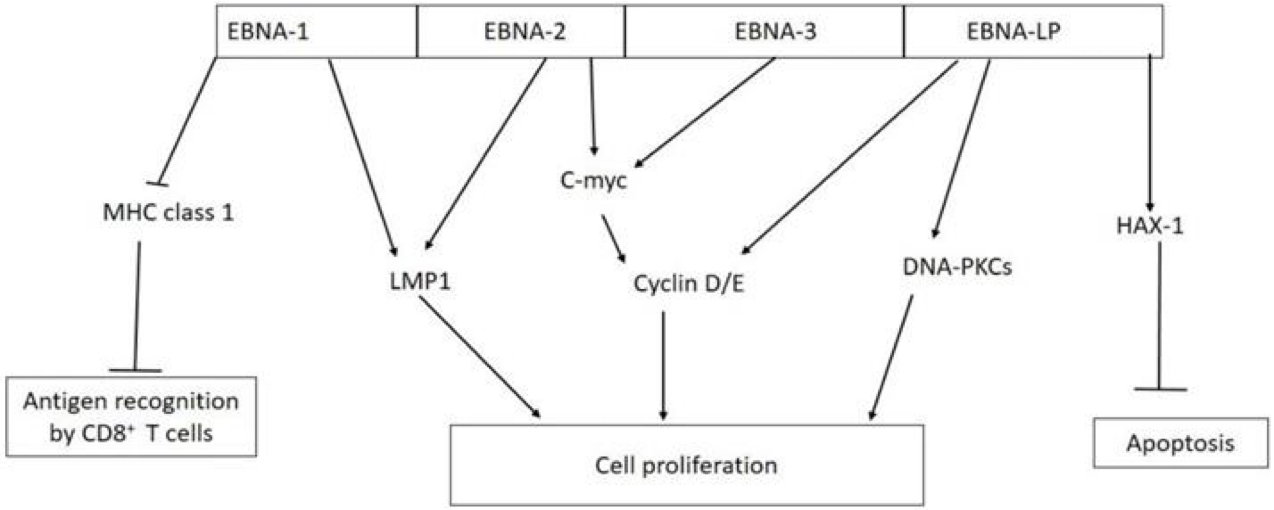
EBNA-1 inhibits antigen presentation by major histocompatibility complex (MHC) class I; EBNA-1 and EBNA-2 activate the expression of LMP-1; EBNA-2 and EBNA-3 interact with C-myc which constitutively activate cyclin D/E leading to unregulated cell proliferation; EBNA-LP can directly activate cyclin D/E and DNA-dependent protein kinase (DNA-PKCs) to promote cell proliferation; EBNA-LP promotes cell survival by interacting with antiapoptotic protein hematopoietic cell-specific protein 1 (HS-1)-associated protein X-1 (HAX-1).
Biological activities of latent membrane protein-1 (LMP-1) in tumorigenesis
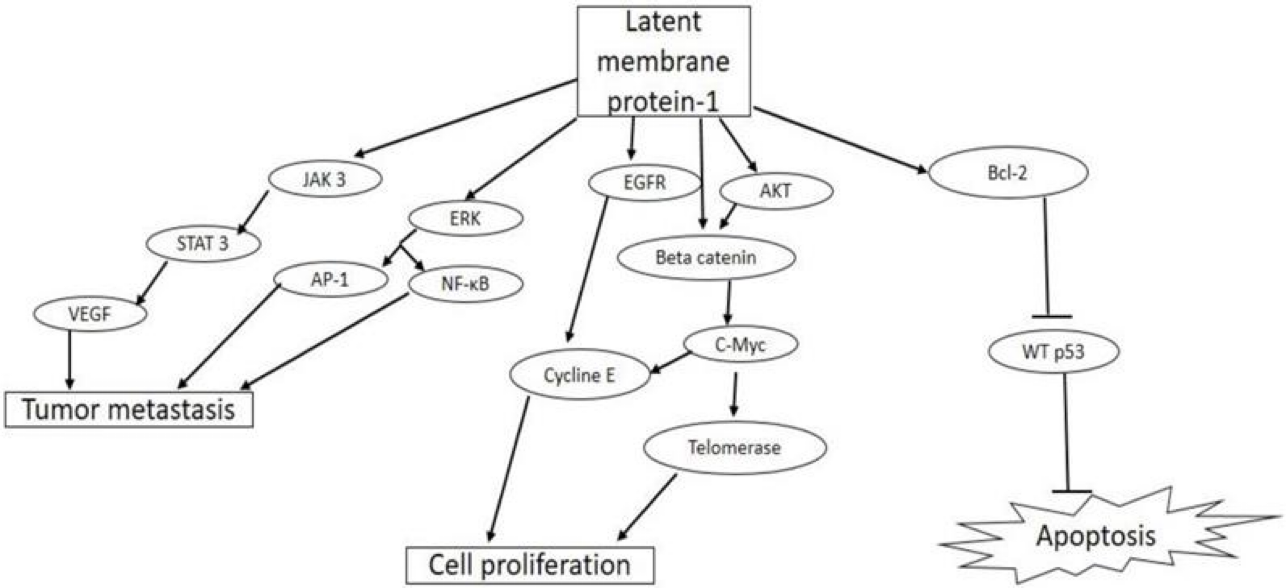
EBV LMP-1 activates cellular pathways that lead to tumor invasiveness and metastasis, cell proliferation, and inhibition of apoptosis