Lecture 9: Glycemic Responses and Glycogenesis
Learning Outcomes:
Dynamics of post-prandial glucose disposal and the key hormones involved
Understand the post-prandial glucose responses for subjects with glucose intolerance, insulin resistance and diabetes
Understand what glycemic index is, how it is measured and how it is clinically useful
Appreciate the role of different glucose transporters in different tissues and how these function
Review the chemical structure of glycogen and the chemical strategy for its synthesis
Understand how increased glycogen synthesis stimulates glycolysis
Explain the consequences of the different activities of hexokinase and glucokinase
Summarise the similarities and differences in glycogenesis in different tissues (liver vs muscle)
Building glycogen = glycogenisis
Glucose is toxic and can react with every protein in our body
No enzymes required
Glycation = no enzymes for glucose and protein reaction
Glycation is bad – leaves proteins sugar coated and impacts their function
The rate this occurs in proportional to blood glucose concentration
Even happens at 4-5mM – but out brain requires 4-5mM
Time of exposure is also important
Want to return blood glucose levels to normal asap after eating, or damage to proteins is at risk
Determining risk of diabetes:
Don’t consume food 8-12 hours, measure BG concentration – fasting blood glucose
HbA1c test – measures how many haemoglobin molecule in the blood are glycated (have glucose attached) – indicator of [blood glucose] over the last 3 months
