AP chem Unit 5: Kinetics
5.1: reaction rates
rate: ∆amount/∆time
the rate of a reaction can only be determined experimentally
avg. rate of reaction: ∆molarity/∆time
instantaneous reaction rate: slope of line tangent to curve
initial rate: rate for first interval on the graph

collision theory
in order for a reaction to take place, 2 things must happen
particles must collide with the right orientation
particles must collide with sufficient energy
factors that influence the rate of reaction
reactant concentration | higher molarity increases the chance of a collision |
temperature | higher temperature = particles are more likely to collide with energy greater than the activation energy(mostly not moving faster!!) |
surface area | higher surface area = higher chance of particles colliding |
catalysts | • homogenous catalysts provides an alternate pathway to complete the reaction that takes less Ea • heterogenous catalysts alter the orientation of the molecules so they form the correct orientation |
5.2: reaction rates
we can use experimental data to determine rate law
a rate law describes how the rate of reaction is proportional to the concentration of each reactant raised to a power
ex) D + 2C → AB
possible rate law: rate = k[D]^0[C]^1
rate constant: k, dependent on temp.
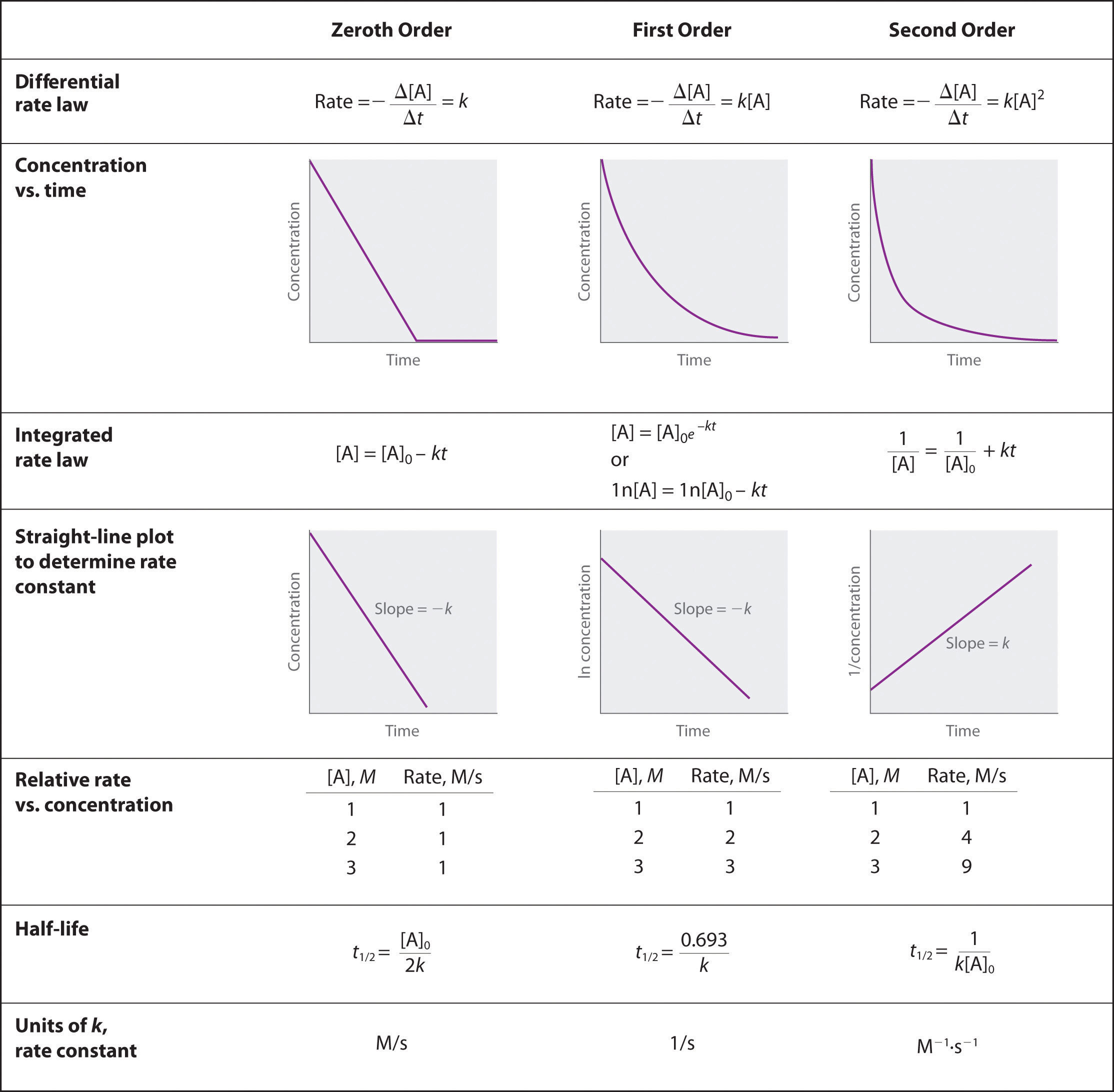
reaction order | unit for k | example |
0 | M/time | rate = k[A]^0 |
1 | 1/time | rate = k[A] |
2 | 1/(M*time) | rate = k[A]² rate = k[A][B] |
3 | 1/(M²*time) | rat |
n | 1/(M^(n-1)*time) |
5.3: concentration changes over time
graphing the concentration of a reactant vs time allows us to determine the reaction order
each order produces predictable curves
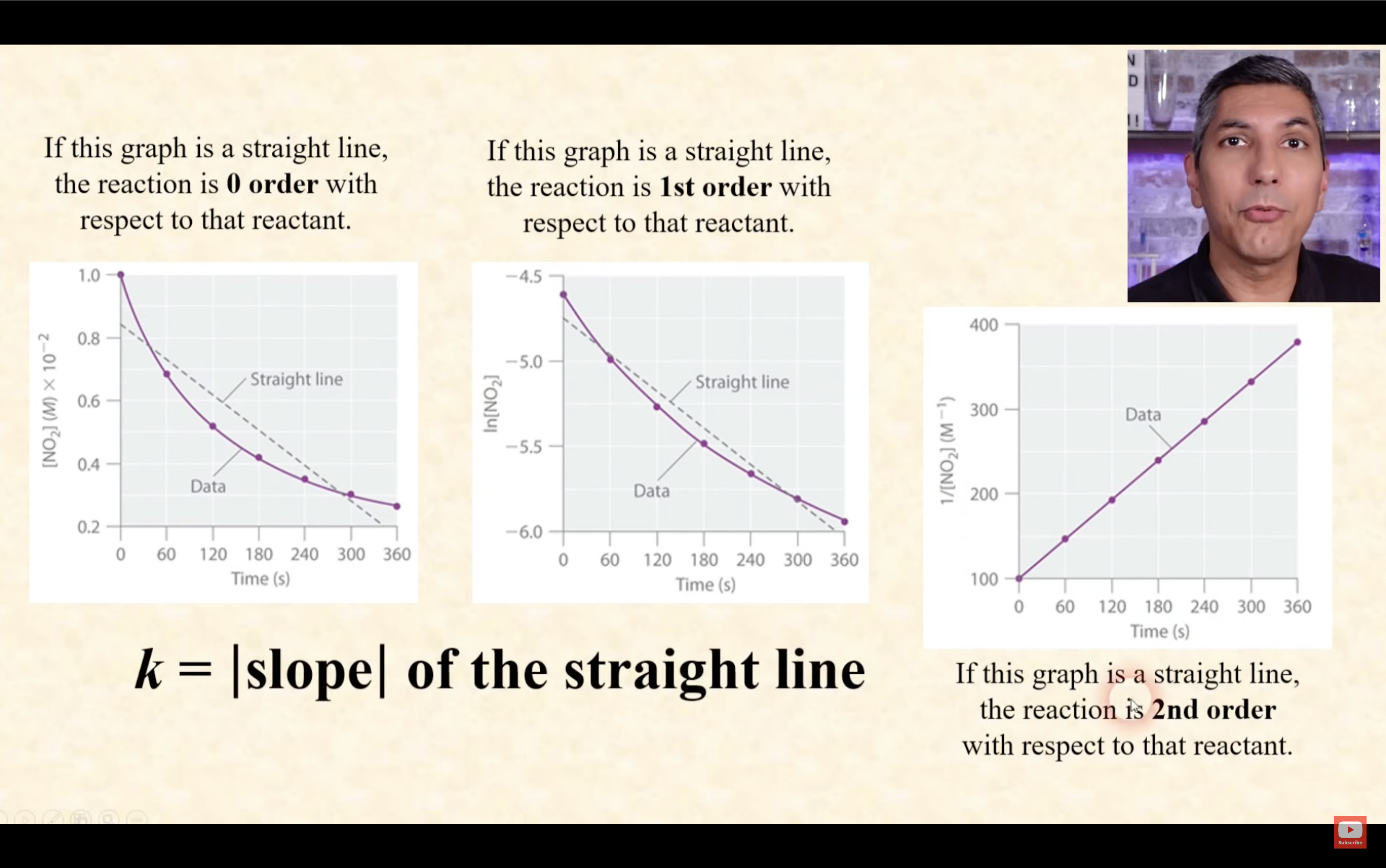
order | 0 | 1 | 2 |
half life decreases while [A] decreases | half life remains constant(independent of [A]) | half life increases while [A] decreases |
5.4: elementary reactions
an elementary step is a single step within the reaction mechanism
ex) 2NO → N2O2(rate = k[NO]²)
N2O2 + O2 → 2NO2(rate = [N2O2][O2])
2NO + O2 → 2NO2 cross out N2O2(intermediate)
intermediate: species that are neither reactants nor products, formed during the reaction and consumed by it
