In-Depth Notes on Luminescence Spectroscopy (PPT)
Overview of Luminescence Spectroscopy
Luminescence and fluorescence are based on multiplicity
Fluorescence (emission) doesn’t involve a spin flip and Ms=1 (singlet state) electrons still paired
Phosphorescence (emission) involves a change in spin state, leading to a triplet state (Ms=3) where the electrons are unpaired, allowing for longer-lived excited states and delayed emission of light.
Spin flip shouldn’t happen but can in heavy atoms due to interactions in the magnetic fields
A technique widely utilized in chemistry for the emission of photons.
Notably significant for biochemical applications due to its high sensitivity and low detection limits, making it effective for trace analysis.
nanomolar or picomolar detection limit
Electronic states influenced by environment so luminescence can give a lot of info.
Instrumentation
Basics
Laser-Based System: Sample is irradiated with photons, which are absorbed.
fluoresce is proportional to the intensity of the source
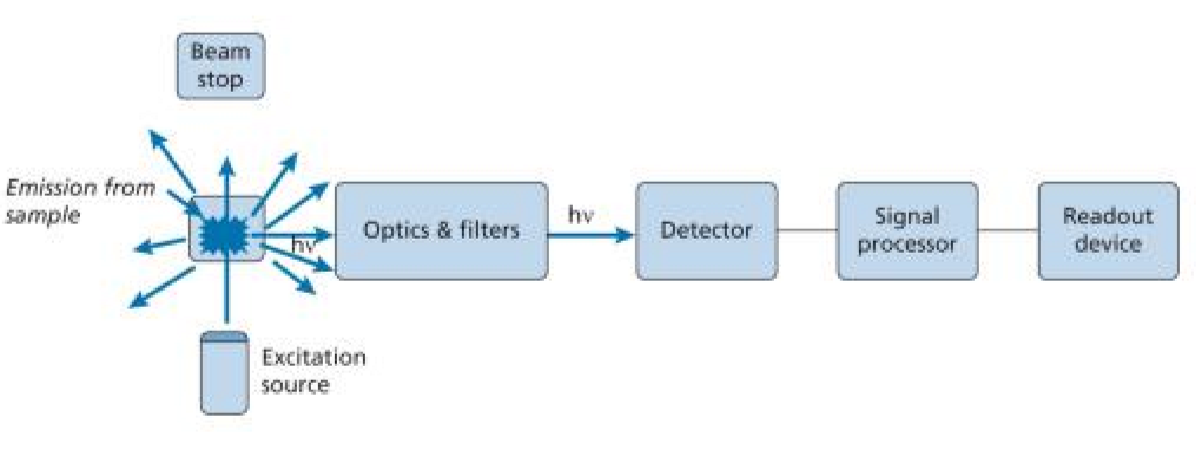
Steady State: Two principal types of instruments based on spectral analysis.
Source is continually exciting the sample and equal amounts of ground state and excited state atoms
Have an excitation wavelength and scan the emission wavelength
Example: Pyrene absorbs at 338nm
Frequency Domain: looking at the actual wave itself
wavelength hits sample and there is a delay in the absorption and emission
see the difference in the intensity and wavelength shift
Time Domain: pulsing the domain and seeing emission at some time t
usually in nanosecond
time resolved spectroscopy: pulsing the sample and slowly going to higher or shorter energies overtime
red edge effects: start exciting at the red edge of the band then you can look at molecules in different environments. Viscous solutions you can see the different electronic effects
Blue edge effects (higher energy state molecules)
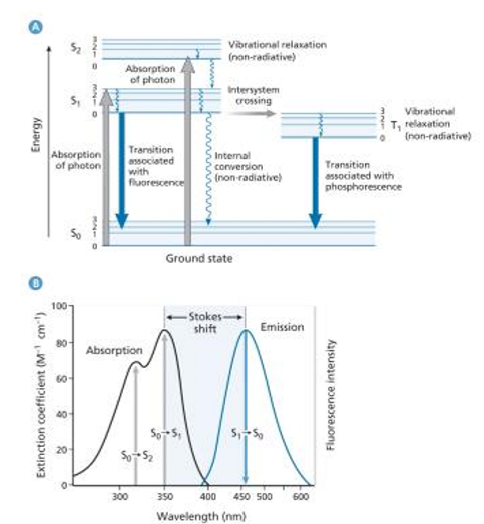
Jablonski Diagram: Illustrates radiative (photon emission) and non-radiative processes (energy loss without photon emission; thermal energy).
S0 = ground state
S1 = first excited singlet state (Majority of molecules emit from the lowest state of S1)
Manifold: all the vibrational and electronic states that are a part of that level (S1 is a level)
S2 = second excited state (only one molecule the emits from S2)
S2 → S1; S1→S0: internal conversion; non-radiative energy loss (thermal)
All these processes taking place simultaneously
Intersystem crossing: the process where a molecule transitions between different spin states, typically from a singlet state to a triplet state, resulting in a longer lived excited state.
Delayed fluorescence: electron crossed over to the T1 state but there is enough energy to push it back and the spin can flip and cause emission
The rates constants determine the fluoresce or phosphorescence output
Stokes shift: The lose of energy of the excited photon before emission
wavenumber of max wavelength absorption - wavenumber of max wavelength the emission = delta v
Mirror image rule: emission spectrum is identical to the absorption spectrum in shape but occurs at a different energy, typically lower due to the Stokes shift.