Post-Prandial Glucose Disposal:
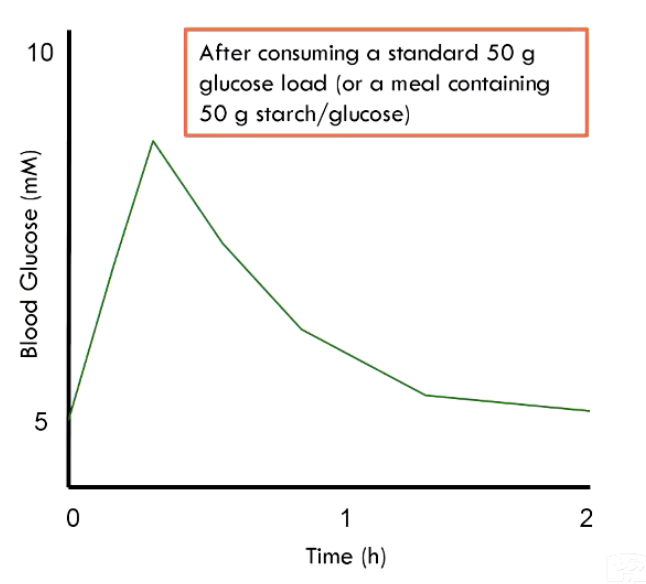
Oral glucose tolerance test
Patient consumes a standard glucose load
Monitor BG concentration over 2 hours
Accomplished by:
Insulin secretion
Liver sponging up lots of glucose
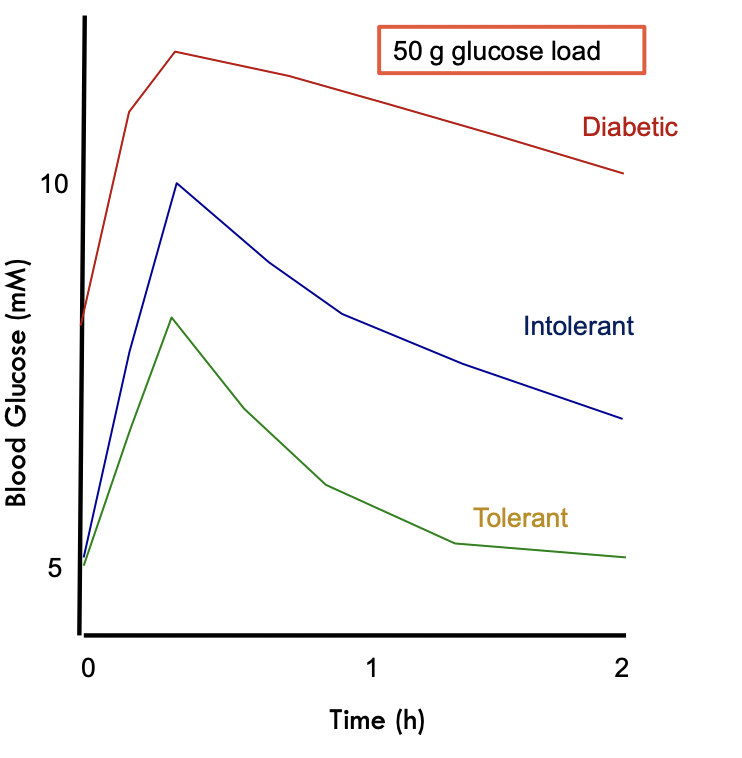
Glucose Tolerant – you are able to return to fasting blood glucose by 2 hours after consumption
Glucose Intolerant – Normal fasting blood glucose but slow clearance; doesn’t return to fasting concentration within 2 hours
Diabetic – Fasting hyperglycaemia, doesn’t return to fasting concentration within 2 hours, relentless exposure to high [glucose]
The more glucose exposure, the more the damage
Insulin Production:
Released by the beta cells of the pancreas when [glucose] > 5mM
Small amount of insulin can also be released in response to amino acids
Each time a meal is consumed, insulin spiked
Insulin stimulates glucose uptake upregulating GLUT-4 recetptors in the muscle and adipose tissue
Cells won’t keep bringing in glucose if they aren’t doing anything with it – glycogenesis or lipogenesis
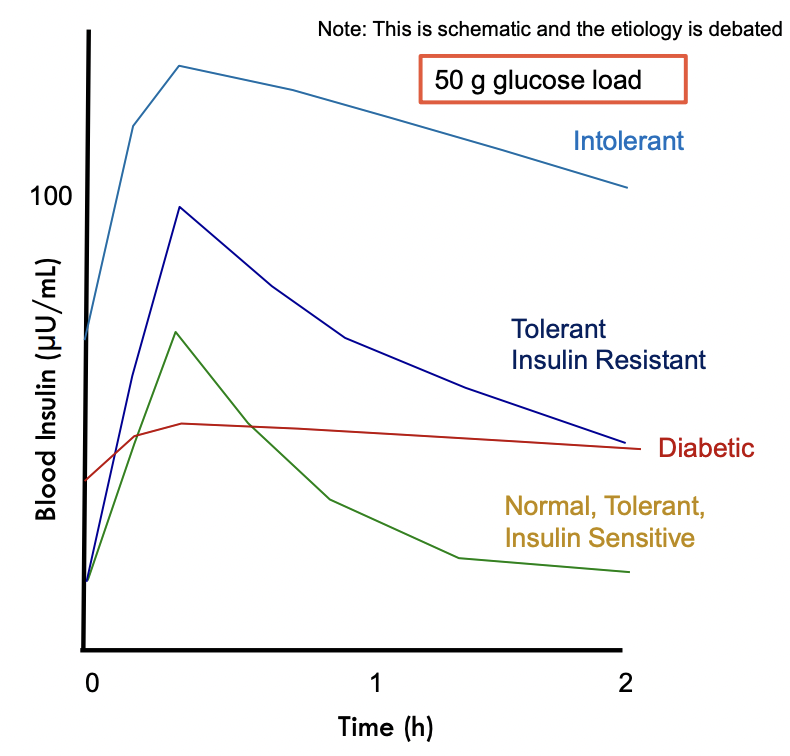
Insulin Resistance:
Normal, Tolerant, Insulin Sensitive: Insulin spikes in response to food intake and then lowers within 2 hours
Tolerant Insulin Resistant: Need to secrete more insulin to get rid of glucose
Intolerant (pre-diabetic): Secreting more insulin but not enought o overcome resistance, constant hyperinsulinemia
