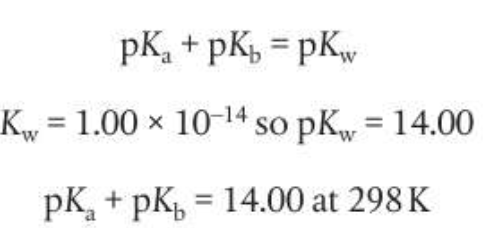Proton Transfer Reactions
Overview:
Bronsted-Lowry acids and bases
pH scale
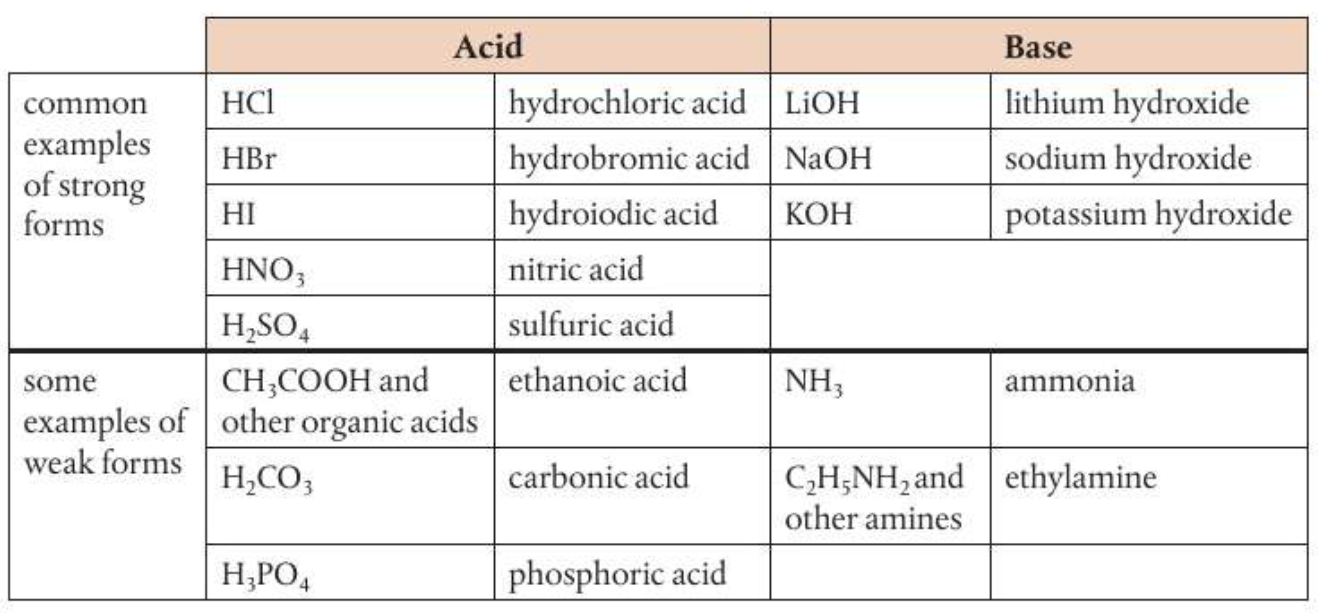
Strong and weak acids and bases
Neutralization reactions
Bronsted-Lowry acids and bases:
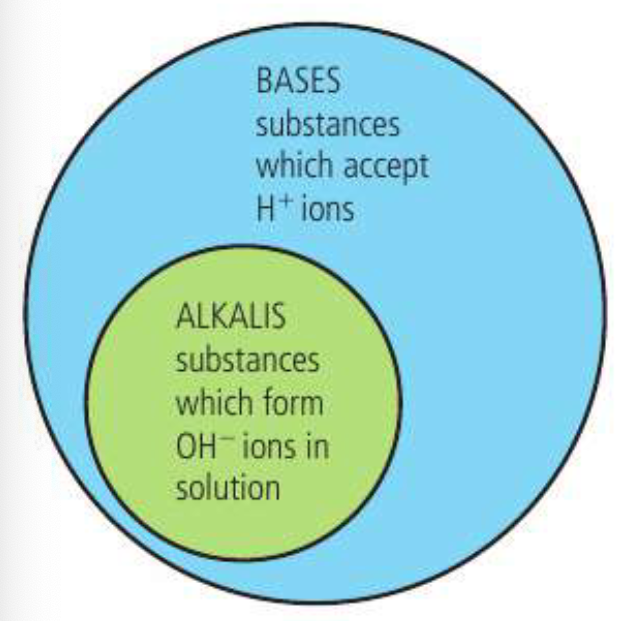
The Brownsted-Lowry theory describes acids and bases in terms of proton transfer. It was initially proposed that oxygen was the universal acidfying principle, and that an acid was composed of oxygen and a none-metal. This was disproven by the existence of HCl. Then Arrhenius suggested that an acid could be defines as a substance that dissociates in water to form H+ ions and anions, while a base dissociates to form OH- and cations. Also recognizing that hydrogen and hydroxide form water and the cations and anions could form a salt. The Bronsted-Lowry theory is a similar bur broader theory that says acids donate H+ while bases accept H+.
Reacting species in Bronsted-Lowry acid-base reactions form conjugate pairs, consider:
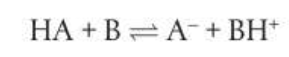
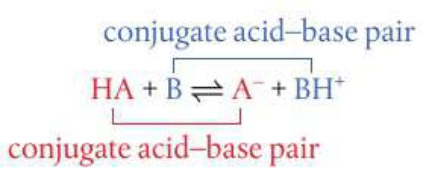
