Clinical Investigation Exam 2
~3 questions per lecture
Observational Studies [8 questions]
Recognize situations where an observational study design may be appropriate to answer a research question.
Limitations
cannot show causality — only associations
Used
when RCTs cannot be done
Why Used
provide preliminary data to inform the design and how to conduct a RCT when feasible
may have more generalizability than RCT
Identify key features that distinguish each of the different types of observational study designs from each other, and from interventional studies: cross sectional, case control, cohort- retrospective and prospective.
Cross sectional study
What:
prevalence study — examines relationships between disease (outcome) or drug (exposure) and other characteristics in a population at one point in time, cannot measure incidence of disease
Set Up: gather data at one time on both exposure and outcome
exposure and outcome present
exposure and no outcome
unexposed and outcome present
unexposed and no outcome
Use: study conditions that are frequent with long duration of expression
Case control study
What: compares people who have disease/outcome (cases) to those who do not (controls) with respect to variables of interest (causes or exposures)
Set Up
Use
Strengths
Weaknesses
Cohort study
What: incidence study measures outcome(s) in a population that didn’t start with outcome, relates hem to development of outcome over time
Retrospective or prospective — ALWAYS longitudinal
Set Up
Use
Strengths
Weaknesses
Identify where each type of observational study design sits in the evidence hierarchy pyramid.
Higher quality of evidence w/ lower risk of bias
systematic reviews and meta-analyses of RCT
RCT
cohort studies
case control studies
cross sectional studies, surveys
case reports
mechanistic studies
editorials, expert opinion
Lower quality of evidence w/ higher risk of bias
Identify advantages and disadvantages of each type of observational study design.
Cross Sectional
Advantages
quick, inexpensive, ethically safe
measures multiple exposures/outcomes at once
Good for hypothesis generation
Disadvantages
cannot establish causality or temporality
ONLY measures prevalence, not incidence
Prevalence = the amt of people with a disease or condition at a point in time
highly prone to bias (recall, observer, sampling, nonresponse)
unequally distributed confounders
NOT for rare or short duration diseases
Case-Control
Advantages
for rare diseases or long latency [effective]
faster and cheaper than cohort designs
study of multiple exposures for one outcome
Disadvantages
Bias (recall, selection)
difficult to select appropriate controls
only estimate odds ratios
odds of an event occurring in one group versus another, estimating means you don’t know the true risk or probability
Confounders may distort associations
Cohort (prospective)
advantages
temporal relationship can be established between exposure and outcome
Temporal relationship = exposure happens before the outcome, you must be exposed before it can cause an effect logically
measure incidence and risk
study multiple outcomes for one exposure
data collection more complete and standardized
Disadvantages
Time consuming, expensive
Bias (loss to follow up)
not for rare diseases
blinding difficulty
Cohort (retrospective)
Advantages
faster, less expensive than prospective
when data already exists its good
study of exposures that don’t happen anymore
Ex. Atomic bomb radiation exposure
Disadvantages
poor or incomplete data quality
prone to confounding and bias (selection)
cannot control for unmeasured variables
Recognize types of bias that may be present in observational studies.
Selection bias
What: systematic error in how groups are created or chosen, differ in prognosis or baseline factors
Which studies: Cohort Retrospective, Case-Control,
Recall Bias
What: participants with disease may remember past exposures differently
Which studies: Case-Control, Cross sectional
Observer Bias
What: investigator’s knowledge of exposure/outcome influences data collection or interpretation
Which studies: Cross Sectional
Sampling/ Nonresponse bias
What: Study sample not representative of population or certain groups less likely to respond
Which studies: Cross Sectional
Confounding [by indication]
What: indication for a treatment (not the treatment itself) is what causes the outcome
Which studies: Case-Control
Regression to the Mean/Hawthorne effect
What: extreme values tend to move toward the average upon repeat measurement/ behavior changes because subjects know they’re being observed
Which studies:
Recognize potential confounders that may be present in observational studies.
What is a confounder?
a variable associated with both the exposure and the outcome, but not on the casual pathway
Measured confounders
collected and adjusted for.
Examples: age, gender, comorbidities
Unmeasured confounders
unknown or unavailable variables
Residual confounding
leftover bias even after adjustment
Confounding by indication
reason for treatment is related to outcome
Choose appropriate strategies to minimize the effects of confounders in observational studies.