the rate law can be inferred from an elementary step based on the number of particles that collide
the slowest step is the rate determining step, limits how fast the reaction can go
5.5: collision model
collision theory explains why different reactions occur at different rates
particles must collide
they must have sufficient energy
they must be in the correct orientation
activation energy must be met in order to react(breaks bonds)- if there isn’t sufficient energy, the particles will bounce off each other
5.6: reaction energy profile
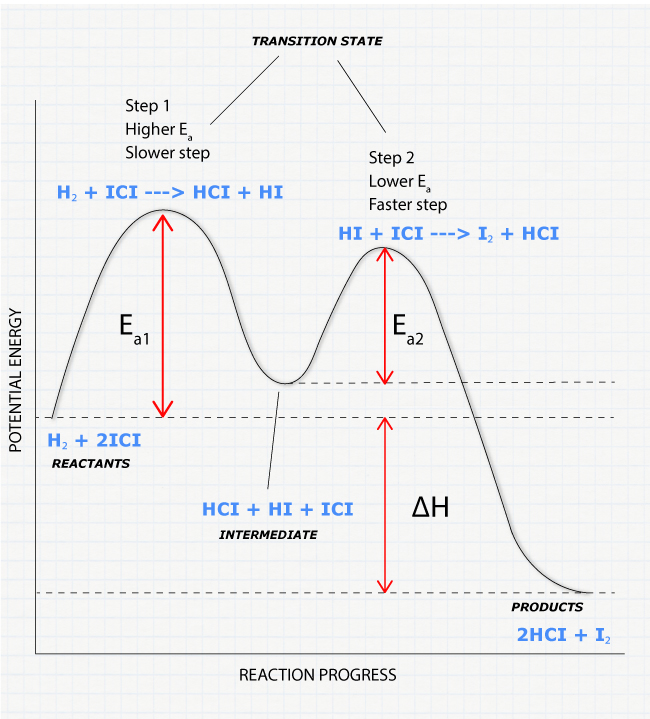
the transition state is where the bonds of the reactants are being broken and bonds in the products formed
*kinetic energy is proportional to temp.
a catalyst changes the reaction pathway
5.7: introduction to reaction mechanisms
in reactions involving multiple steps, the overall reaction occurs in a series of reactions
5.8: reaction mechanism and rate law
5.9: steady state approximation
when the first step of a reaction is fast, we approximate the reaction rate
5.10: multistep reaction energy profile
5.11: catalysts