In a conjugate pair, the acid always has one more proton than its conjugate base. To form a conjugate base from an acid remove one H+ and to form a conjugate acid from its base, add one H+. Many of the polyatomic ions can act as Bronsted-Lowry bases by accepting H+ and forming Bronsted-Lowry acids with the exception of NH4
Amphiprotic species can act as a Bronsted-Lowry acid or base. Water for example, as well as HCO3:
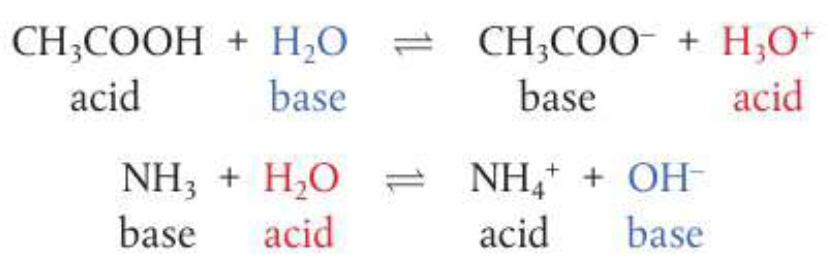
Therefore they can both dissociate and release H+ to be an acid and accept H+ to be a base, meaning they will have a lone pair of electrons.
Moving right across a period the oxides transition from basic metal oxides to amphoteric oxides. Basic metal oxides react with acid to form salt and water. Amphoteric oxides may react with an acid or a base to form salt and water. Non-metal oxides form weak salts in solution. Nitrogen and sulfur form weak oxides known as NOx and SOx, SO2 reacts with moisture in the air to form weak acids, which can form stronger acids which lowers the pH to cause acid rain.