During study design
Restriction
limit study to subjects within one category of a confounder
Matching
pair cases and controls with similar confounding factors
Propensity score matching
statistical method to match exposed/unexposed subjects based on probability of receiving treatment
During Data Analysis
Multivariate regression
adjust for multiple confounders simultaneously
Purpose: controls for age, sex, comorbidities
Propensity score adjustment
uses predicted probability of exposure to balance groups
Purpose: mimics randomization statistically
Instrumental Variable Analysis
uses a variable associated with exposure but not with outcome to simulate randomization
Purpose: provider preference for a drug
Sensitivity analysis
tests robustness of results by varying assumptions
Purpose: assess impact of unmeasured confounders
Conceptual Frameworks
Known knowns: measured and adjusted
Known unknowns: known but unmeasured → listed as limitations
Unknown knowns: indirectly measured through proxies
Unknown unknowns: completely unrecognized residual confounders
Interventional Studies [3 questions]
Distinguish how interventional studies differ other types of study designs.
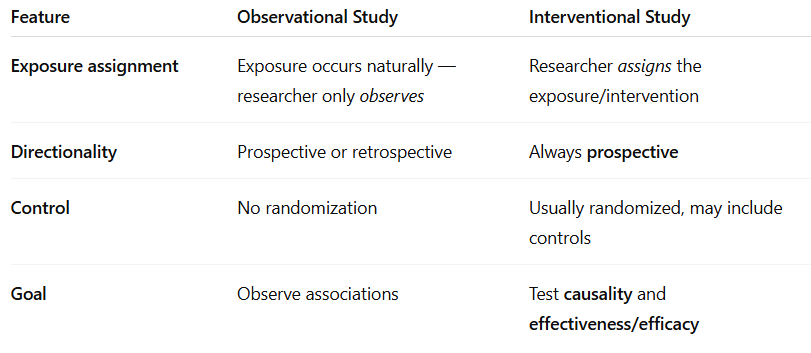
Key difference: interventional → investigator determines who receives intervention
Identify how superiority designs differ from non-inferiority intervention designs and the appropriate situations for each.

Choose an appropriate study design based on the research question and outcome of interest.
Is a new treatment effective compared to placebo?
superiority RCT
Is a new treatment as effective as standard care but more convenient or cheaper?
non-inferiority RCT
Is an intervention feasible or produces a signal for benefit?
non-randomized or pre-post study
Does a treatment have a temporary effect that can be reversed?
crossover study
Can a real-world intervention improve outcomes in practice?
pragmatic or cluster RCT
Are two interventions interacting or synergistic?
factorial design
Understand differences in how control groups are selected for each intervention design (e.g. RCT, Cross-over, pre-post, etc.)
Traditional RCT
Type: randomized to treatment vs control/placebo
GOLD STANDARD, minimize bias
Non-Randomized controlled trial
Type: assignment by investigator or patient choice
HIGH BIAS
Historical Control
Type: compare to data from prior patients or past standard
temporal and selection bias
Pre-Post (before-after)
same patients before and after intervention
regression to the mean and temporal confounding
Crossover design
each subject serves as their own control, separated by washout
efficient, sensitive to order and carryover effects
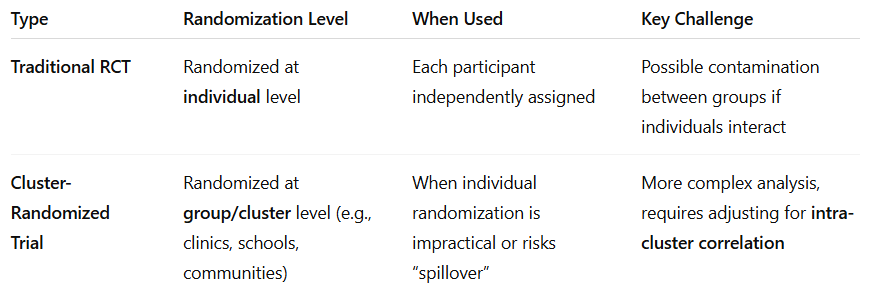
Cluster randomization
whole groups randomized
when individual randomization would contaminate other participants
Recognize the key features of a factorial design.
Purpose — evaluate two or more interventions at same time
Design — 2×2 randomization
Main question — can both treatments work independently or synergistically?
Interaction (effect modification) — occurs when effect of one intervention depends on presence/absence of other
Advantages — test multiple interventions with fewer subjects; examines interaction
Disadvantages — added complexity; risk of adverse effects from combos
Understand how randomization differs in traditional RCT vs cluster-randomized trial.
Identify key features that make a trial pragmatic.
