Amino Acid Metabolism and Pentose Phosphate Pathway
Amino Acids as Neurotransmitters and Modulators
Many amino acids and their derivatives (e.g., noradrenaline, dopamine) act as neurotransmitters or modulators in the brain.
Peptides also play a significant role in brain signaling.
Signaling in the brain is mediated by amino acids or their derivatives.
Example: Glutamate Neurotransmission and D-Serine
Rare disease example: mutations in a protein associated with glutamatergic neurotransmission.
Glutamate is released by one neuron and received by an adjacent neuron through glutamate receptors.
D-serine acts as a modulator of the glutamate receptor.
D-serine is produced in astrocytes from L-serine, which crosses from blood vessels into the brain.
L-serine is converted into D-serine in astrocytes, released, and diffuses into the synapse to improve neurotransmission.
Oral treatment with L-serine can improve motor function and quality of life in patients with impaired neurotransmission due to the mutation.
L-serine normalized EEG patterns and reduced seizure frequency in some affected children.
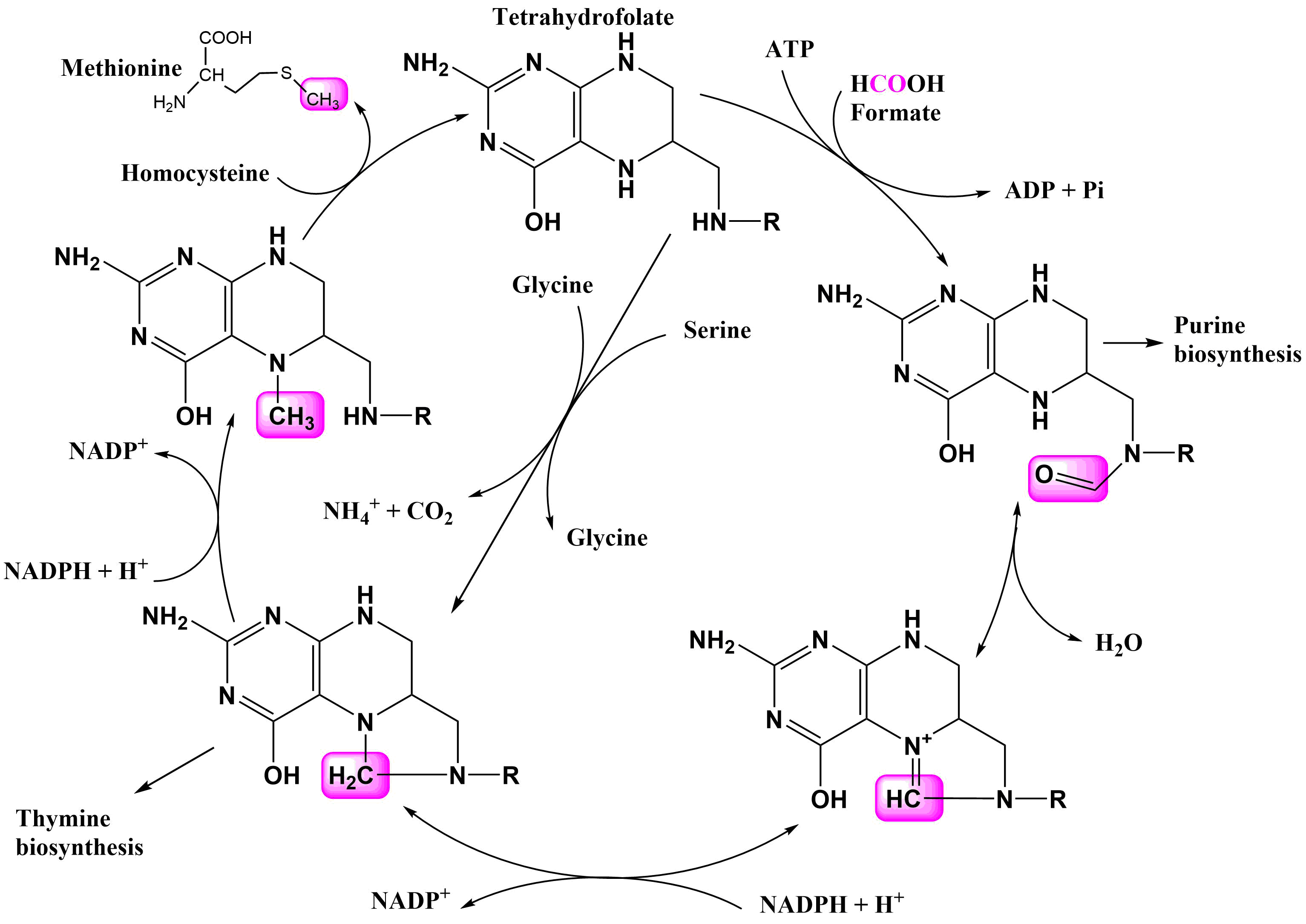
Methyl Groups in Metabolism
Methyl groups are present in various molecules like creatine, adrenaline, thymidine, and choline.