T2 Diabetes: Beta-cells exhausted, unable to respond dynamically, unable to maintain basal euglycemia (normal blood sugar)
Starch:
Main source of dietary carbohydrate
A polymer of glucose
Two major forms:
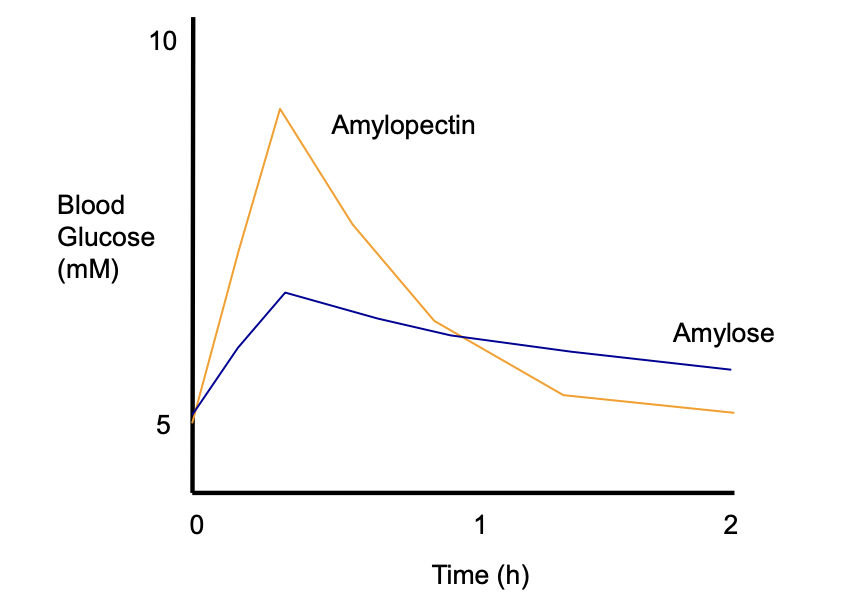
Amylose
Linear, forms helices
Difficult to digest – digestive enzymes can only act on one end of the chain
Difficult for amylases to penetrate
Makes itself all the way to the lower bowel before it is digested, causes Flatulence
Low GI foods
Amylopectin
Branched
Easy hydrolysis/digestion
Easy and quickly to digest
Lots of digestive enzymes can work on the many branches at the same time
Faster spike in [glucose]
Processed foods
High GI
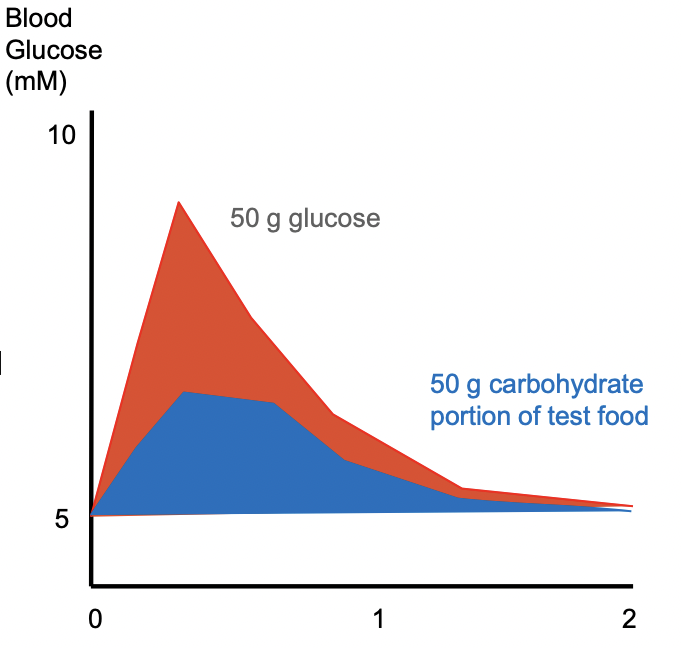
Glycemic Index:
Invented to try and help us chose foods that didn’t expose our body to very high [glucose] or great fluctuations – which are linked to the development of T2 diabetes
Describes post-prandial glucose response
Area under the test food curve divided by area under the reference curve
Reference food is usually 50g glucose
Test food given in an amount that will give 50g digestible carbohydrate
E.g. the amount of food that will provide 50g of digestible carbs – 2 apples
Measure ration of area under each curve = GI index (as a %)
GI of amylopectin foods (modern grains) have a high GI >80
GI of legumes <30
Notes:
Area under slowly absorbed may be the same as area under quickly absorbed
Should GI apply to non-starches?
Fructose can sweetened foods but your body doesn’t respond with insulin production when you consume it
Fructose is a non-glycemic sugar
Sucrose (table sugar) is only half glucose
Using sucrose to sweeten foods = only consuming half as much glucose
Dairy foods are low GI
Lactose is glucose and galactose
only half glucose
GI is reduced
e.g. yogurt is low GI because of the galactose but it will eventually be converted anyway
Low GI is considered healthier, generally
Spikes tend to predispose people to T2 diabetes rather than slow release
Claims of “slow burning energy”
Not supply driven – we burn energy at the rate we use it
Glucose Disposal:
Every cell has GLUT1 transported to transport blood glucose to your tissues
First blood in your body to look at the sugar you consume if in the hepatic portal vein (liver) – liver gets the first look at food
Liver has GLUT2 transporters – takes a lot of blood glucose out of the blood – insulin independet
GLUT2 are wide open doors (don’t need insulin)
Muscle – GLUT4
Strongly dependent on insulin
Very high capacity to get rid of glucose – muscle requiring energy to do things
Has more GLUT4
White adipose tissue – GLUT4
Strongly dependent on insulin
Used glucose to build fat and store it for later
We have lots of white adipose tissue
Need to do something with the blood glucose
High glucose levels inhibits hexokinase
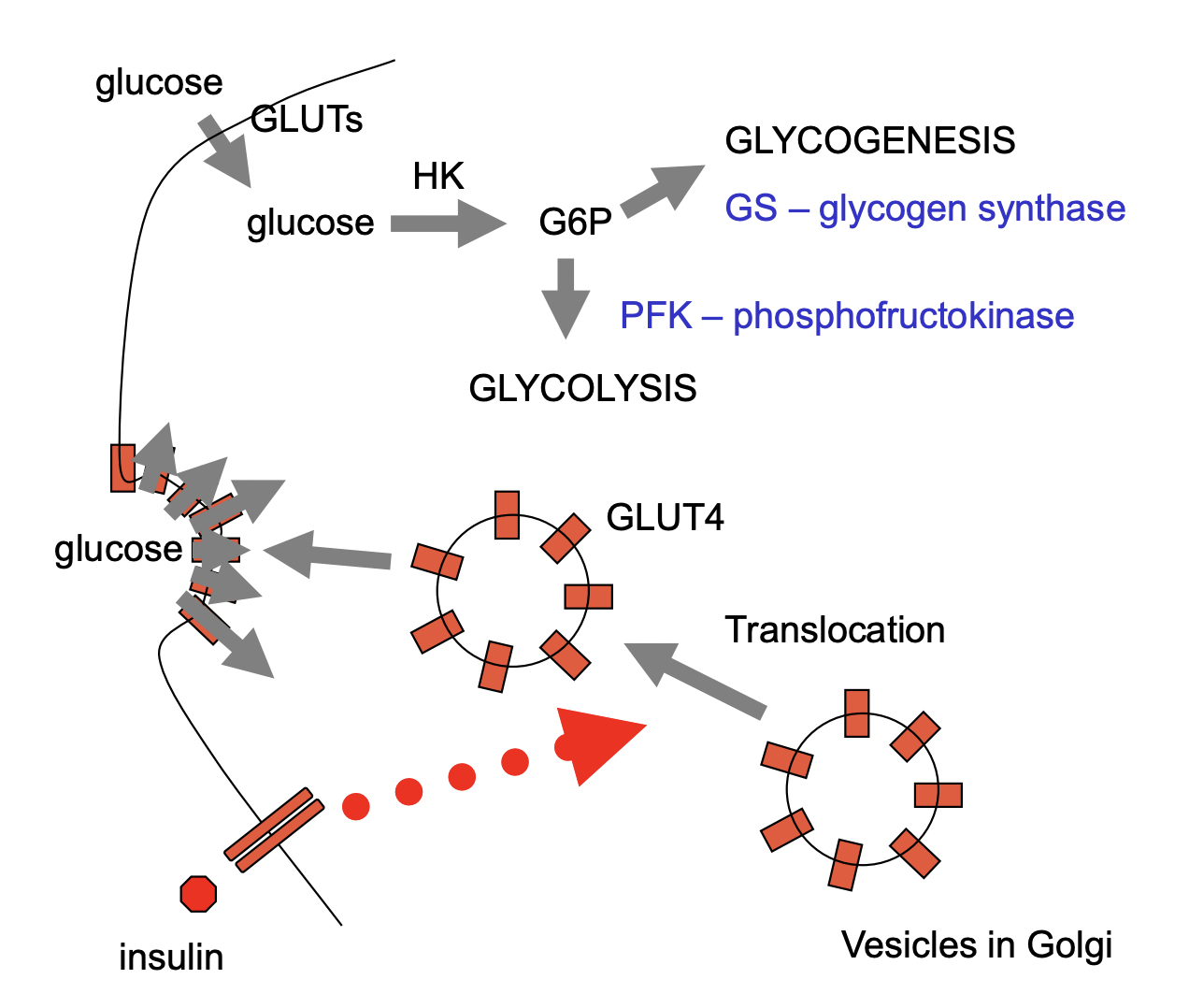
In Muscles:
Glycogen synthase is used to turn glucose 6 phosphate into glycogen – glycogenesis
GLUT4 live in Golgi apparatus and translocate to the membrane with insulin stimulation
Glucose in the Muscle:
Glucose comes in and it phosphorylated by hexokinase
Could go down glycolysis
Or it gets converted to glucose-1-phosphate via isomerisation reaction
UTP comes, gets rid of 2 phosphates (PP), becomes UMP, and attaches to glucose-1-phosphate to make UDP glucose (Activated Glucose)
This prepares it to add on to the end of the glycogen chains – the end specifically
