Alkyl Halides Sn1 Reactions
Recap of Sn2 Reactions
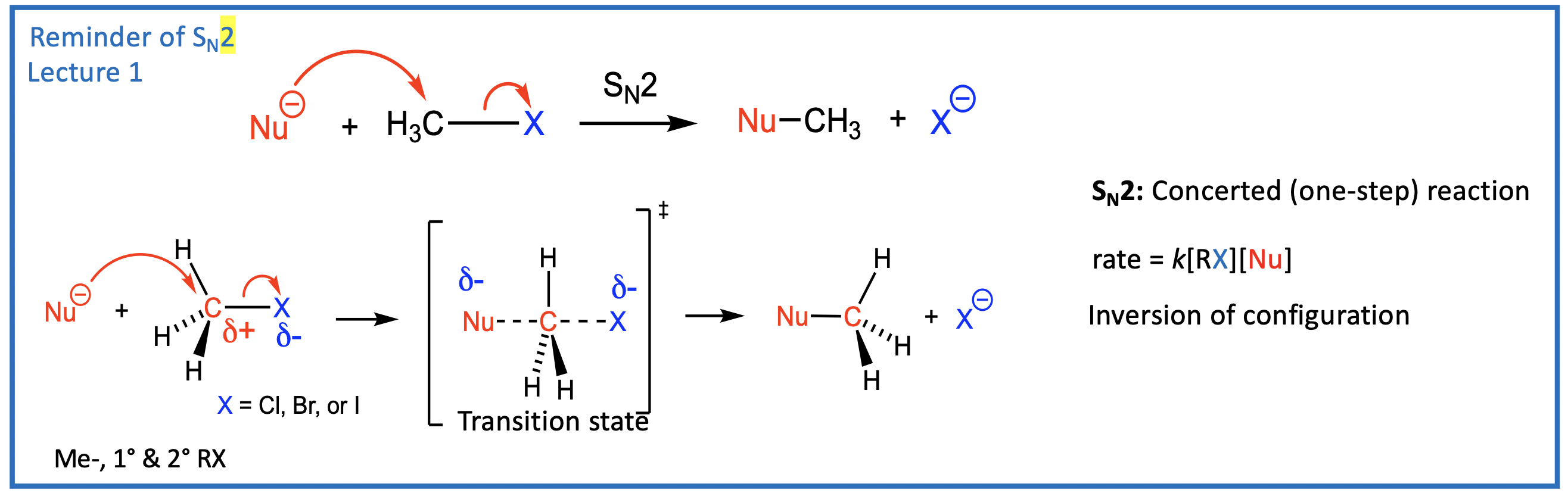
“backside attack“ pushes electron from left to right.
transition state - shows whats happening, starting material to product.
one step reaction
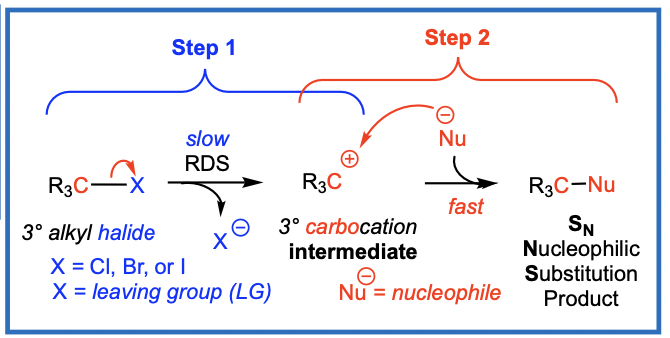
The Sn1 reaction with 3deg alkyl halides
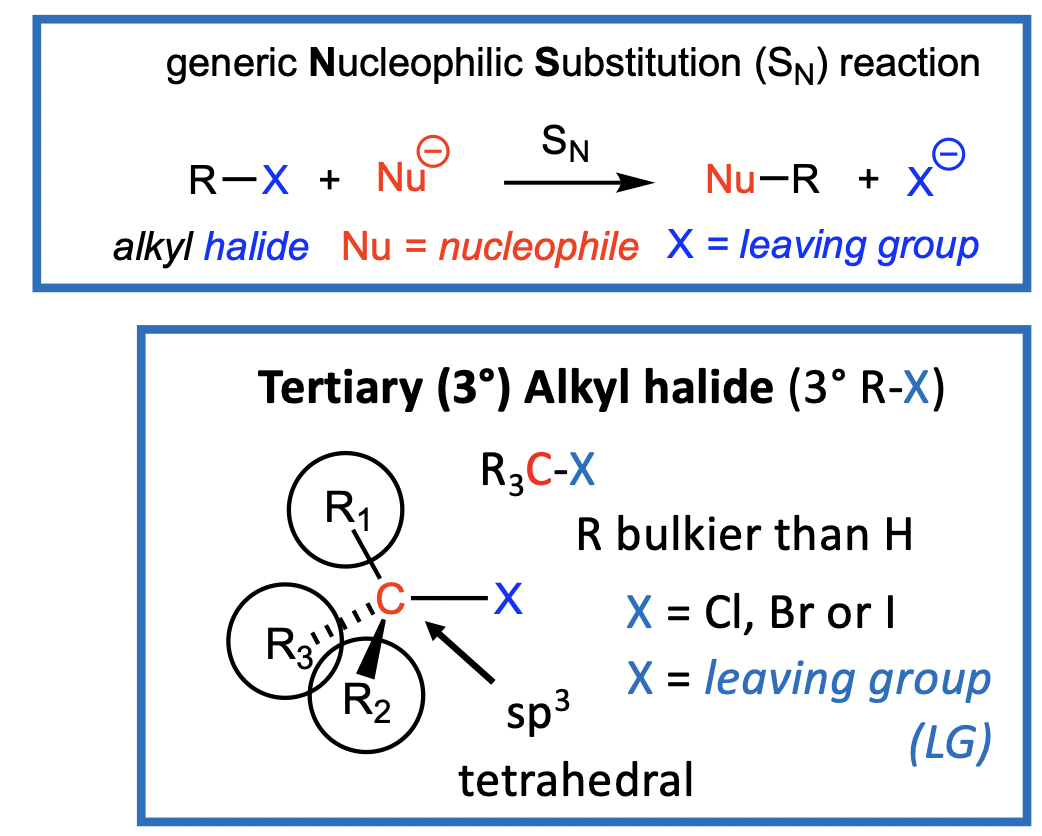
Generally, tertiary (3°) alkyl halides undergo SN1 reactions with nucleophiles
SN1: Substitution, Nucleophilic, Unimolecular
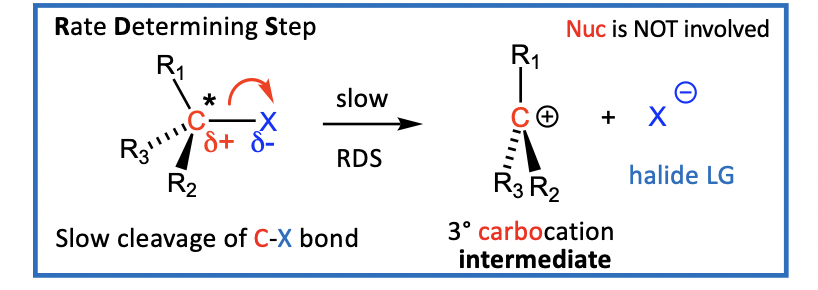
The general SN1 reaction has a two-step mechanism
Steps
C-X bond breaks to form a carbocation (R3C+) and a halide (X-)