THF (tetrahydrofolate) transfers methyl groups to other compounds.
it contains 2xN which can temporarily hold a methyl group in different oxidation states before transferring it.
Source of Methyl Groups
Methyl groups come from amino acids, either glycine or serine (converted to glycine).
Glycine is broken down into ammonia, carbon dioxide, and a methyl group.
The methyl group is initially a methylene group when wedged between the nitrogens.
The oxidation state of the methyl group can be changed within the tetrahydrofolate cycle using NADPH.
Tetrahydrofolate Cycle
Methylene tetrahydrofolate can be reduced to methyl tetrahydrofolate with NADPH.
Methyl tetrahydrofolate is used to methylate DNA bases, such as thymidine.
Methylation of DNA is important for epigenetic modulation and silencing of genes. (turning some genes on or off)
The methyl group is transferred to methionine, which then forms S-adenosyl methionine (SAM), the molecule that methylates DNA.
Methylene group is also used for thymine biosynthesis
Formyl tetrahydrofolate, another oxidation state, is used for purine biosynthesis.
Formate can be directly loaded onto tetrahydrofolate to generate formate tetrahydrofolate.
The reaction converting methyl tetrahydrofolate to methionine is irreversible.
The cycle involves cycling methyl groups through different oxidation states (methylnyl, methylene, methyl, formyl).
C1 compounds (methyl groups in different oxidation states) are used for various chemical reactions.
NADPH is required to go through the different oxidation states.
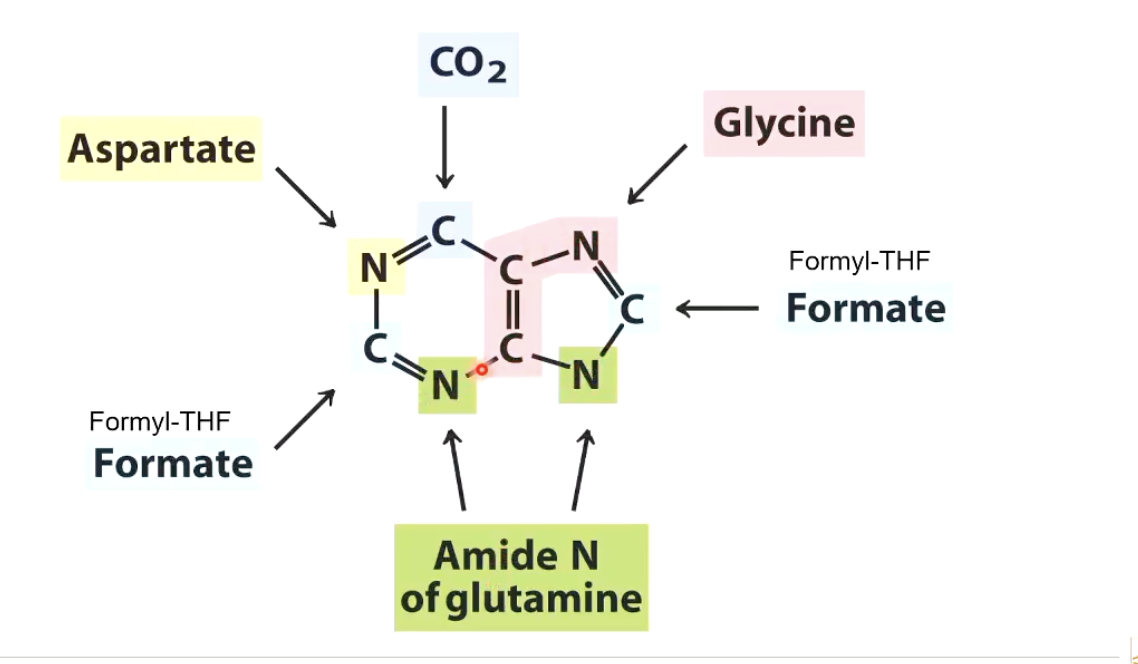
Purine Biosynthesis
Formyl tetrahydrofolate is used to form carbon atoms in purine bases.
