Topic 2 - Learning Objectives - Membrane Transport & Potential
Define amphipathic and explain why phospholipids are amphipathic
amphipathic - molecules that contains hydrophobic and hydrophilic regions
a phospholipid contains:
a hydrophilic head - phosphate glycerol head
a hydrophobic tail - fatty acid tail
Explain why membranes are describes as fluid mosaic models
model stats that a membrane is a fluid structure with various proteins embedded in it
List two classes of phospholipids in animal cell membranes
phosphoglycerides - most abundant in animal cell membranes
Sphingolipids
Describe and identify general chemical structure of a phosphoglyceride
where a phosphate group links with a polar head group
phosphatidylethanolamine - a phosphate group links to a ethanolamine
phosphatidylcholine - phosphate group links to a choline
phosphatidylserine - phosphate group links to a serine
phosphatidylinositol - phosphate group links to inositol
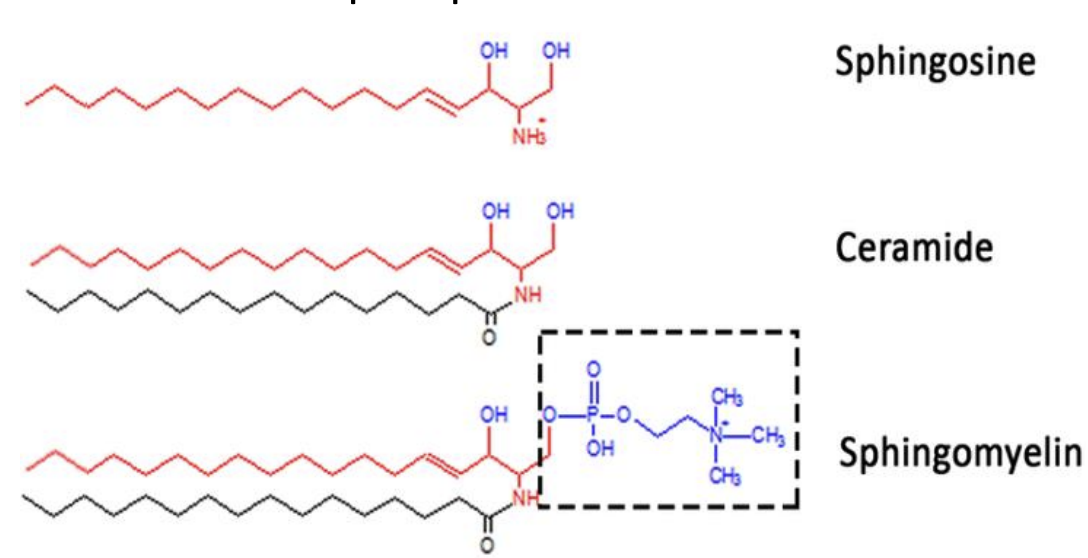
Describe and identify general chemical structure of a sphingolipid
ceramides
sphingomyelin
glycosphingolipids
Differentiate between general chemical structure of a phosphoglyceride and a sphingolipid
sphingolipids are chemically different but have similar 3D shape to phosphoglycerides
sphingolipids have:
sphingosine as backbone - instead of glycerol
sphingosine - an 18-carbon amino alcohol with an unsaturated hydrocarbon chain
one fatty acid chain - instead of two
fatty acid chain is bound to sphingospine with an amide bond - instead of an ester bond
highly enriched in cell membranes of the brain and nervous tissue
List (and identify the chemical structure of) the three types of sphingolipids
ceramides (simplest type) - where one fatty acid chain is attached sphingosine with an amide bond
sphingomyelin - ceramide bound with either phosphocholine/phosphoethanolamine
glycosphingolipids - cereamides with one or more sugar residues
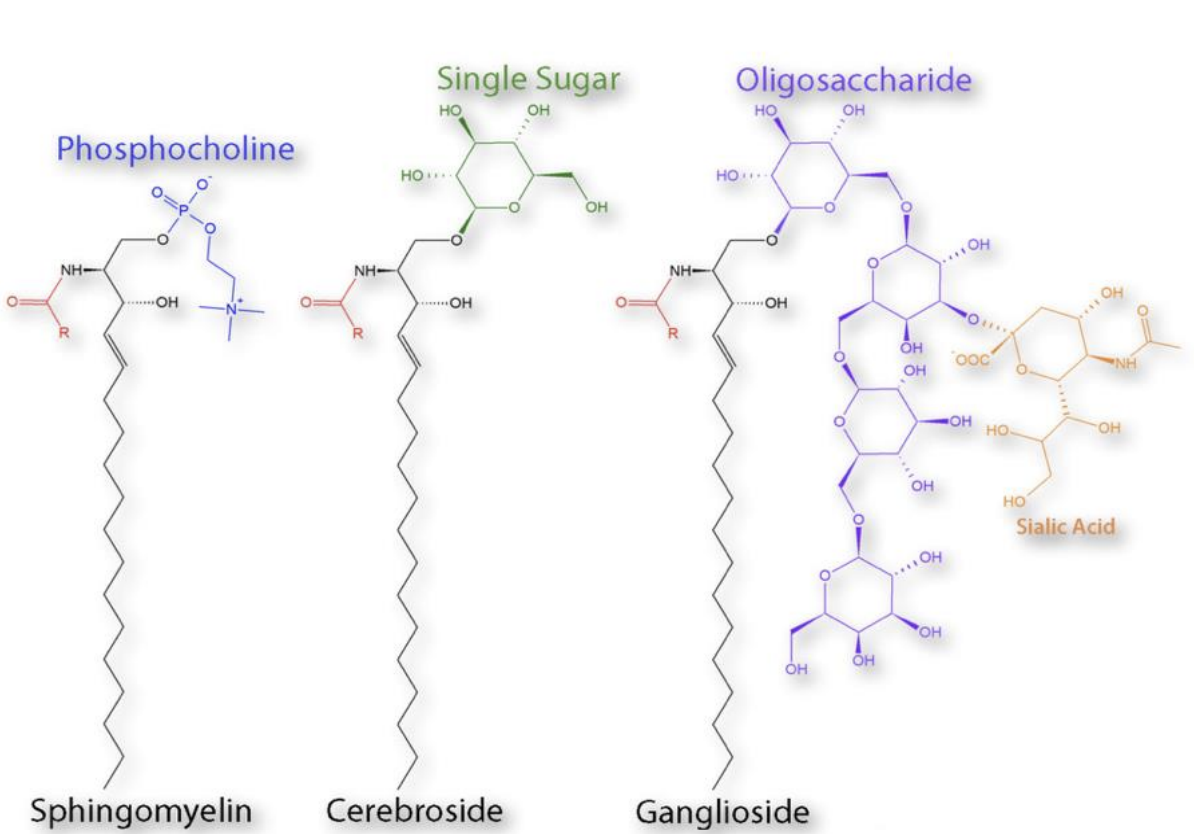
List (and identify the chemical structure of) the two common types of glycosphingolipids
types of glycosphingolipids:
cerebrosides - single glucose/galactose as sugar residues
gangliosides - at least three sugar as residues (one sugar must be sialic acid)
List and describe factors that can increase and decrease membrane fluidity
temperature - when temperature increases, membrane moves from a solid gel state to a more fluid state
fatty acid chains - shorter fatty acid chains are more fluid than those with longer fatty acid chains
shorter fatty acid chains means less surface area to allow for stabilizing van der Waal’s/hydrophobic interactions that occur between fatty acid chains of neighbouring phospholipid molecules
unsaturated fatty acid chains - membrane rich in unsaturated fatty acid chains are more fluid than those rich in saturated fatty acids
the kinds (bends) in unsaturated fatty acids prevent tight packing between phospholipids
Explain how cholesterol affects membrane fluidity
cholesterol acts as a fluidity buffer against temperature extremes
helping to maintain membrane integrity by preventing it from becoming too rigid in cold temperatures and too fluid in warm temperatures.
at warm temperatures - cholesterol restrains movement of phospholipids (cholesterol acts to decrease membrane fluidity)
at cold temperatures - cholesterol prevents tight packing between phospholipids (cholesterol acts to increase membrane fluidity)
*an increase in cholesterol decreases the membrane permeability - cholesterol limits the phospholipid’s movement and interaction