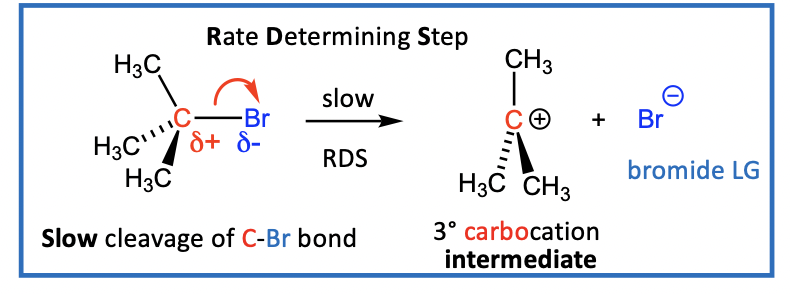
step one is slow, RDS Rate Determining Step (RDS)
C-X bond cleaves to give a 3° carbocation intermediate (R3C+) and the leaving group, a halide (X-)
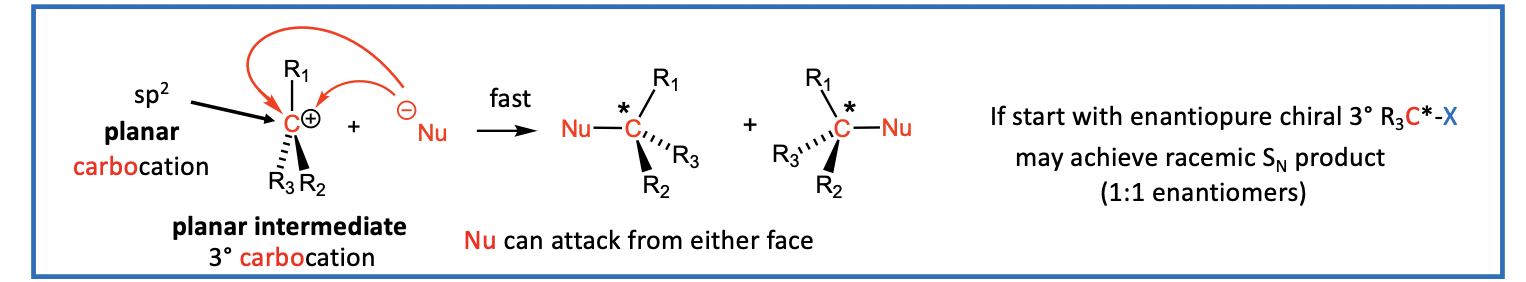
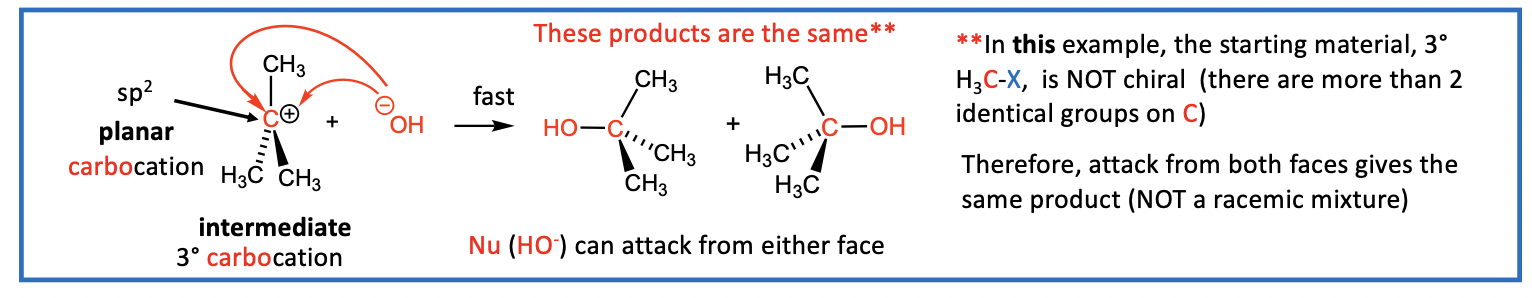
Step 2: Now the nucleophile (Nu) attacks the carbocation (R3C+) intermediate to form a new bond (Nu-C)
step 2 is fast
Nu attacks the 3° R3C+ intermediate to give SN product
Overview
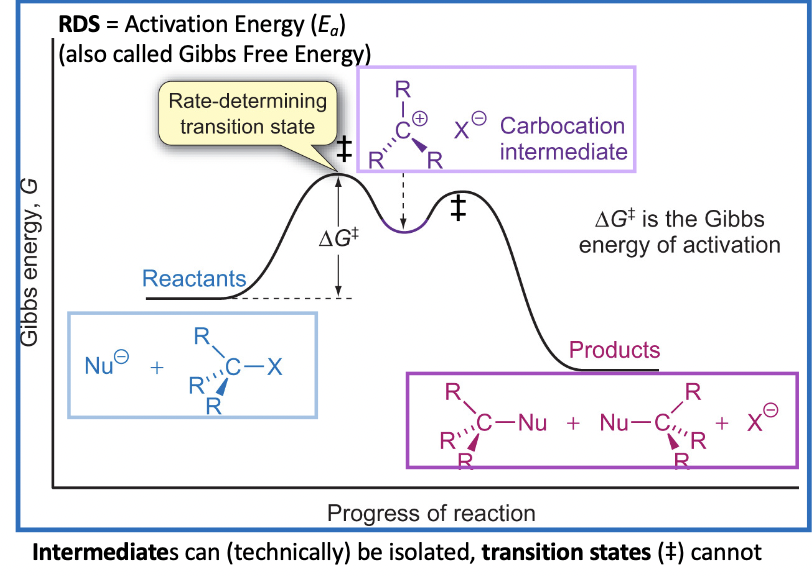
Remember: Mechanism is deduced from kinetic studies
In a Rate Equation, the molecularity of a reaction is the
number of molecules taking part in the RDS of the reaction In this case, the nucleophile is not involved in the RDS