UDP is left over and needs to be converted back to UTP to be used again
This uses ATP
UDP + ATP = UTP + ADP
Activate glucose by turning it into UDP glucose so it can be added onto a glycogen chain and that costs 1 ATP
Branching:
Need branching in glycogen or it will be entirely linear
Glycogen branching enzyme takes some glycogen chain, cuts it, and joins it to form a branch.
Glycogen Synthase:
Regulated by reversible phosphorylation
Has a kinase and a phosphatase that can regulate it
Insulin can stimulate phosphatase I that will remove the phosphate from glycogen synthase to make it active to add glucose to glycogen
Kinase removes adds the phosphate groups – makes it inactive
Insulin stimulated the building of new glycogen molecules
Phosphofructokinase
Rate limiting step of glycolysis
Not directly stimulated by insulin
Regulated allosterically
Especially by AMP which is stimulates by low energy charge
Stimulation of glycolysis by insulin creates an energy demand
Glycogenesis drops cellular ATP and increases ADP and AMD
This drop in energy stimulates PFK and glucose oxidation
The anabolic pathway requires and stimulates the catabolic pathway – coupling
Signals to store fuels also causes fuels to be burn
Liver Glucose Disposal
GLUT2 used to take up glucose
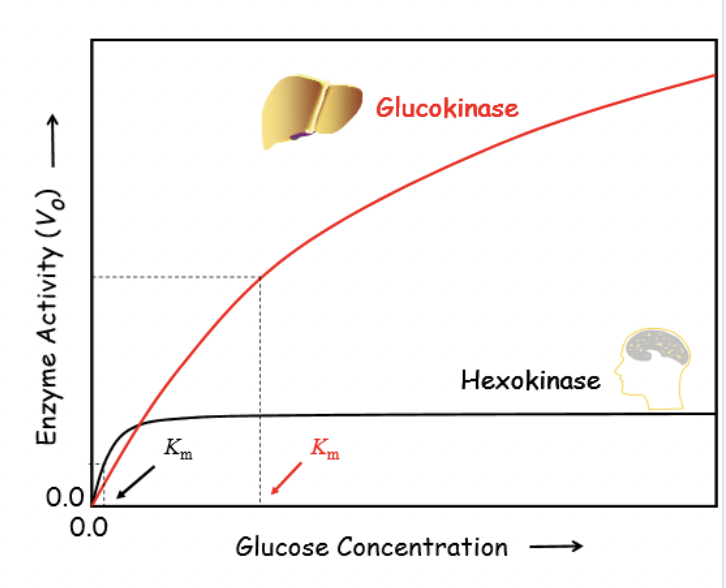
Glucokinase
Keeps trapping glucose-6-phosphate
Won’t be inhibited by glucose-6-phosphate build up
High Km (10mM) for glucose – not saturated by high levels of liver glucose
[GP6] rapidly increases as blood [glucose] rises
GP6 can stimulate inactive glycogen synthase in the liver
Don’t need insulin
In Liver | In Muscles |
|
|
Glucokinase (GK) | Hexokinase (HK) |
|
|
Glycogen synthase needs to see protein at the core (glycogenin) of the glycogen molecule to keep adding glucoses
Makes glycogen granules
Each glycogen is only 12 to 14 residues
2 ATPs are required for the incorporation of a glucose into the glycogen chain
G to G6P and UDP to UTP