Regulatory Mechanisms and Fibrinolysis Student Outline.docx
MLS 340
Regulatory Mechanisms and Fibrinolysis
- The Hemostatic System- Initiation & Propagation
- Regulator mechanisms
- Fibrinolysis
The Hemostatic System: Initiation and Propagation
- Pathways are not distinct, independent – actually interdependent.
- Normal coagulation requires
- Cells that express TF (usually extravascular)
- Platelets, usually intravascular
- 2 Phases
- initiation
- Occurs on TF expressing cells
- 3-5% thrombin
- propagation
- Occurs on platelets
- 95% or more of thrombin
- initiation
Initiation
- Begun by formation of extrinsic tenase complex (VIIa, TF, +Ca++, phospholipids)
- Generation of small amounts of thrombin
- Less efficient than intrinsic tenase complex
- But platelets, cofactors and procoagulants become activated
- Low level of thrombin generated in the initiation:
(1) activates platelets through cleavage of protease activated receptors PAR-1 and PAR-4
(2) activates factor V released from platelet a-granules
(3) activates factor VIII and dissociates it from VWF
(4) activates factor XI, the intrinsic accessory procoagulant that activates more factor IX
(5) Splits fibrinogen peptides A and B from fibrinogen and forms a preliminary fibrin network. The initial platelet plug is thus formed.
Propagation
- More than 95% of thrombin generation
- Occur on the surface of activate platelet
- Large number of platelets adhere to site of injury
- By low levels of thrombin (initiation)
- Adhering to exposed collagen
- This creates COAT platelets (collagen and thrombin)
- COAT Platelets
- higher level of procoagulant activity than platelets exposed to collagen alone
- provide a surface for formation and amplification of complexes
- Both platelets and tissue factor-bearing cells are essential for physiologic coagulation
- Deficiencies of key proteins (VII, IX, VIII, X, V, or prothrombin) compromise thrombin generation and manifest as significant bleeding disorders
 Coagulation Regulatory Mechanisms
Coagulation Regulatory Mechanisms
- Balance between procoagulant & anticoagulant systems.
- Activated factors and/or platelets must be kept at the site of injury so fibrin formation is limited to the site of vascular injury