So we can delete this from our equation
rate = k [R3C-X][Nu ] rate = k [R3C-X]
Rate of this Nucleophilic Substitution (SN) depends on theconcentration of only RX (not the Nu)
So, the reaction is: first order = Sn1 (unimolecular)
Why do 3° RX undergo SN1 rather than SN2 reactions?
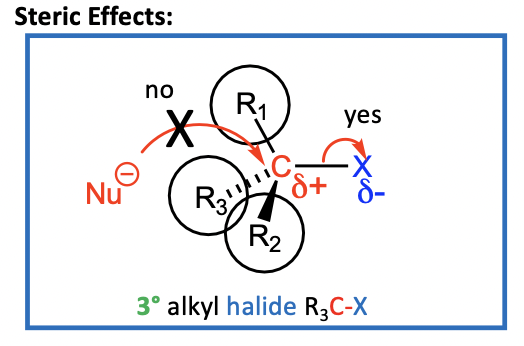
Steric effects
Remember: SN2 requires Nu to approach electrophilic C of R3C-X close enough to form a bond & displace the halogen leaving group, X
• Must approach C from the opposite side to the halide (X) for: • orbital overlap AND to avoid negative charge of halogen
• Recall that alkyl groups are bigger than H’s
• Approach by the nucleophile to the electrophilic C of R3C-X is hindered
by the three bulky R (alkyl) groups
So, with 3° RX, SN2 is difficult to achieve and SN1 is favoured
Also, with the release of steric strain, with loss of LG & the formation of carbocation, drives reaction forward “steric acceleration”
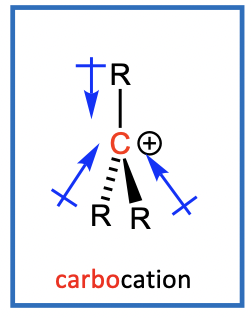
Electronic effects
Alkyl groups help stabilise positive charge on carbocation & electrophilic C of R3C-X
More alkyl groups = more stabilisation
If electrophilic C of R3C-X is stabilised, easier to cleave C-X bond
If carbocation intermediate is stabilised, helps reaction move forward
More alkyl groups = SN1 is more favoured

methyl, primary, secondary
tertiary
Example of a tertiary alkyl halide undergoing a SN1 reaction with a nucleophile
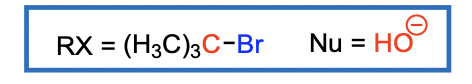
C-Br bond breaks to form carbocation (R3C+) and a halide (Br-)
the nucleophile (Nu) attacks the carbocation (R3C+) to form a new bond (Nu-C)
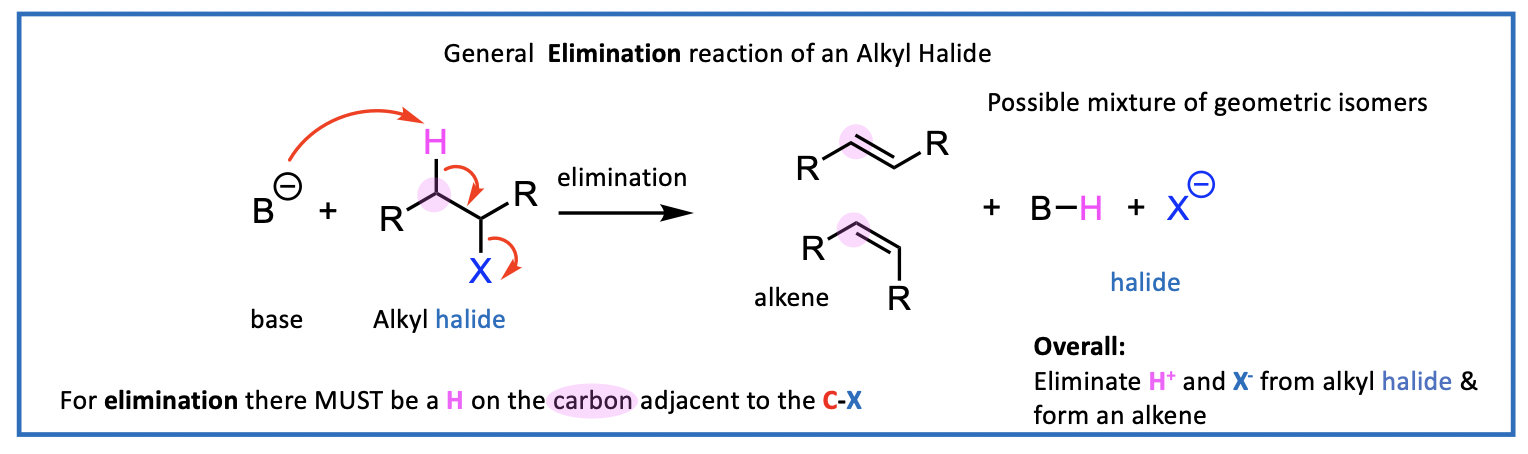
Elimination reaction with a base
using a base instead of a nucleophile
still losing halide, also loses a hydrogen (H+) and (X-)
The base captures H+ and this is generally shown as B-H on the reaction schemes.
forming an alkene
identify secondary alkyl halide (one carbon away, does it have a hydrogen attached?)
base does not attack carbon - attacks hydrogen (proton) and pushes to form alkene
different from Sn reactions - forms alkene
pink shows where lost hydrogen comes from
Competition may occur between Sn and elimination reactions
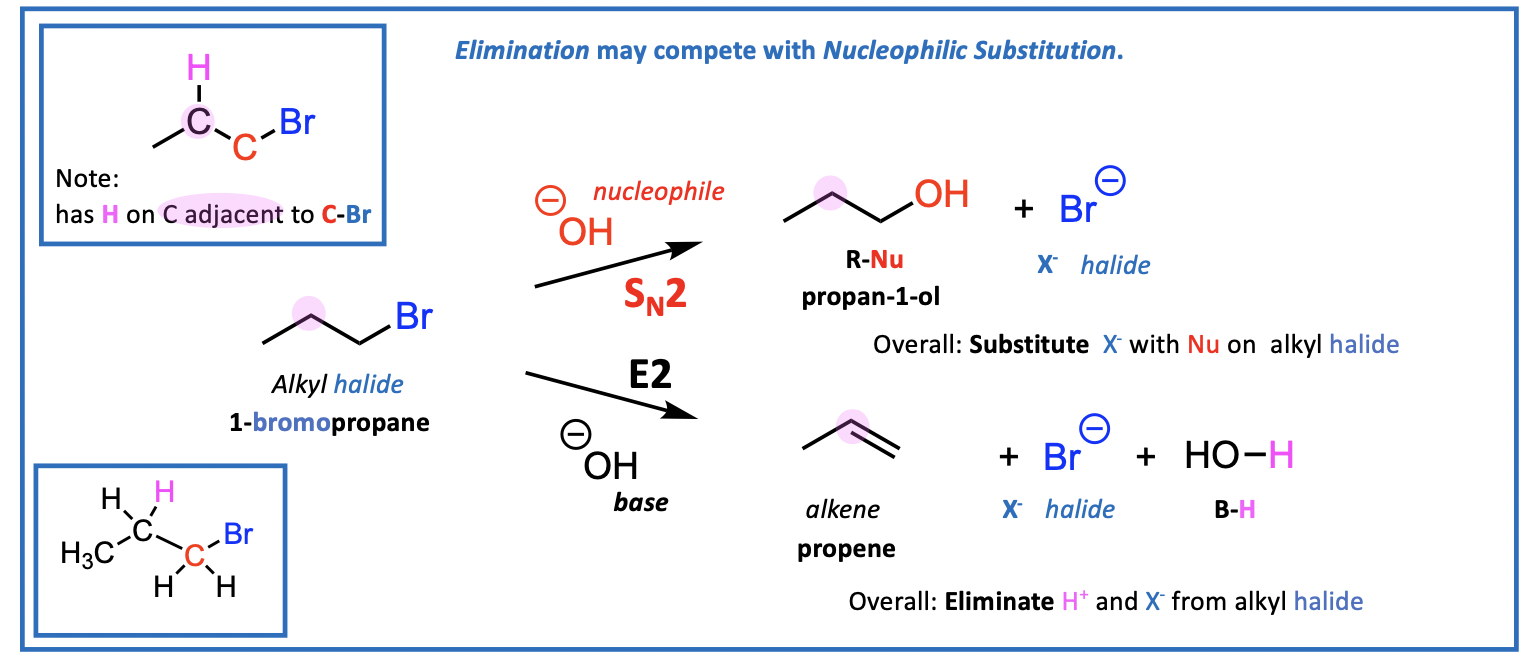
can act as a base or a nucleophile
Example of E2 reaction
The E2 reaction has a one-step (concerted) mechanism
E2: Elimination, Bimolecular rate = k [R-X][base]
{E2 Goes through a transition state but we will not explore these details}
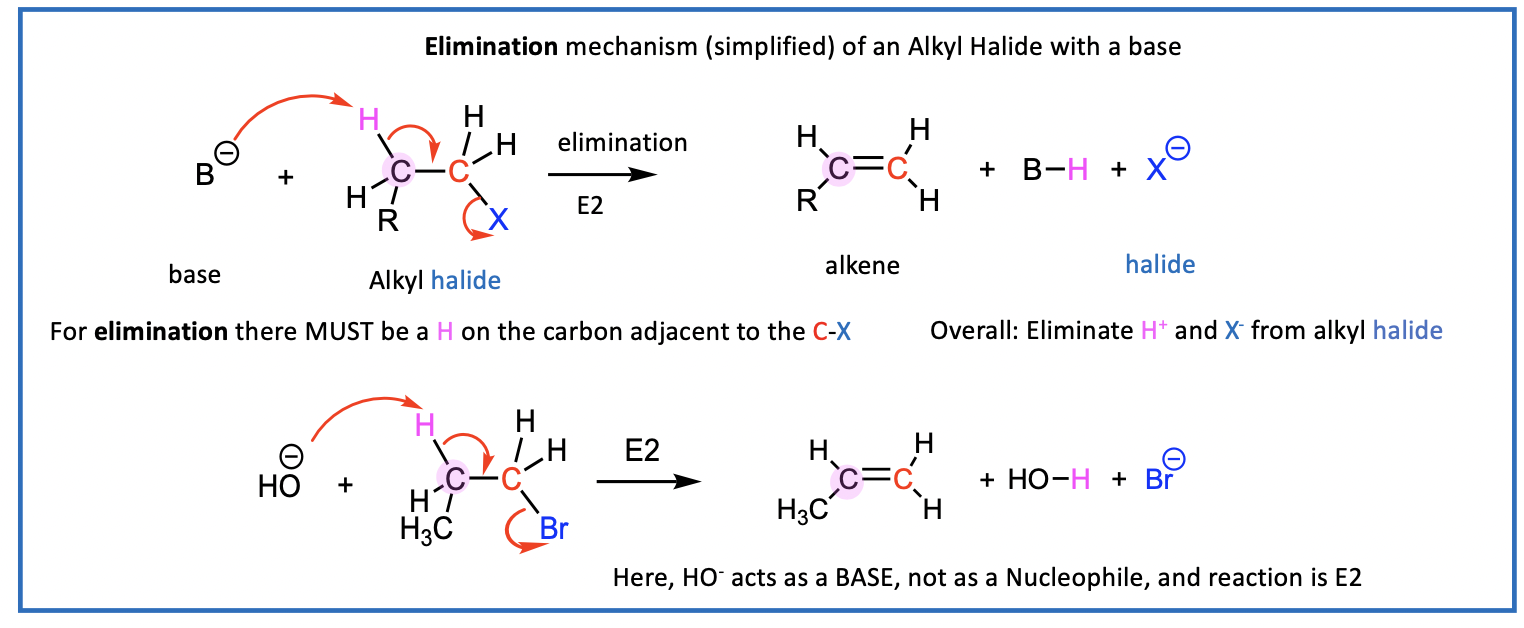
imagine what the product will look like
Alternative elimination products