Timescales of Emission:
Absorption: 10^{-15} seconds (femtoseconds).
Internal conversion/vibrational relaxation: 10^{-14} to 10^{-10} seconds.
Fluorescence: 10^{-9} to 10^{-7} seconds. (intersystem crossing)
Phosphorescence: 10^{-3} to 10^{-2} seconds (very slow due to forbidden transitions).
All affect whether or not you get emission
Cell Geometries
Different geometries facilitate the varying requirements for luminescence measurements.
45 deg used for solid samples.
direct is not really used
Key Concepts
Rates of Luminescent Processes
Fluorescence: Transition from the first excited singlet state to the singlet ground state.
random phenomenon, all molecules have an equal probability to emit after being excited
Wavefunctions of the ground state to the excited have to be similar
Phosphorescence: Transition from the first excited triplet state to the singlet ground state; requires a spin flip, which is quantum mechanically not allowed.
Kasha’s Rule: Photon emission typically occurs from the lowest excited state rather than higher states, ensuring effective emission.
Emission should arise from E=0 in singlet S0 and E=0 in T1
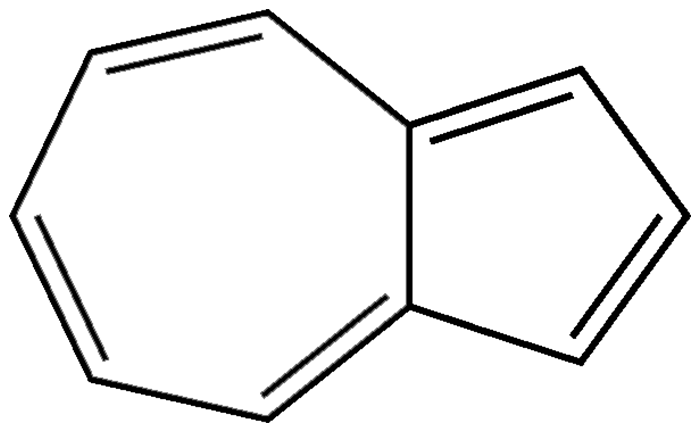
Azulene is the only exception to Kasha’s rule
Delayed Fluorescence
Types
E-type (Eosin): Temperature-dependent; excitation leads to intersystem crossing; significant for understanding relaxation under variable thermal environments.
P-type (Pyrene): Intensity proportional to the square of excitation intensity; requires two photons and two molecules interacting to regenerate fluorescence.
Recombination Fluorescence: Involves photooxidation leading to an excited molecule without immediately returning to the ground state.
Excimers and Exciplexes
Excimer: Dimer formed in an excited state involving the same molecule, where the excitation energy is shared.
Exciplex: Involves two different molecules; one in an excited state and one in the ground state, typically functioning as electron donor and acceptor.
Quantum Mechanics in Luminescence
Frank-Condon Factor: Transition probability correlated with vibrational wave function overlap between two states; larger overlap leads to more intense fluorescence.
Quantum Yield: A measure of the efficiency of photon emission; high quantum yields correlate with effective luminescence.
External Influences
Heavy Atom Effects: Influence on spin-orbit coupling that enhances intersystem crossing.
Variability in Quantum Yield: Explored in both fluorescence and phosphorescence contexts.
Single Photon Counting Techniques
Purpose and Application
Utilizes Time-Correlated Single Photon Counting (TCSPC) for detecting short-lived excited states of molecules on the ns timescale.
Experimental Setup: Incorporates advanced electronics to facilitate the timing and correlation of excitation and emission signals.
Necessary Equipment
Components include Constant Fraction Timing Discriminators, Time-to-Amplitude Converters, Pulse Height Multichannel Analyzers, and delay lines to boost accuracy in measurements.
Quenching Phenomena
Types of Quenching
Dynamic and Static Quenching: Involve nonradiative energy transfer to neighboring molecules or interactions forming ground state complexes.
Stern-Volmer Equation: A tool to evaluate dynamic quenching effects and calculate quantum yield in the presence of quenchers.
Classifications of Quenchers
Inorganic (e.g., heavy atoms), Organic (various organic compounds), and Self-quenching where the quencher is the same as the detecting molecule.
Molecular Structure and Its Impact on Luminescence
Molecular Features: Compounds with rigid polyconjugated π-systems and aromatic moieties promote fluorescence.
Nonradiative Processes: Can be minimized in rigid matrices or at low temperatures, ensuring maximum emission efficiency.
Instrumentation Details
Fluorescence Microscope
Typically uses a Xe arc lamp generating broad output, necessary for high-intensity measurements.
Detectors
PMTs (Photomultiplier Tubes) and CCDs (Charge-Coupled Devices) are prime for luminescence.
PMTs are faster with pulsed applications, whereas CCDs excel in imaging applications due to higher quantum efficiency.
Summary
Luminescence spectroscopy is a valuable analytical technique involving intricate processes governed by quantum mechanics. Its versatility across scientific disciplines underscores the significance of thorough understanding and improved instrumentation outcomes.