- Factors and platelets must be controlled so they are inactive when distant from a site of vessel damage and blood remains fluid in uninvolved vessels.
Principle Regulators
Tissue Factor Pathway Inhibitor (TFPI)
Antithrombin (AT)
activated protein c (APC)
Tissue Factor Pathway Inhibitor (TFPI)
- TFPI is synthesized primarily by ECs and is also expressed on platelets
- Binds to and inhibits
- factor xa
- VIIa:TF complex
- 2 Step Process
- First TFPI binds factor Xa and inactivates it
- then TFPI:Xa complex binds and inactivates TF: VIIa, preventing more activation of Xa.
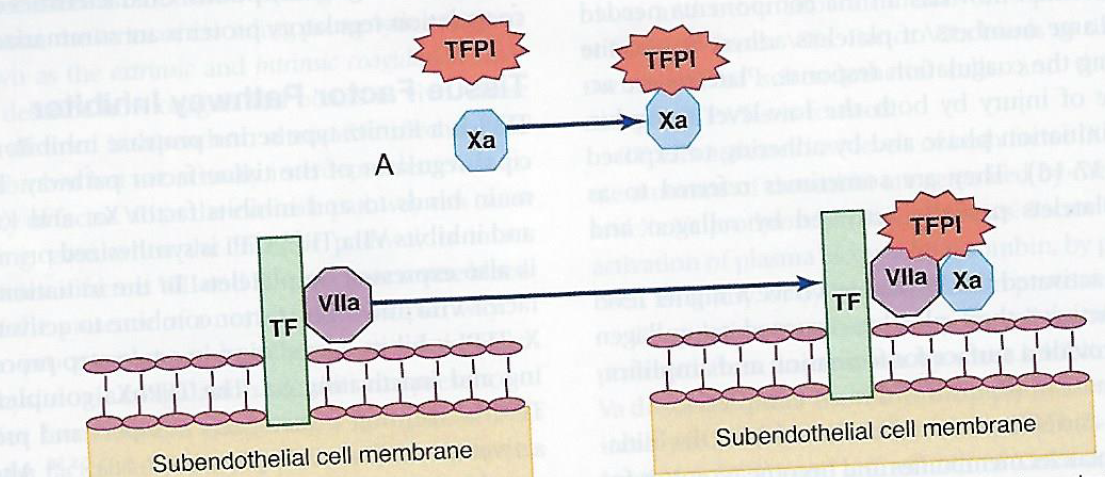 Alternatively, TFPI may bind directly to Xa and VIIa in the TF:VIIa:Xa complex
Alternatively, TFPI may bind directly to Xa and VIIa in the TF:VIIa:Xa complex
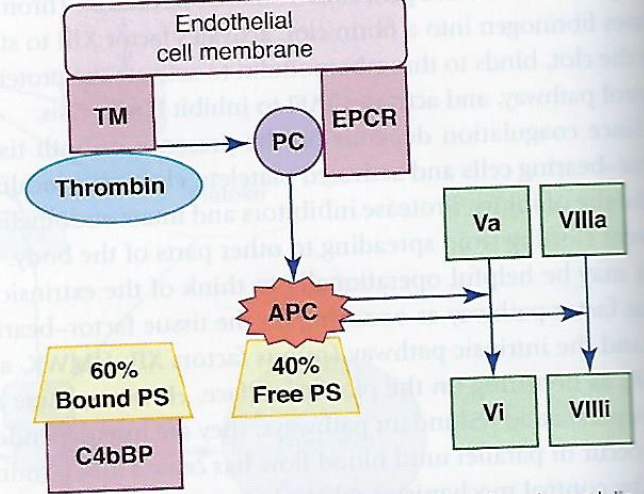
Protein C Regulatory System
- Thrombin cleaves fibrinogen generating a fibrin clot
- activates factors v, viii, xi, and xiii
- Propagating more thrombin generation
- In intact normal vessels
- thrombin avidly binds thrombomodulin
- triggers protein c regulatory system
- revises thrombin’s function from a procoagulant enzyme to an anticoagulant.
- EC protein C receptor (ECPR)
- transmembrane protein
- binds protein C adjacent to the thrombomodulin-thrombin complex.
- augments the action of thrombin-thrombomodulin at least fivefold
- Activated protein C (APC)
- Dissociates from EPCR and binds its cofactor, free plasma protein S.
- stabilized APC-protein S complex hydrolyzes and inactivates factors
- Va and VIIIa
- slowing or blocking thrombin generation and coagulation.
- Protein S
- cofactor that binds and stabilizes apc
- synthesized the liver and circulates in the plasma in two forms:
- 40% of protein S is free
- 60% is covalently bound to complement control protein C4b-binding protein (C4bBP)
- bound protein S cannot participate in the protein C anticoagulant
- free plasma protein S can serve as the APC
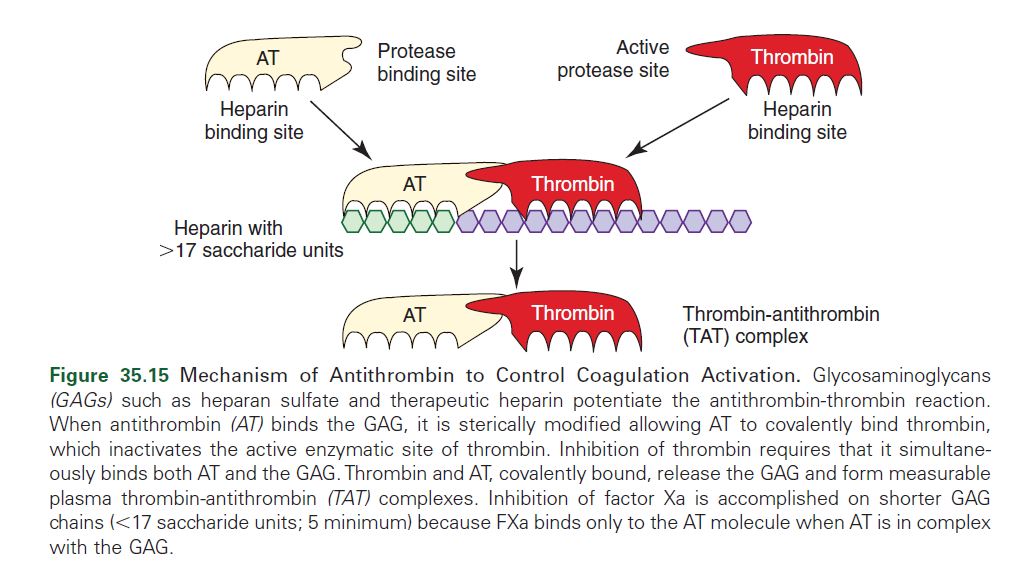
Antithrombin (AT)
- serine protease inhibitor (serpin)
- binds and neutralizes serine proteases
- thrombin
- Ixa
- Xa
- XIa
- XIIa
- Prekallikrein
- Plasmin
- Requires heparin for effective anticoagulant activity.
