Regulated Exocytosis
4: Exocytosis is the process of vesicle fusion with the plasma membrane whereas secretion is the release of soluble vesicular content from the cell following exocytosis. Exocytosis is also important in delivering membrane proteins to the plasma membrane.
5: There are two different pathways of exocytosis - constitutive and regulated.
Constitutive exocytosis occurs constantly and is where vesicles bud from the Golgi and fuse directly with the membrane. Important for continually transporting newly synthesised lipids and proteins to the membrane, and secretion of molecules such as ECM components. Found in all eukaryotic cell types.
Regulated exocytosis occurs in many different specialised cells. Vesicles are retained in the cytoplasm until a signal is received, usually increase in cytoplasmic calcium. The signal promotes vesicle fusion with the membrane.
Similar proteins involved in both pathways.
6: Regulated exocytosis important for many different systems and cell types eg:
neurotransmitter release at synapse
insulin secretion in pancreatic cells
translocation of GLUT4 in response to insulin stimulation
7:
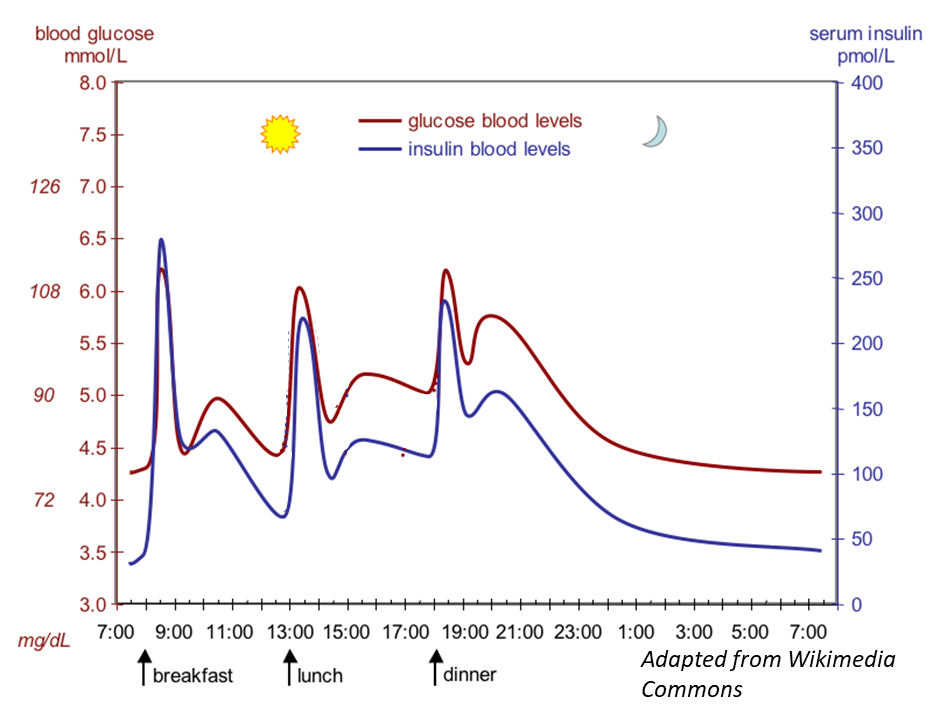
8: Insulin released from pancreatic beta cells in response to increased blood glucose. Beta cells found in pancreatic islets.
9: Insulin secretion occurs via regulated exocytosis in response to calcium ion influx:
increased glucose uptake and metabolism → ATP:ADP increase
ATP sensitive potassium channels close
membrane depolarised
voltage gated calcium channels open
increased cytosolic calcium → exocytosis of insulin granules (vesicles containing insulin)
insulin secreted
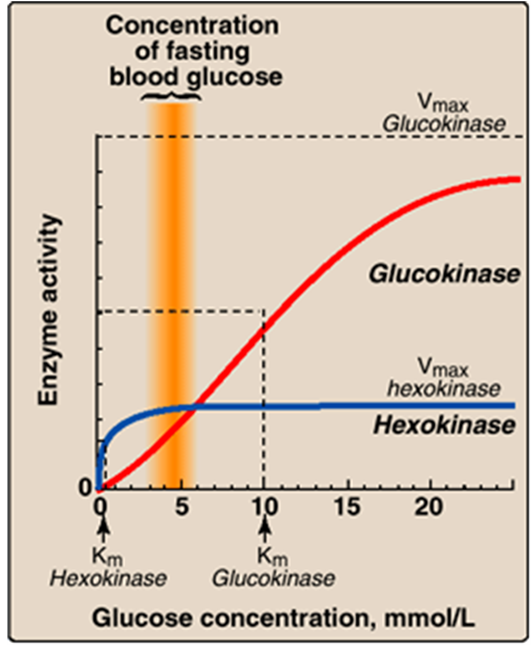
10: Glucose phosphorylation → glucose-6-phosphate traps glucose in cell committing it to further metabolism. Catalysed by hexokinase and glucokinase.
Hexokinase ubiquitously expressed (expressed in all cells). Works at full capacity at low glucose conc as it has high affinity for glucose ensuring cells have sufficient glucose metabolism even when BGC low
Glucokinase (mainly expressed in beta cells) activity varies within physiological range of BGC as it has lower affinity for glucose than hexokinase. When glucose enters cell there is proportional increase in glucose-6-phosphate formation and ATP. Therefore insulin release directly correlated with BGC.
11: Insulin secretion from beta cells occurs in a biphasic manner with a rapid first phase of insulin secretion followed by a slower second phase.