For elimination there MUST be a H on the carbon adjacent to (next to) the C-X
Overall: Eliminate H+ and X- from alkyl halide
But what happens if there are more than one adjacent carbons, each with a H? You can get a variety of elimination products
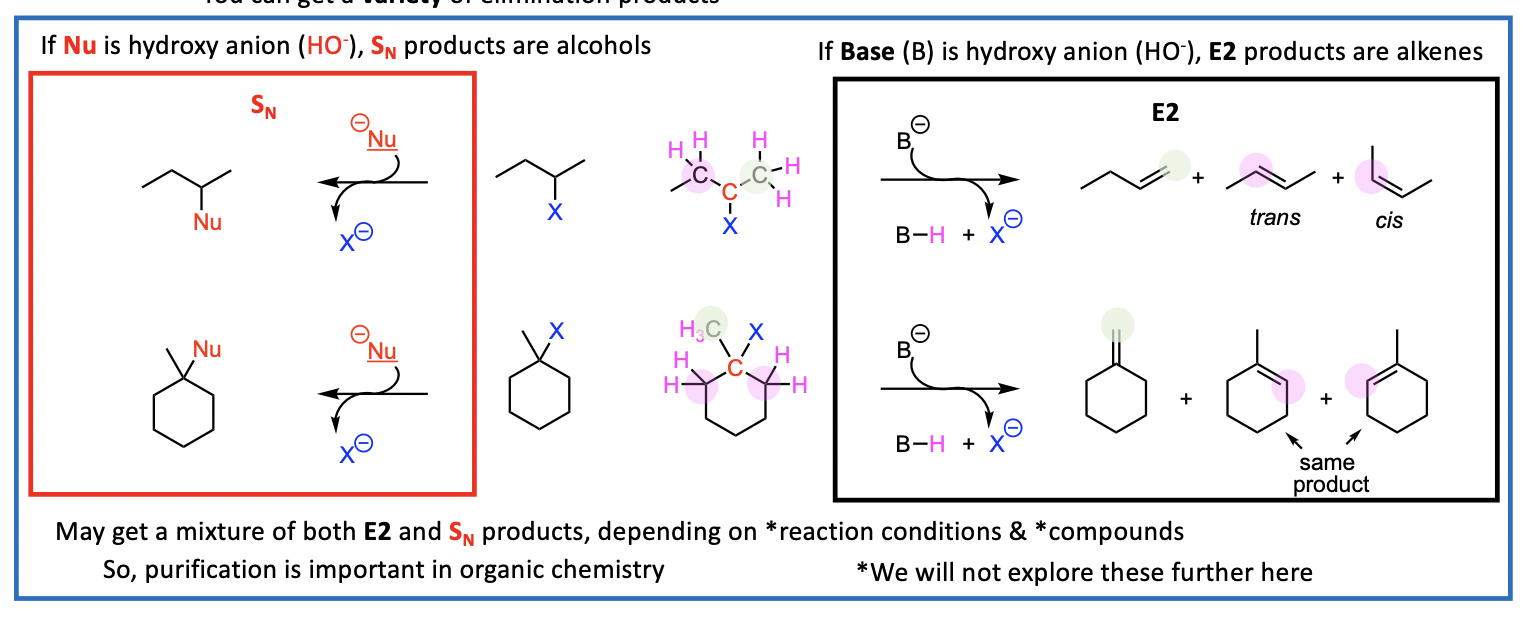
May get a mixture of both E2 and SN products, depending on reaction conditions & compounds
Therefore, purification is important in organic chemistry
Grignard Reagents
Alkyl halides are used to create Grignard Reagents
Grignard Reagents are organomagnesium compounds with the general formula RMgX Victor Grignard awarded a Nobel prize in 1912
How to make a Grignard Reagent?