- available from endothelium-associated mast cell granules or as EC heparan sulfate.
- AT’s amplified 2000-fold by binding to heparin
- Heparin induces a conformational change in the AT molecule
- allows binding of activated coagulation factors, causes inactivation
- Inhibition of thrombin, factor X, and other serine proteases by AT is dependent on:
- length of the heparin chain
- Longer heparin chains are able to bind both molecules to produce inhibition of thrombin
Heparin Cofactor II (HCII)
- 2nd line inhibitor of thrombin
- primarily targets thrombin
- requires heparin
Other SERPINS
- ZPI
- protein C inhibitor
- a1-protease inhibitor (a1-antitrypsin)
- a2-macroglobulin
- a2-antiplasmin
- PAI-1
Fibrinolytic System
- Activation of coagulation also activates fibrin lysis.
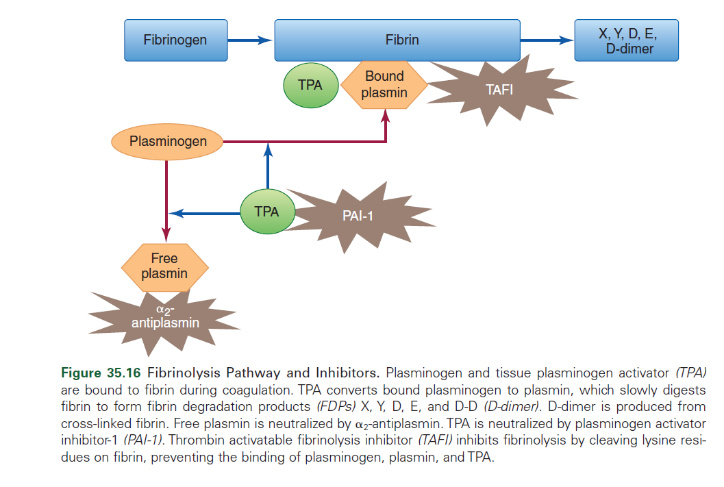 Removal of unwanted fibrin deposits by bound plasmin.
Removal of unwanted fibrin deposits by bound plasmin.- Re-establishes blood flow and facilitates the healing process
- Two activators of fibrinolysis
- tissue plasminogen activator (TPA)
- Urokinase plasminogen activator (UPA),
- Convert fibrin-bound plasminogen into plasmin
- the principle enzyme of the fibrinolytic system
- Convert fibrin-bound plasminogen into plasmin
- delicate balance between the activators and inhibitors in this system.
Plasminogen
- Plasmin is a serine protease that systematically digests fibrin polymer
- Synthesized by the liver.
- Circulates as a zymogen.
- Plasminogen is activated to plasmin by tissue plasminogen activator (tPA), urokinase, or other endogenous activators:
- Factor XIa, XIIa fragments, Kallikrein, HMMK
Plasmin
- Bound plasmin digests clots and restores blood vessel patency.
- free plasmin can be found in the circulation
- capable of digesting plasma fibrinogen, factor V, factor VIII, and fibronectin.
- x, y, d, e d-dimers
Tissue Plasminogen Activator (TPA)
- ECs secrete TPA
- hydrolyzes fibrin-bound plasminogen
- converting it to plasmin
- initiating fibrinolysis
- Circulating TPA is bound to inhibitors such as PAI-1
- complexes are cleared from circulation
- synthetic recombinant TPAs
- mimic natural TPA
- “clot-busting”
Urokinase Plasminogen Activator
- Secreted in urinary tract epithelial cells, monocytes, and macrophages
- Doesn’t bind to fibrin
- Becomes incorporated into the mix of fibrin-bound plasminogen and TPA at the time of thrombus formation
- Intrinsic plasminogen activator
- Small amounts circulate in the plasma and thus it plays a minor role in in-vivo fibrinolysis.
- Purified urokinase preparations are widely used to dissolve clots
Plasminogen Inhibitors
Plasminogen activator inhibitor-1 (PAI-1)
- Principle inhibitor of plasminogen
- Inactivate tpa and upa
- is produced by ECs, megakaryocytes, smooth muscle cells, fibroblasts, monocytes, adipocytes, hepatocytes, and other cell types
- Behaves as an acute phase reactant protein.
- Increased after major surgery, MI and severe trauma.