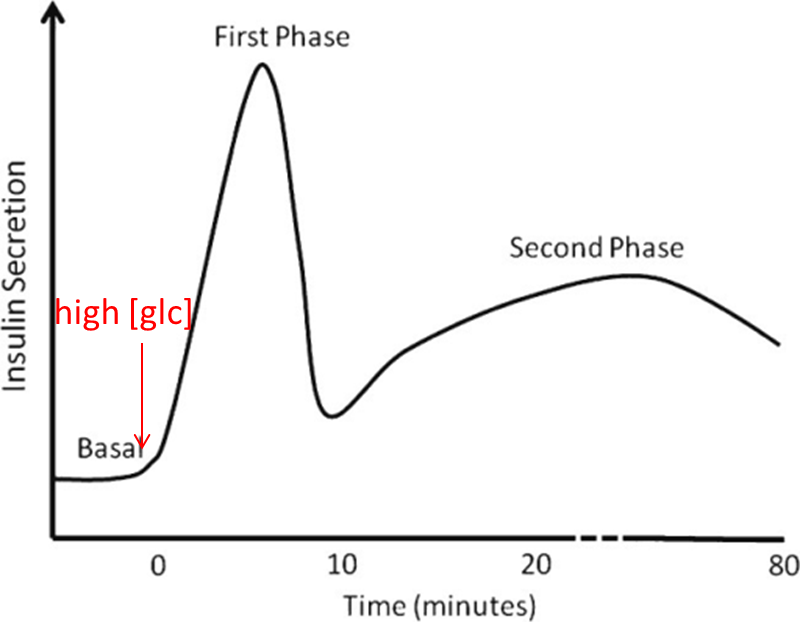
12: Biphasic insulin secretion is thought to reflect distinct insulin granule pools in cells. First phase insulin secretion occurs in approx 5 minutes and is released from granules docked at membrane - readily releasable. Second phase requires recruitment of cytosolic pool - reserve pool. Many different proteins regulate exocytosis of insulin granules.
13: Exocytosis mediated by SNARE proteins including vSNAREs eg synaptobrevin (aka VAMP) which forms complex with tSNAREs in membrane eg SNAP25 and Syntaxin. Formation of these three SNARE proteins drives fusion of vesicle with membrane.
15: Synaptotagmin is a calcium binding protein present on insulin granules. Its calcium bound form interacts with SNARE protein and membrane phospholipids to enhance probability of membrane fusion occurring This is how increased cytosolic calcium leads to increased insulin secretion. This is known as calcium-secretion coupling.
16: Insulin acts on many body cells to reduce blood glucose, its major targets being the liver, adipocytes and skeletal muscle. Insulin activates a different regulated exocytosis pathway in adipocytes and skeletal muscle. It stimulates exocytosis of vesicles containing GLUT4 which is a pathway that doesn’t involve calcium.
17: Insulin binding → receptor auto-phosphorylation (tyrosine kinase region). Phosphorylated residues act as binding sites for insulin receptor substrate proteins (IRS). IR phosphorylates 4 tyrosine residues in IRS proteins. Phosphoinositide 3-kinase binds phosphorylated residues on IRS proteins then converts membrane PIP2 to PIP3. Binding PIP3 causes recruitment of PDK1’s PIP3 dependent kinase, activating PDK1 which phosphorylates and activates kinases eg PKB (aka Akt). Activated PKB phosphorylates proteins on GLUT4 vesicles to promote exocytosis
18: GLUT4 vesicle exocytosis requires interactions between SNARE proteins: VAMP on vesicle and syntaxin and SNAP23 on plasma membrane. Instead of contents being released into extracellular environment receptor is inserted
19: vesicle fuses with plasma membrane
20: vesicle fusion allows insertion of GLUT4 receptors into membrane. Glucose enters cells via GLUT4
21:
22: When not needed, GLUT4 is contained in storage vesicles in the cell. Protein AS160 functions to retain vesicles in cell. Activated PKB inactivates AS160 by phosphorylating it so that GLUT4 vesicles can fuse with the membrane.
23: Type 1 diabetes is a chronic autoimmune condition where destruction of beta cells results in insulin deficiency and hyperglycaemia. Occurs in genetically susceptible individuals, likely due to environmental trigger eg viral infection which incorrectly activates immune response
24: Hyperglycaemia occurs due to loss of insulin-stimulated glucose uptake into target cells and loss of insulin-mediated repression of gluconeogenesis and glycogen breakdown in the liver. People with Type 1 diabetes therefore need to take insulin injections for the remainder of their lives
25: Type 2 diabetes is linked with insulin resistance. It’s characterised by high BGC due to insulin resistant target tissues (adipocytes and skeletal muscle) and insufficient production and/or secretion of insulin (beta cell dysfunction. Pancreas may respond by producing more insulin but this causes too much stress to pancreas, leading to pancreatic dysfunction. Diagnosed later in life but onset is getting increasingly earlier. Accounts for 90 to 95% of diabetes cases and prevalence rising rapidly due to increased obesity, lack of exercise, poor diet and ageing population
26: Strong link between obesity and insulin resistance/type 2 diabetes although it’s not clearly defined how excess body fat causes insulin resistance/type 2 diabetes. Many different mechanisms implicated in development of insulin resistance via disruption of insulin signalling.
27: Type 2 diabetes treated by lifestyle changes eg healthier diet and regular exercise to control symptoms. Progressive disease therefore oral medication may eventually be required eg metformin which blocks production of glucose by liver - inhibits gluconeogenesis. May also take sulfonylureas which bind gated ATP potassium channel, closing it causing membrane depolarisation and calcium channel activation triggering insulin vesicle exocytosis and insulin secretion. If glucose control not adequate insulin may also be prescribed