The pH scale:
pH is a log expression of [H+]. The equations are as follows:
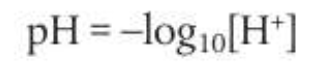
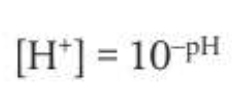
Features of the pH scale:
pH numbers are positive and have no units
Fall within the range pH 0-14
pH is inversely related to [H+], so stronger and more concentrated acids will have a lower pH, whereas weaker and more dilute acids will have a higher pH
A change of one pH unit is 10x the conc, eg

The inverse relationship can be illustrated by this graph:
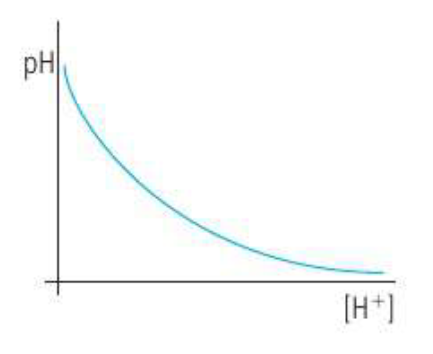
pH can be measured using a universal indicator paper or solution, or using a pH meter.
From pH equations, we can calculate the pH value given a concentration or the concentration given the pH. Though the pH scale is a measure of [H+], it can also be used to measure the alkalinity of a solution since there is an inverse relationship between [H+] and [OH-] concentrations. The sum of [H+] and [OH-] is 1 and the sum of pH and pOH is 14.

Majority of acid base reactions involve ionization in aqueous solution therefore the role of water must be considered in more detail. Water only ionizes a little bit so the equilibrium expression can be written as:
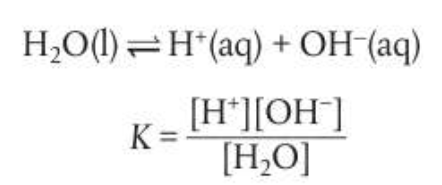
**ion products/water
The concentration of water is a constant, creating a modfied equilibrium constant known as the ionic product constant of water, Kw
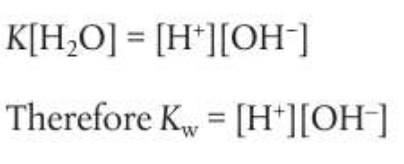
At 298K Kw= 1.00 × 10-14
Meaning [H+]=1.00 × 10-7 and pH = 7.00
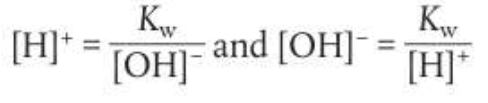
However Kw is temperature dependent, as an equilibrium constant. The reaction for the dissociation of water is endothermic as it requires bond breaking. So,
An increase in temperature will shift the equilibrium to the right which will increase the value of Kw and pH will decrease (acidic)
A decrease in temperature will shift the equilibrium to the left which will decrease the value of Kw and pH will increase (basic)
Strong and weak acids and bases:
Strong and weak acids and bases differ by extent of ionization.
Acids: if an acid dissociates fully it is a strong acid, in this case the reverse reaction is negligible so not equilibrium sign. If an acid sissociates paertially it is a weak acid, therfore it needs an equilibrium sign in the dissociation reaction. A strong acid forms a weak conjugate base. Weak acids are poor proton donors and therefore form strong conjugate bases.
Bases: if a base ionized fully it is a strong base, if it ionizes only partially it is a weak base. Similarly, a strong base forms a weak conjugate acid and a weak base forms a strong conjugate acid.
*Do not confuse acidic/basic strength with concentration
Strong and weak acids additionally differ by their equilibrium constants. At equilibrium strong acids have a high concentration of product (ionized form) and therefore hae large equilibrium constants. Weak acids have a lower concentration of product at equilibrium and therfore hae smaller equilibrium constants. The strength of dissociation is dependent on the bond that has to be broken to release H+, considering hydrogen halides, the halogen atom increases in size going down group 17 which increases the length of the bond meaning theyare weaker and require less energy to break therefore acid strength increases down the group.