Describe lipid rafts
identify the components that are enriched in the structure of lipid rafts
cholesterol
sphingomyelin
gangliosides
phosphoglycerides with saturated fatty acid chains
explain why lipid rafts have reduced fluidity
due to all the components in the structure, they are tightly packed together which minimizes movement = fluidity
list examples of functions associated with lipid rafts
serves as an organizing centre for the assembly of signalling molecules
recruits actin cytoskeleton - allows for further stabilization of lipid raft
facilitates formation of transport vesicles
List the 6 major functions of membrane proteins
transport
enzymatic activity
signal transduction
cell-cell recognition
intercellular joining
attachment to the cytoskeleton and extracellular matrix
With respect to plasma membrane asymmetry
identify primary location (inner or outer leaflet of glycolipids)
glycolipids/glycosphingolipids - outer leaflet
identify primary location (inner/outer leaflet) of the 4 phosphoglycerides (PS,PI,PE,PC)
PS/PE/PI - inner leaflet
PC - outer leaflet (can also be found on inner leaflet, but are predominantly found on the outer leaflet)
state the two that have a net negative charge at physiological pH
PS/PI have a net negative charge
identify primary location of sphingomyelin
sphingomyelin - outer leaflet
List and explain important reasons for maintaining plasma membrane asymmetry
to create surface potential difference across membrane
can interact with positively-charged amino acid residues of membrane-bound proteins - keeps the proteins anchored to the membrane
peripheral membrane proteins (ie. proteins for cell signalling) contain positively-charged residues that can associate with negatively charged PS/PI
creates curvature within the membrane
preservation of cell viability
From the following list (PC, PE, PI, PS and the Sphingomyelin) state which are cylindrical shaped phospholipids and which are conical-shaped phospholipids
Cylindrical-shaped phospholipids:
Phosphatidylcholine (PC)
Sphingomyelin
Phosphatidylinositol (PI)
Phosphatidylserine (PS)
Conical-shaped phospholipids:
Phosphatidylethanolamine (PE)
Define concentration gradient, electrical gradient and electrochemical gradient
concentration gradients - differential concentrations of a substance across a space or a membrane
electrical gradient - a difference of charge across plasma membrane
electrochemical gradient - the combined gradients of concentration and electrical charge that affects an ion
List two types of passive transport and provide examples of molecules that use each type of transport
simple diffusion - where no transport proteins are needed
examples:
gases
small uncharged polar molecules (urea, water, and ethanol)
facilitated diffusion - transport proteins speed passive movement of molecules across the plasma membrane (ideal for polar molecules + charged ions)
List an define three main types of transport proteins that carry out facilitated diffusion
porins
large / barrel shaped
nonselective
moves hydrophilic molecules across the membrane based on size rather than charge
permeases
transports specific molecules across a membrane by binding to the molecule and undergoes conformational changes
permease can become saturated when all of the binding sites are occupied by molecules (leads to max transport rate)
ion channels
forms smaller pores where only specific ions may pass based on size and charge
typically gated
channels are specific to one/somtimes two ions
