uses of alkanes in crude oil
Crud roil is a mixture of different hydrocarbons (mainly alkanes)
Crude oil itself is useless. We must separate the alkanes until useful fractions. Alkanes can be separated from the crude oil because they have different boiling points.
Because the different fractions made from crude oil have different properties they also have different uses. Many fractions are used for fuels, such as petroleum gas.
How is crude oil separated into fractions?
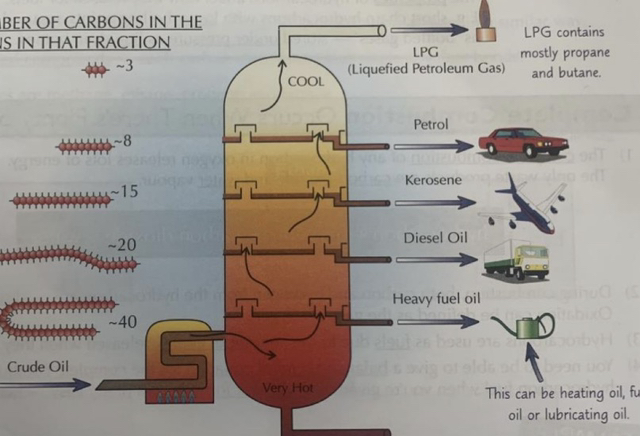
the crude oil is heated outside the column where most of it vaporises
The mixture, of vapours and liquids, is pumped into the bottom of the column
The liquid is drained off from the bottom of the column
Inside the column is a temperature gradient, meaning the bottom is hot and the top is cool
The vapours rise up the column
Longer hydrocarbons have higher boiling/ condensation points. This is because they are larger molecules with stronger IFs, which takes more energy to break
Longer chain hydrocarbons condense back into liquids towards the bottom of the column where the temperature is higher. The liquids are collected and used for different purposes.
Shorter chain hydrocarbons have lower boiling points. This is because they are smaller molecules with weaker intermolecular forces, which takes less energy to break.
Shorter chain hydrocarbons condense back into liquids towards the top of the column where the temperature is lower.
The liquids are collected and used for different purposes.
In short
the crude oil is heated outside of the column where most of it evaporates
The mixture of gas and liquid enters the bottom of the column where its hot
The liquid is drained off and the gas begins to rise through the column where its hot at the bottom and cool at the top
This allows the gases to separate out and condense at separate points so they can be used