Reaction of Mg with alkyl halides (in dry diethyl ether)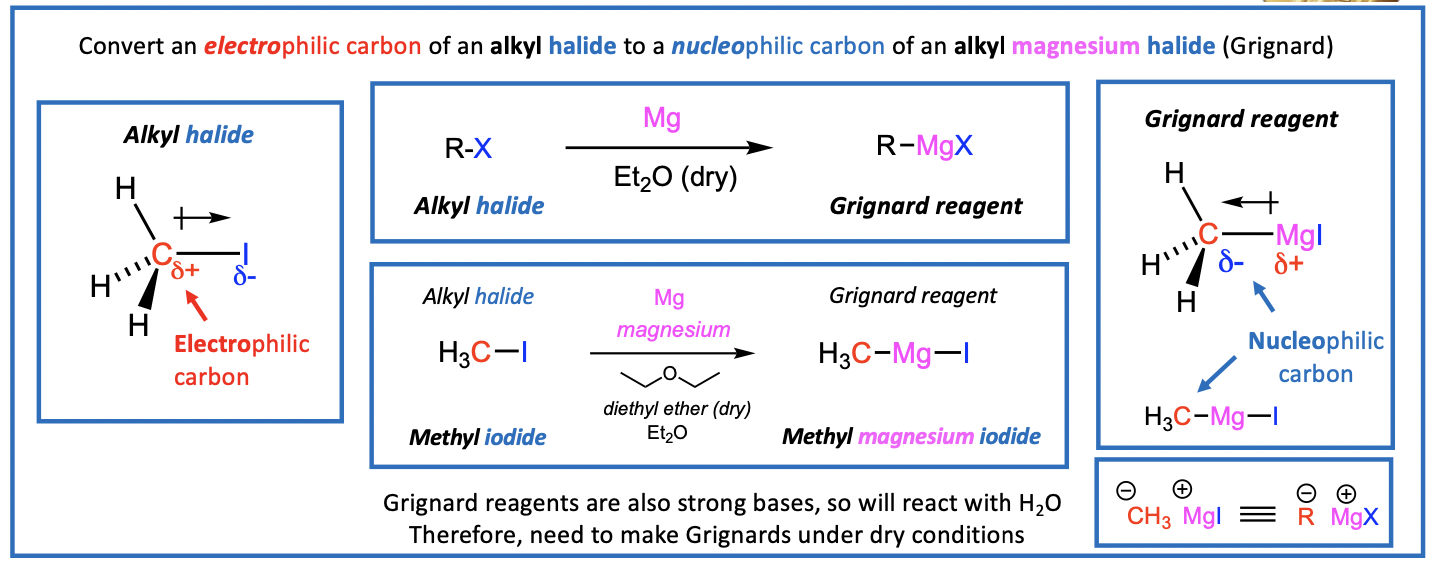
Mg inserts itself into the bond, flipping the electronegativity.
carbon becomes nucleophilic