List and define the 5 types of ion channel proteins
gated ion channels
ligand-gated channels - open when specific regulatory molecules bind
voltage-gated channels - open/closes in response to different membrane potentials
mechanogated channels - regulated through interactions with subcellular proteins that make up the cytoskeleton
signal gated channels - opens/closes in response to a specific intracellular molecule
non-gated ion channels
leak channels - no trigger is required for their opening/closing
intrinsic rate of switching between open and closed states
Differentiate between primary and secondary active transport
primary active transport - involves permease carrier proteins that use an exergonic reaction (ex. ATP hydrolysis) to provide the energy to transport the molecule
secondary active transport - involves permease carrier proteins that move one molecule down its electrochemical gradient to help move another molecule against its concentration gradient
doesn’t use ATP directly - instead uses electrochemical energy
List the types of primary active transporters (provide specific examples for each)
P-type ATPases
Na+/K+ ATPases
Ca2+ ATPases
V-type ATPases
H+ ATPases
F-type ATPases
mitochondrial ATP synthase
ABC (ATP-binding cassette)
multi-drug resistance proteins - p-glycoproteins
Define uniporter, antiporter (exchanger), symporter (co-transporter)
uniporter (primary active transport) - a carrier that moves one specific ion/molecule
antiporter (secondary active transport) - a carrier that involved moving two different ions/molecules in opposite directions
symporter (secondary active transport) - a carrier that involves moving two different ions/molecules in the same directions
List examples of secondary active transporters that are symporters and antiporters
symporter
sodium glucose contrasporter protein
antiporter
sodium-calcium exchanger
Define Osmosis
the passive transport of water across a selectively permeable membrane
Define osmolarity
refers to number of particles in a solution
osmolarity takes into consideration of the TOTAL number of solutes (permeable and impermeable)
iso-osmotic - solutions are equal in osmolarity → no net osmosis occurs between them if they are separated by a selectively permeable membrane
hypo-osmotic - osmolarity in a solution is lower than that of another solution
hyper-osmotic - osmolarity solution is greater than that of another solution
Determine dissociation coefficient of a particular solute in a solution + calculate osmolarity
equation - osmolarity = sum of each solute (molarity x n)
where n is the coefficient of dissociation
Define tonicity
refers to what happens to a cell when placed in a solution
isotonic - solution with same concentration of non-penetrating solutes as that found in cells
hypertonic - solution with higher concentration of non-penetrating solutes than that found in cells
hypotonic - solution with lower concentration of non-penetrating solutes than that found in cells
Differentiate penetrating and non-penetrating solutes
penetrating solutes - substances that are permeable to the cell → can pass through the membrane
non-penetrating cells - substances that are not permeable to the cell → cannot pass through the membrane
Differentiate between osmolarity and tonicity
osmolarity - takes in consideration the total number of solutes (both permeable and impermeable)
tonicity - takes into account concentration only relating to impermeable solutes only
typically penetrating solutes will reach equilibrium → doesn’t effect tonicity