To distinguish between strong and weak acids and bases:
strong acids and bases have a higher ion concentration making them more conductive than their weaker counterparts, this can be measured using the conductivity setting on a pH meter
Rate of reaction is increases with stronger acids
pH scale can be used directly to compare strengths of acids of equal concentration which can be determined using an indicator or pH meter
Neutralization Reactions:
In a neutralization reaction, acids react with bases to form water and salt (ionic compound). A salt is derived from a parent acid and a parent base, for example NaCl comes from the acid HCl and the base NaOH. These salts may then dissociate, therfore the addition of salt may impact pH depending on the strength of the parent acid and base. NaCl will not impact pH because its parent acid and base are both strong, however if one was strong and the other was weak, the dissociation of that salt would alter the pH of a solution in favour of its strongest parent.
Metal oxides and hydroxides are based which react with acids to form salt and water.
Metal carbonates and hydrogen carbonates react with acids to produce salt, water, and carbon dioxide. A reaction which involves a gas being given off wil visibly produce bubbles, this is known and effervescence.
When acids react with reactive metals they also form salts when the hydrogen from the acid is replaced by the metal. In these reaction there is no transfer of a proton and the hydrogen is just released as gas. Hydorgen ions because electrically neutral by accepting an electron from the metal, this is known as a redox reaction where the metal is oxidized and the hydrogen is reduced.
pH curves:
pH curves are generated by an acid base titration where controlled volumes of a reactant are added gradually from a burette to a fixed volume of a second reactant. The reaction in the flask takes place until the equivalence point is reached where they exactly neutralize each other. A pH meter or indicator is used to detect this point. In most titrations a big jump in pH occurs at the equivalence point AKA the point of inflection.
For a strong acid and strong base: equivalence point is pH =7
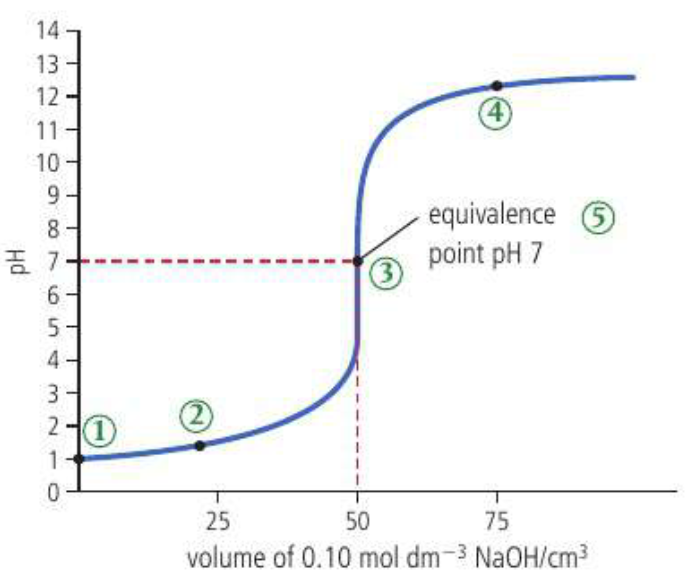
*for a weak acid and strong base the equivalence point is >7 and for a strong acid and weak base the equialence point is <7
Dissociation constants express the strength of weak acids and bases. They can be represented as equilibrium expressions with their own equilibrium constant. The constant of an acid is Ka and the constant of a base is Kb


These values allow us to calculate ion concentrations present at equilibrium, meaning we can calculate the pH and pOH values as well. Remember that the given concentration of an acid or base is its initial concentration (before dissociation). The pH or pOH refers to the concentration of ions at equilibrium. So to find dissociation constants the concentration values must be at equilibrium for reactants and products. When dissociation is small, the constantants are very low and therefore initial concentrations are approximately equal to concentrations at equilibrium.
Steps to solve for dissociation constant:
write out dissociation eqn with water
determine change in concentration of products by using given pH, for acid convert to conc using pH, for base find pOH from pH then convert to conc
plug into respective equation
* may be asked to calculate the pH and pOH from a given K value, in this case change the conc by x and plug into eqn, then use log to convert to pH
pKa and pKb are log expressions of the dissociation constants:

Relationship between conjugate acid-base pairs: