NOMENCLATURE OF ORGANIC NOMENCLATURE OF ORGANIC COMPOUNDS AND ISOMERISM
Naming of branched hydrocarbon (1 branch)
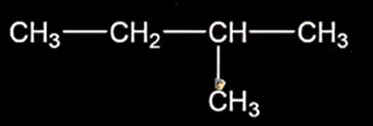
The longest chain containing maximum number of carbon atoms should be considered as the main chain and the remaining as branches.
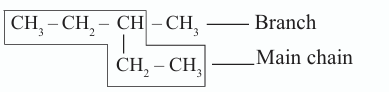
Each branch is named as an alkyl group, and its position is indicated by the number of the carbon atom to which it is attached. use the lowest number
Name of alkyl group = Word root corresponding to the number of carbon atom / atoms in the branch + ‘yl
full writing = Position number of branch+ hyphen + name of alkyl group + word root + suffix (ane)
(2 branch) Naming
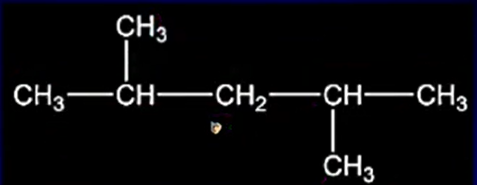
If more than one branch is present, select the longest carbon chain. Number the carbon atoms from left to right or right to left in such a way that the carbon atoms with branches get the lowest position numbers.
If the same branch appears more than once in a carbon chain, the number of branches is to be indicated using prefixes like di (two), tri (three), tetra (four) etc. Position numbers should be separated by commas.
FUCTIONAL GROUP
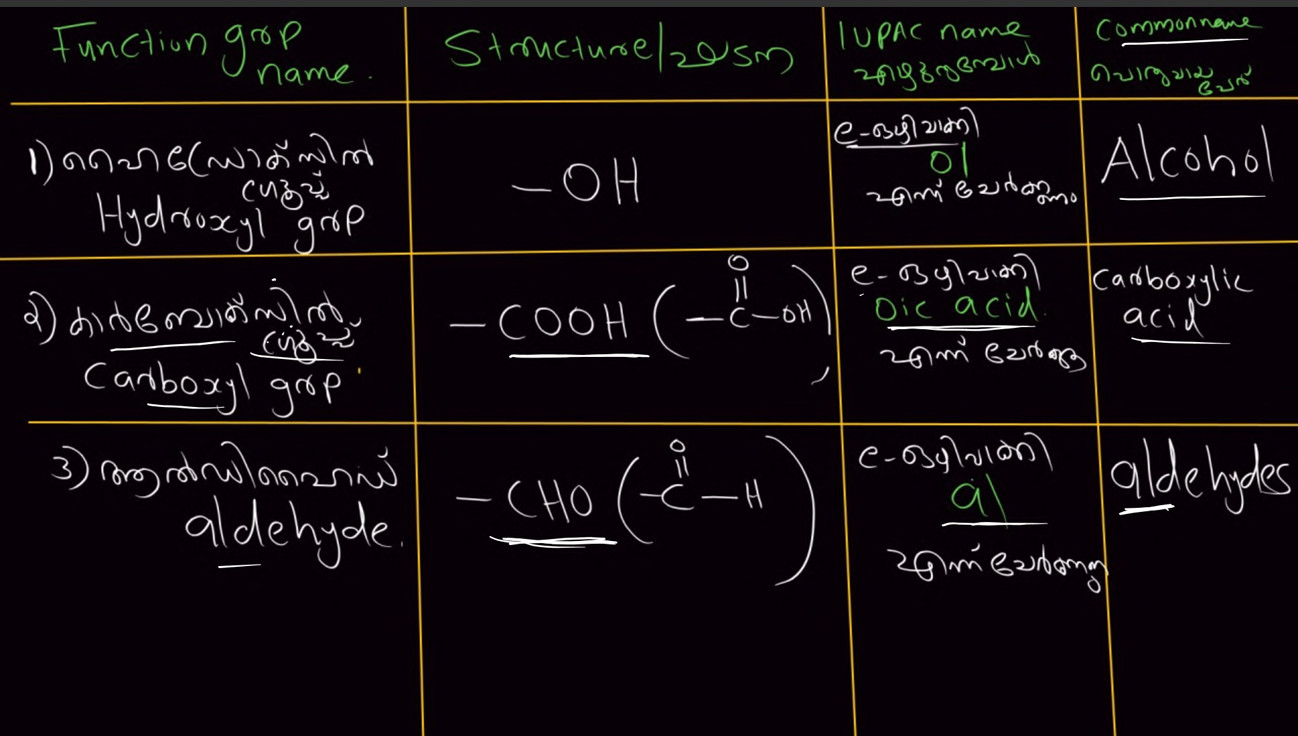

Function grp should be in the main chain or attached into the main chain
Function grp should have small number while counting
ISOMERS / ISOMERISM
Compounds having same molecular formula but diffrent chemical and physical properties are called isomers
the phenomenon is called isomerism
4 types isomerism
chain [compounds with same molecular formula but possess a diffrence in the chain structure ]
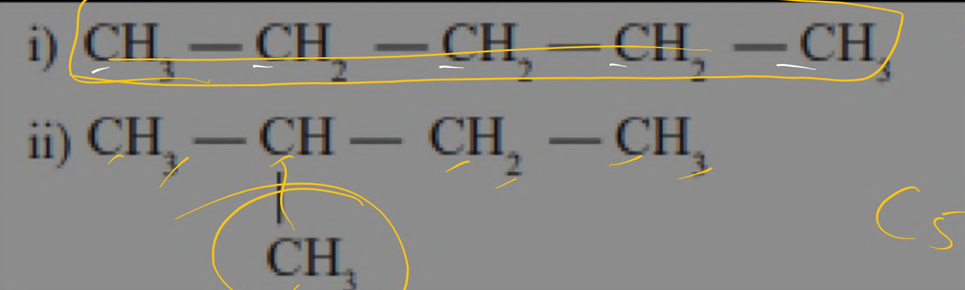
position [ position of the functional grp is differ in 2 compounds having same molecular formula and functional grp]
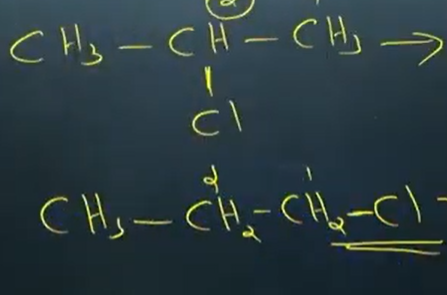
functional grp [ compunds having same molecular formula , but having a differ in their functional grp ]

metamerism ( CO , O )
It will only affect CO , O
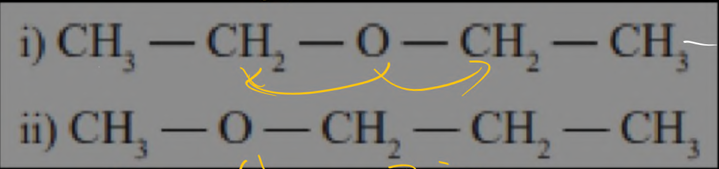
here only diffrence is the number diffrence to the left and right of O .that is metamerism
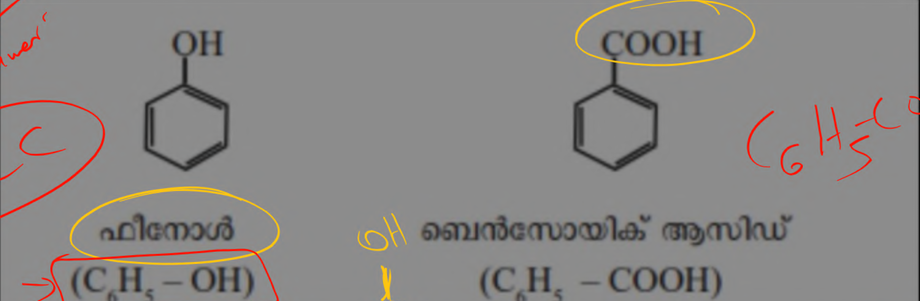
Aromatic hydrocarbon ( benzene )
cyclic compounds having their own aroma . benzene have industrial importance [ molecular formula = C6H6 ]