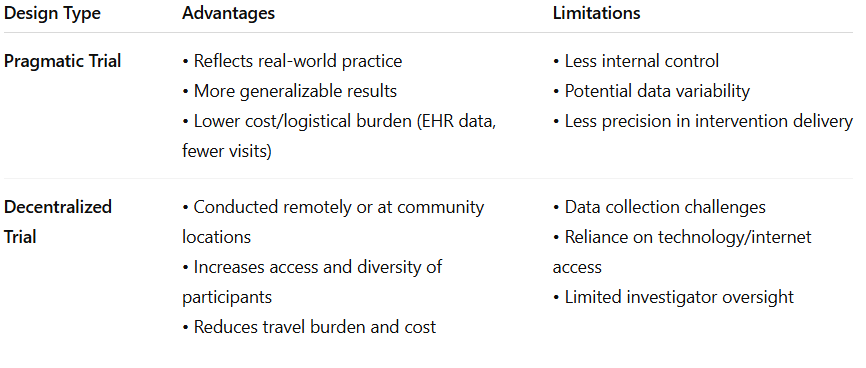
Purpose — real world effectiveness tested
Population — broad inclusion criteria; clinical practice patients
Intervention — implemented in routine care
Follow-Up and data collection — EHRs, registries, remote systems
Randomization — preserved to maintain internal validity
Strength — high external validity
Limitation — less control → more variability and potential bias
Recognize advantages and limitations of alternative study designs such as pragmatic and decentralized trails.
Randomized Control Trials: Superiority [8 questions]
Identify strengths and challenges/limitations of randomized trial designs.
Strengths
Gold standard for causality
balances known and unknown confounders
blinding minimizes bias in treatment and outcome
internal validity due to standardized protocols and SOPs
Challenges
expensive and time consuming
ethical barriers
efficacy does not mean effectiveness
volunteer bias, Hawthorne effect, regression to the mean
not great for rate outcomes or long-term endpoints
Define equipoise in clinical research.
The ethical and scientific state of genuine uncertainty about whether one treatment is better than another.
Trial is justified when there is true uncertainty about which option is superior
Purpose: participants are not knowingly deprived of effective therapy, maintains ethical balance in randomization
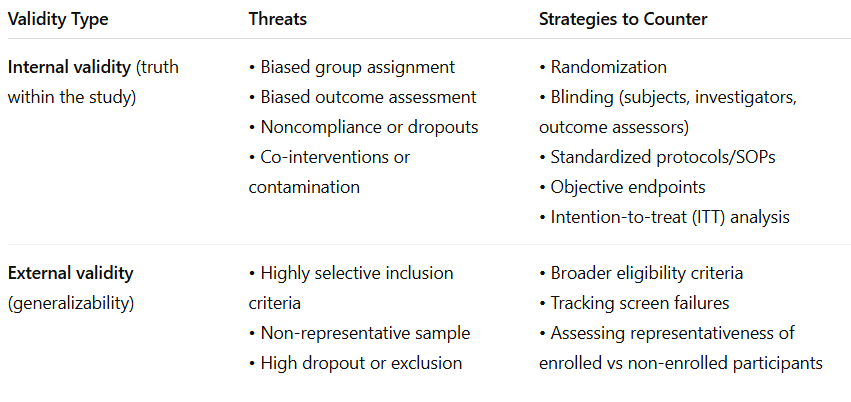
Identify threats to a RCTs internal and external validity, and strategies used to counter them.
Recognize when stratified/block randomization should be considered.
Use stratified/block randomization in:
small sample size
when prognostic factors influence outcomes strongly (age, sex, comorbidity)
Subgroup analyses are planned
Example: if age and sex affect outcomes, create strata (male/female; >65/>65) and randomize within each stratum to maintain balance
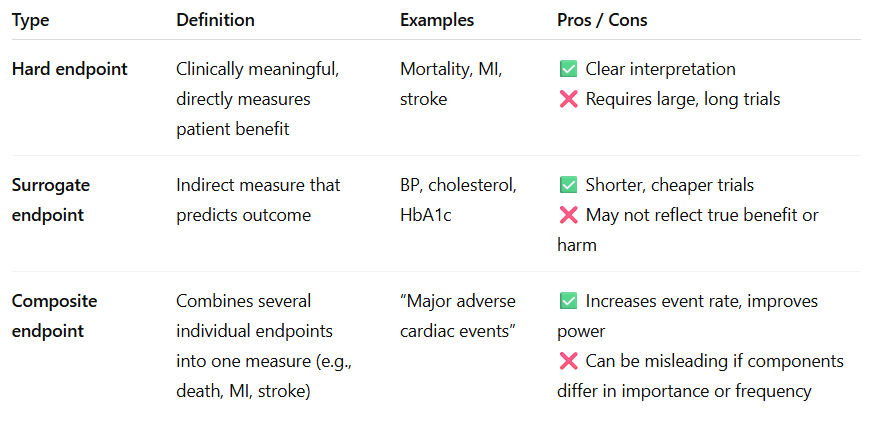
Distinguish between hard vs surrogate endpoints, and how composite endpoints are created and used.
Understand what a win-ratio analysis is and how it is calculated.
When composite endpoints include events of varying clinical importance.
Compares pairs of patients (treatment vs control) hierarchically by severity
compare for most important outcome
if tie → next outcome
continue until winner is determined
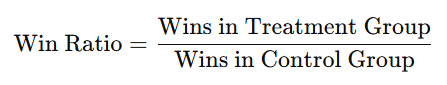
Advantages
prioritizes clinically important outcomes
Prevents trial events from overshadowing serious ones
Limitations
complex, careful hierarchy design required
less commonly used outside of cardio trails
Identify the reasons why RTC may be stopped early.
Benefit — intervention shows clear, overwhelming efficacy, continuing is unethical
Harm — excess adverse events or mortality in treatment arm
Futility — statistically impossible to show a difference with remaining data
Understand the role of subgroup analyses as well as the limitations of multiplicity testing.
Subgroup analysis
examines whether treatment effects differ among specific populations
Key rules
pre-specified (a priori)
multiplicity increases chance of false positives aka Type 1 error
findings are hypothesis-generating unless preplanned
Multiplicity
multiple statistic comparisons increase false-positive risk — must adjust alpha or use hierarchical testing
Recognize the difference between a priori vs post-hoc analysis
Priori
defined before seeing data
theory driven, part of stat plan
limited comparisons
ex. predefined subgroups or secondary outcomes
Post-Hoc
after seeing data
exploratory, hypothesis generating
many comparisons → increase type 1 error
Ex. unplanned new analysis after results seen
Interpret a Kaplan-meier survival curve.
Time to event graph
x-axis = time
y-axis = proportion of pts free from event
separation of curves → difference in survival, log rank test assesses statistical significance
Describe what a time-to-event analysis and how it is used.
how long until specific event occurs (death, relapse)
includes if and when event occurs
accounts for censoring
cox proportional hazards model → hazard ratio
Understand the difference between an intention-to-treat vs per-protocol analysis
Feature | Intention-to-Treat (ITT) | Per-Protocol (PP) |
|---|---|---|
Who is analyzed | All randomized participants, regardless of adherence | Only participants who followed protocol |
Preserves randomization? | ✅ Yes | ❌ No |
Reflects real-world use? | ✅ Yes | ❌ No |
Bias risk | Lower | Higher (attrition bias) |
Effect estimate | Conservative | May overestimate effect |
Regulatory preference | Primary analysis (FDA/EMA) | Supportive/sensitivity analysis |
ITT answers - does it work in practice
PP answers - does it work if taken exactly as prescribed
Understand the relationship among the determinants (α, β, etc.) that go into calculating sample size.
Term | Meaning | Effect on Sample Size |
|---|---|---|
α (alpha) | Type I error — probability of false positive (usually 0.05) | Lower α → larger sample needed |
β (beta) | Type II error — probability of false negative (usually 0.1–0.2) | Lower β (higher power) → larger sample |
Power (1–β) | Probability of detecting a true effect (typically 80–90%) | Higher power → larger sample |
Effect size | Expected difference between groups | Smaller effect → larger sample |
Event rate | Frequency of outcome | Lower event rate → larger sample |
Underpowered studies risk TYPE II ERRORS
Pragmatic, Randomized Controlled Trials [2 questions]
Distinguished how efficacy of an intervention differs from effectiveness in context of traditional RCT vs pragmatic clinical trial designs.
Concept | Traditional RCT (Explanatory) | Pragmatic Clinical Trial (PCT) |
|---|---|---|
Focus | Measures efficacy — Can it work under ideal conditions? | Measures effectiveness — Does it work in real-world practice? |
Setting | Controlled research environment with expert investigators | Conducted in everyday clinical settings (e.g., community clinics, health systems) |
Participants | Highly selected, homogenous, adherent | Diverse, typical patients seen in real-world care |
Protocol | Strict adherence, standardized procedures | Flexible, embedded within normal clinical workflow |
Goal | Determine biological or mechanistic cause–effect relationship | Evaluate impact on clinical decisions, patient outcomes, and policy |
Efficacy = ideal world → can it work?
Effectiveness = real world → does it work in practice?
Identify the main limitations to traditional randomized controlled trials that pragmatic trials are intended to address.
