Hess' Law
Hess’ Law
States the overall standard enthalpy change of a reaction is same regardless of whether the reactions takes place in one step or several steps
This allows you to break a reaction into steps and the enthalpy changes to find total enthalpy change for an overall reactions
Example
Calculate enthalpy change for the reaction - 2C + 2H + O2 → CH3COOH
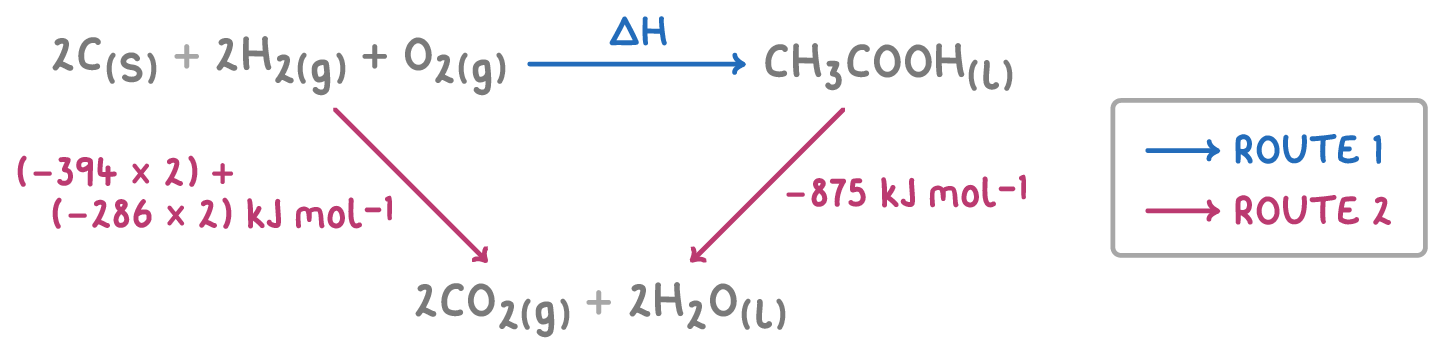
∆H =