- Increased PAI-1 levels correlate with reduced fibrinolytic activity and increased risk of thrombosis
Plasmin inhibitors
Alpha 2 antiplasmin
- Main inhibitor of plasmin and therefore the fibrinolytic system.
- Interferes with absorption of plasminogen to fibrin.
- Same binding site as fibrin therefore the plasmin absorbed onto fibrin is protected from the action. Only free, circulating plasmin is attacked
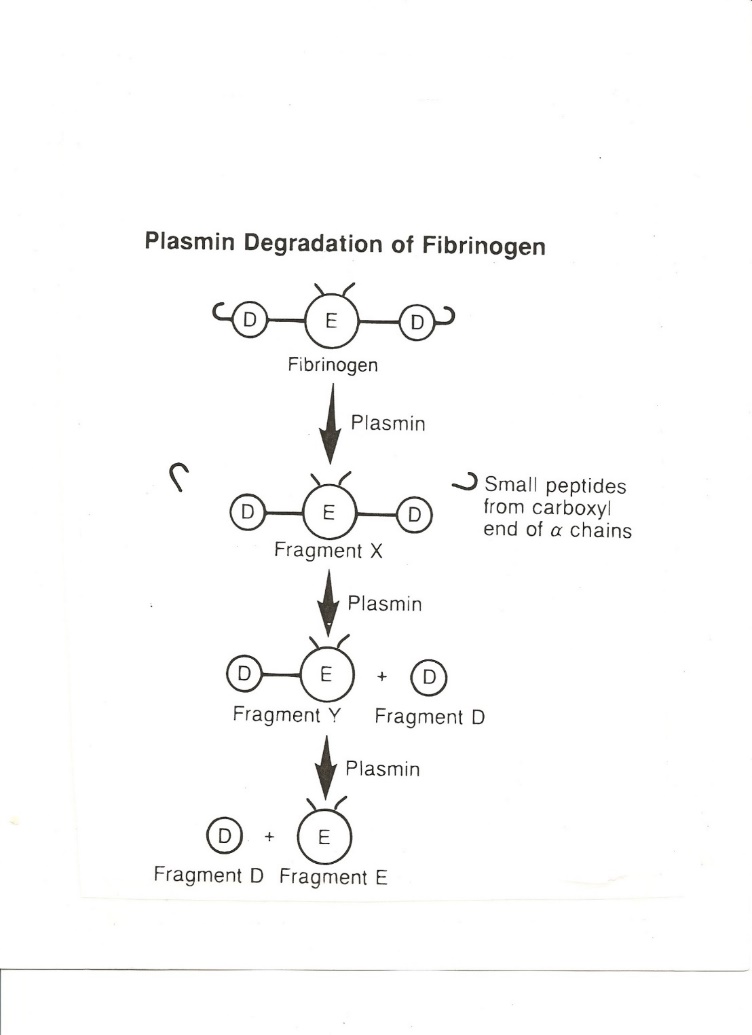 Fibrinolysis by plasmin
Fibrinolysis by plasmin
- Proteolytically degrades both fibrin and native fibrinogen in circulation.
- Products termed fibrin/fibrinogen degradation products (FDPs)
- Fragment X (still capable of clotting).
- Fragment X into Fragment Y + Fragment D
- Fragment Y into Fragment D + Fragment E
- Result = 2 D’s and 1 E
- D-Dimer specific to fibrin degradation (to rule out DIC, DVT, PE)
Fibrin Degradation Products FDPs
- Interfere with further thrombin–induced fibrin formation and increase vascular permeability
- Degrade V, VIII, XIIa, fibrinogen, fibrin, and GP1b
- Cleave C3 into fragments and activate complement
Kinin System
- Important in inflammation, vascular permeability, and chemotaxis
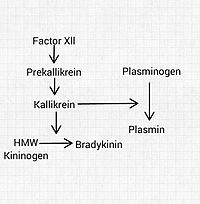 Involved in contact activation of intrinsic pathway
Involved in contact activation of intrinsic pathway- Prekallekrein circulates complexed to HMWK
- XIIa (+HMWK) activates prekallekrein to kallekrein and XI to XIa
- Kallekrein and XIa then reciprocally activate XII to XIIa in a feedback system that amplifies the reaction
- Kallekrein
- An enzyme, also activates plasminogen to plasmin
- Acts on HMWK to release bradykinin
- Increase vascular permeability
- Contract smooth muscle
- Dilate small blood vessels
- Induce inflammation and pain
- Release prostaglandins from tissues