Traditional RCT Limitation | How Pragmatic Trials Address It |
|---|---|
Slow adoption of findings — research results take years to influence practice | PCTs embed research directly in health systems to speed translation |
Poor generalizability — highly selected populations | Include diverse, routine-care patients to reflect real-world use |
Unrealistic conditions — rigid protocols, perfect adherence | Flexible protocols aligned with usual care |
Expensive and time-consuming | Use EHR data and routine workflows to reduce cost and time |
Limited relevance to decision-makers | Prioritize outcomes meaningful to patients, clinicians, and policy makers |
Overreliance on surrogate or composite outcomes | Focus on practical, patient-centered outcomes |
Recognize the key features of a trail that make it pragmatic.
Pragmatic Clinical Trial → designed to improve practice and policy, not just test hypothesis
Core characteristics
real world clinical settings
health-system stakeholders involved
routine workflows and standard care procedures used
electronic health records is how data is collected
compares real-world alternatives, not just placebo
diver populations
measures outcomes important to decision makers like pts, clinicians, payers
cluster randomization used
seeks to generate real-world evidence more than lab-based efficacy data
Identify the potential challenges/disadvantages of a pragmatic design.
Challenge | Explanation |
|---|---|
Complex analysis | Multiple sites, variable implementation, and missing data complicate interpretation. |
Funding constraints | Large-scale, system-based research can still be costly. |
Regulatory uncertainty | Fewer formal FDA/NIH standards for pragmatic approaches. |
Ethical considerations | Consent can be complex when trials are embedded in routine care. |
Investigator experience | Many researchers lack training in pragmatic methods. |
Blurred responsibilities | Providers act as both clinicians and investigators — potential conflicts. |
Identify the domains of pragmatism of a trail that are evaluated using the PRECIS-2 scoring wheel.
Evaluates: how a pragmatic trial is across 9 domains
scored: 1 (very explanatory) → 5 (very pragmatic)
Wheel = closer to rim → more pragmatic; Trials near center → explanatory
Domain | What It Evaluates |
|---|---|
1. Eligibility | How inclusive or restrictive the patient criteria are |
2. Recruitment | How participants are identified and invited (routine vs special) |
3. Setting | Whether the trial occurs in specialized research centers or typical care environments |
4. Organization | The level of extra support or infrastructure needed beyond normal care |
5. Flexibility (Delivery) | How much the intervention protocol mirrors everyday practice |
6. Flexibility (Adherence) | Whether strict adherence is enforced or naturally observed |
7. Follow-up | Frequency/intensity of monitoring compared to normal care |
8. Primary Outcome | Whether outcomes are directly meaningful to patients/clinicians (vs surrogate) |
9. Primary Analysis | Whether analysis includes all real-world participants or only highly compliant ones |
RCT Designs: Noninferiority
Understand the key concepts in noninferiority trial design. Why does this design exist? What are the advantages, disadvantages? How does equipoise impact these trials?
Concept | Superiority Trial | Noninferiority Trial |
|---|---|---|
Goal | Show treatment is better than control | Show treatment is not unacceptably worse than control |
Null Hypothesis (H₀) | No difference (H₀ = 0) | New treatment is worse by ≥ Δ (H₀ ≥ Δ) |
Test Type | Two-sided | One-sided |
Common α and β | α = 0.05, β = 0.20 | α = 0.025 (1-sided), β = 0.20 |
Reject H₀ when… | CI does not cross 1 or 0 (depending on measure) | CI does not cross Δ (margin) |
Understand the similarity and differences between how superiority and noninferiority trails are conducted, analyzed, and interpreted.
Aspect | Superiority | Noninferiority |
|---|---|---|
Comparator | Placebo or active control | Active control (SOC) |
Hypothesis | H₀: No difference (H₀=0) | H₀: New ≥ Δ worse |
Statistical test | Two-sided | One-sided |
Alpha (α) | 0.05 | 0.025 |
CI threshold | Does not cross 0 or 1 | Does not cross Δ |
Goal | Show new treatment is better | Show new treatment is not unacceptably worse |
Interpretation | If CI excludes null → superior | If CI excludes Δ → noninferior |
Next step | If NI shown, can test for superiority | Must meet NI first before claiming superiority |
Ethical context | Placebo often ethical | Placebo unethical when effective therapy exists |
Be able to read/understand/apply (parts of) a journal article of a noninferiority trial.
Meta-Analysis
Be able to identify the strengths and weaknesses of meta-analysis as a study design.
Understand the steps to conducting a meta-analysis and how the study design impacts the effect estimate.
Understand the difference between random-effects and fixed-effects models.
Understand and apply the various sources and measures of heterogeneity.
Be able to read and interpret tables and forest plots.
Understand the tradeoff between the research questions (inc/exc), number/similarity of studies, bias and heterogeneity.