Carbon dioxide contributes one carbon atom.
Aspartate, glycine, and glutamine contribute other parts of the purine base.
Medical Applications of Tetrahydrofolate
Tetrahydrofolate is essential for nucleobase synthesis, which is crucial during embryogenesis.
During pregnancy, particularly when neural cord closes over the spine, high demand for DNA and RNA biosynthesis makes tetrahydrofolate supplementation important.
Insufficient tetrahydrofolate can lead to spina bifida.
Tetrahydrofolate is also needed for red blood cell production.
Deficiency in vitamin B12 can cause similar problems as tetrahydrofolate deficiency because vitamin B12 is needed in the cycle.
Key Concepts of Amino Acid Metabolism
Transaminase reaction: its importance, mechanism, and what it achieves.
Glucogenic and ketogenic amino acids.
Phenylketonuria.
Urea cycle: how amino groups/ammonia enter, involved molecules, and reactions.
Role of tetrahydrofolate in metabolism.
Creatine acts as a high-energy bond storage compound.
Methyl groups are critical for DNA biosynthesis.
Tetrahydrofolate, along with vitamin B12, is involved in C1 compound reactions.
Glycine and serine are the main donors for generating methyl groups.
Introduction to the Pentose Phosphate Pathway
Pentose phosphate pathway is a key metabolic pathway.
Glycolysis converts glucose to pyruvate, which enters the TCA cycle, producing NADH.
Beta oxidation of fatty acids also produces NADH and acetyl CoA for the TCA cycle.
The pentose phosphate pathway primarily produces NADPH.
NADPH vs. NADH
NADPH and NADH are structurally similar, differing only by a phosphate group.
The phosphate group allows enzymes to differentiate between NADPH and NADH.
NADPH is used for biosynthetic processes, specifically for reduction by adding electrons.
NADH is used for oxidation and the respiratory chain.
The ratio of NADH to NAD is poised toward the oxidized state (NAD), while the ratio of NADPH to NADP is poised toward the reduced state (NADPH).
Uses of NADPH
NADPH is used for fatty acid synthesis, lipid synthesis, cholesterol synthesis, and some amino acid biosynthesis.
NADPH provides protection against oxygen radicals.
Pentose Phosphate Pathway Overview
In the oxidative phase, glucose-6-phosphate is converted to 6-phosphogluconolactone, producing NADPH.
6-phosphogluconolactone is further processed, generating another NADPH and ribulose-5-phosphate (a five-carbon sugar).
One carbon dioxide molecule is lost during this process.
The non-oxidative phase involves reshuffling reactions to generate molecules with three, four, six, and seven carbons.
Glyceraldehyde-3-phosphate and fructose-6-phosphate can rejoin the glycolysis pathway.
Alternatively, ribose-5-phosphate can be produced for DNA and RNA biosynthesis.
revision
Transaminase reaction: importance, mechanism, and what it achieves.
Glucogenic and ketogenic amino acids.
Phenylketonuria.
Urea cycle: how amino groups/ammonia enter, involved molecules, and reactions.
Role of tetrahydrofolate in metabolism.
Creatine's function as a high-energy bond storage compound.
Importance of methyl groups for DNA biosynthesis.
Involvement of tetrahydrofolate and vitamin B12 in C1 compound reactions.
Glycine and serine as the main donors for generating methyl groups.
Pentose phosphate pathway.
NADPH vs NADH.
Uses of NADPH.
Oxidative and non-oxidative phases of the pentose phosphate pathway.